Long term outcome and fusion rate of transdiscal fixation for L5-S1 high grade spondylolisthesis
Publicado en Clinical Neurology and Neurosurgery (2021)
El Dr. R. García de Sola publica sus propios resultados en una técnica de fijación-fusión en las Espondiloistesis de alto grado.
Fue una idea brillante de otros autores (Abdu, et al., Spine,1994), que es posible llevarla a cabo con buen resultado y mínimo riesgo, gracias entre otras cosas a la aplicación de un adecuado sistema de imágenes 3D preoperatorio, Neuronavegador (BrainLab), Arco de Rx motorizado (Ziehm), control neurofisiológico durante la intervención quirúrgica. Así como la utilización de un instrumental especial para implantar los tornillos (BrainLab).
Long term outcome and fusion rate of transdiscal fixation for L5-S1 high grade spondylolisthesis
Juan Delgado-Fernández a,*, Natalia Frade-Porto b, Guillermo Blasco b, Patricia González-Tarno b, Ricardo Gil-Simoes b, Paloma Pulido Rivas b, Rafael García de Sola c
a Department of Neurosurgery, University Hospital 12 de Octubre, Madrid, Spain
b Division of Neurosurgery, Department of Surgery, University Hospital La Princesa, Madrid, Spain
c Department of Neurosurgery, Innovation in Neurosurgery, Universidad Autonoma de Madrid, Hospital del Rosario, Madrid, Spain
ARTICLE INFO
Abbreviations: EQ-5D, Euro Quality of Life; HGS, High grade spondylolisthesis; ODI, Oswestry Disability Index; VAS, Visual Analogic Score.
* Correspondence to: Division of Neurosurgery, University Hospital 12 de Octubre, Avda de Co´rdoba S/N, 28041 Madrid, Spain.
E-mail address: juan.delgado.fdez@gmail.com (J. Delgado-Fernndez).
https://doi.org/10.1016/j.clineuro.2021.106898
Received 19 June 2021; Received in revised form 2 August 2021; Accepted 17 August 2021
Available online 20 August 2021
0303-8467/(c) 2021 Elsevier B.V. All rights reserved.
Supplementary material related to this article can be found online at doi:10.1016/j.clineuro.2021.106898.
ABSTRACT
Objective: High grade spondylolisthesis (HGS) is a quite rare entity and many techniques are available to address this condition. In 1994 Abdu et al. proposed a transdiscal fixation approach that achieved a good clinical outcome. We analyse outcome and fusion achieved in patients treated by transdiscal fixation after 1-year follow-up.
Methods: We reviewed patients operated through transdiscal fixation since 2014 with a follow-up of at least 1 year, and compared preoperative and postoperative clinical measures (ODI, VAS and EQ-5D) and postoperative complications. Also, we analyzed the degree of fusion on CT scan with Lenke and Birdwell criteria.
Results: Twelve patients were included in the study with a mean follow-up of 49.4 months (range 12.8-84.1 months). Three cases presented a Meyerding grade IV spondylolisthesis and 9 cases grade III. At 1-year follow-up mean postoperative ODI, VAS and EQ5D scores improved (ODI 13.2 (range 0-30) vs 49.83 (range 15-71.1); p = .005). Equally this improvement was seen in the last follow-up (ODI 9.28 (range 0-35) vs 49.83 (range 15-71.1); p = .005). CT scan showed fusion grade A in 5 patients (41.6%), another 5 as grade B (41.6%) in Lenke classification. According to the Birdwell criteria 4 patients were classified as grade I (33.3%), 7 patients grade II (58.3%). None showed complications postoperatively or radiolucency in follow-up.
Conclusions: Transdiscal fiXation shows a good clinical outcome that is maintained throughout a long time period and provides a reliable and suitable fusion
Keywords:
fusion rate high grade spondylolisthesis long term outcome neuronavigation transdiscal screws
Dr. R. García de Sola publishes his own results on a fixation-fusion technique in high-grade spondyloarthroplasty.
It was a brilliant idea of other authors (Abdu, et al., Spine,1994), which is possible to carry out with good results and minimum risk, thanks among other things to the application of an adequate preoperative 3D imaging system, Neuronavigator (BrainLab), motorised Rx arc (Ziehm), neurophysiological control during surgery. As well as the use of special instruments to implant the screws (BrainLab).
Video del caso clínico
1. Introduction
High grade spondylolisthesis (HGS) is the slippage of more than the 50% of a vertebral body relative to an adjacent one as defined by Meyerding classification. The first description of a spondylolisthesis clinical syndrome was done in 1782 by HerbiniauX, a Belgian obstetri-cian that reported a prominence anterior to the sacrum that created an impediment to vaginal delivery [1]. Although spondylolisthesis is a relatively common issue in a neurosurgical department, with an incidence of 4.2-11.2% in the general population [2-4], HGS is a quite rare entity accounting for only 19% of spondylolisthesis cases [5]. This fact, has made it difficult to standardize a common procedure for its treat-ment. There are some aspects that should be address when a neurosur-geon faces a patient with HGS. Firstly, there is a major consensus that patients with HGS should undergo surgery to achieve fusion of the space and prevent further progression of the anterior displacement. Secondly, in those cases where the spine is balanced in situ fusion is accepted to prevent neurological damage during slippage reduction that could lead to neurological deficit in 5-45% of patients [1,6-10].
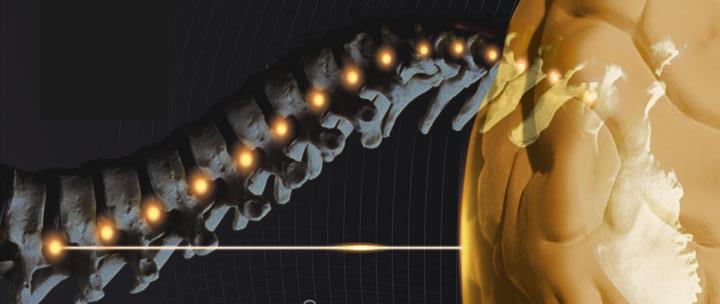
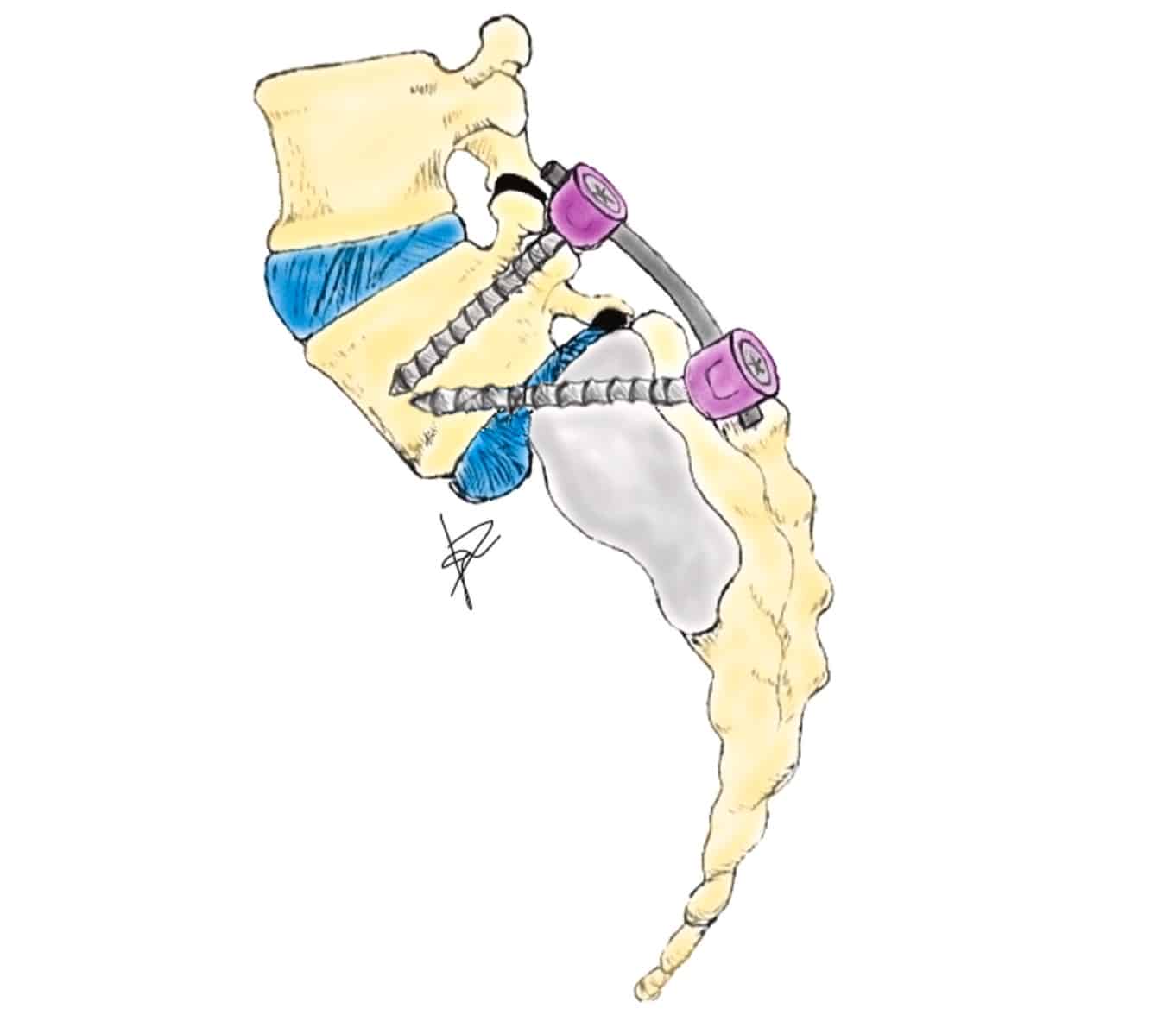
Nevertheless, there is an important controversy about the rate of pseudoarthrosis inthese patients. If in situ fusion is performed without reduction, pseu-doarthrosis rate was reported to be around 15-44% in the oldest series [1,10,11], whereas with the new fiXation systems and anterior techniques pseudoarthrosis rate has declined [12]. Moreover, hardware failure probability increases if the surgical procedure does not include the anterior spinal column. There are several reasons that could explain this increase. Firstly, axial compressive forces are transmitted through the rod to the system creating micro-instability that inhibits fusion and osteogenesis. Secondly, shear forces in the interspace are strong and not recent years different articles have demonstrated biomechanically that limited enough with posterolateral fiXation, especially in patients this construct could be 1.6-1.8 times stiffer than pedicular screws in a suffering from HGS[10]. Many techniques have been proposed to address this condition, both anterior and posterior approaches, trans-discal pedicular screws, Bohlman technique with fibular graft or circular titanium cage, vertebrectomy with sacral dome removal or ALIF and circumferential constructs, among others. However, as Minamide et al. stated we consider that the ideal strategy for HGS must cover the following four aspects [13]. Firstly, it should be performed by a single approach, in order to avoid increasing morbidity; secondly, it should be easy to perform and the surgeon should be familiar with the procedure; thirdly it should try to avoid manipulation of nerve roots and dural sac, and finally the system must resist shear forces in the lumbosacral space, as we have stated previously. These are the reasons why since the description by Abdu et al. [14] of pedicular transdiscal fiXation, our team has been treating HGS with this technique (Fig. 1). We believe that this technique fulfils all the items described above. Furthermore, in HGS model, and at least similar to an interbody plus pedicle screws fiXation [13,15,16].
A lack of evidence remains in the literature regarding long-term outcome and fusion rate of transdiscal fiXation. Therefore, the aim of the present study was to analyse these parameters in our cohort of HGS patients treated by transdiscal fiXation with long follow-up.
2. Materials and methods
2.1. Patient data
We retrospectively reviewed those cases operated with transdiscal L5-S1 fiXation in our institution since 2014-2019, with a follow-up period of at least 1 year. Our local institutional review board approved the study and all patients signed an informed consent form for surgery and further investigations related to their pathology. We included patients over 18 years with correct sagittal balance and Meyerding III or IV spondylolisthesis, with chronic low back pain and/or radiculopathy. In some cases, fiXation included L4 if there were signs of L4-L5 instability. Clinical and demographic characteristics of all patients were recorded including age, sex, symptoms, preoperative pain and medication, and grade of spondylolisthesis. Pain level was evaluated as primary outcome measure with the Oswestry Disability Index (ODI), in which highest values indicate more pain [17]. We also evaluated as secondary outcomes low back pain with visual analogic score (VAS), and the quality of life with EuroQol (EQ-5D) [18], where higher number indicate better quality of life. Preoperative values of ODI, EuroQol and VAS were recorded and compared with those obtained at 1 year of follow-up. Pre and postoperative medication were recorded for every patient. Telephonic assessment was carried out again at the last follow-up before this study started.
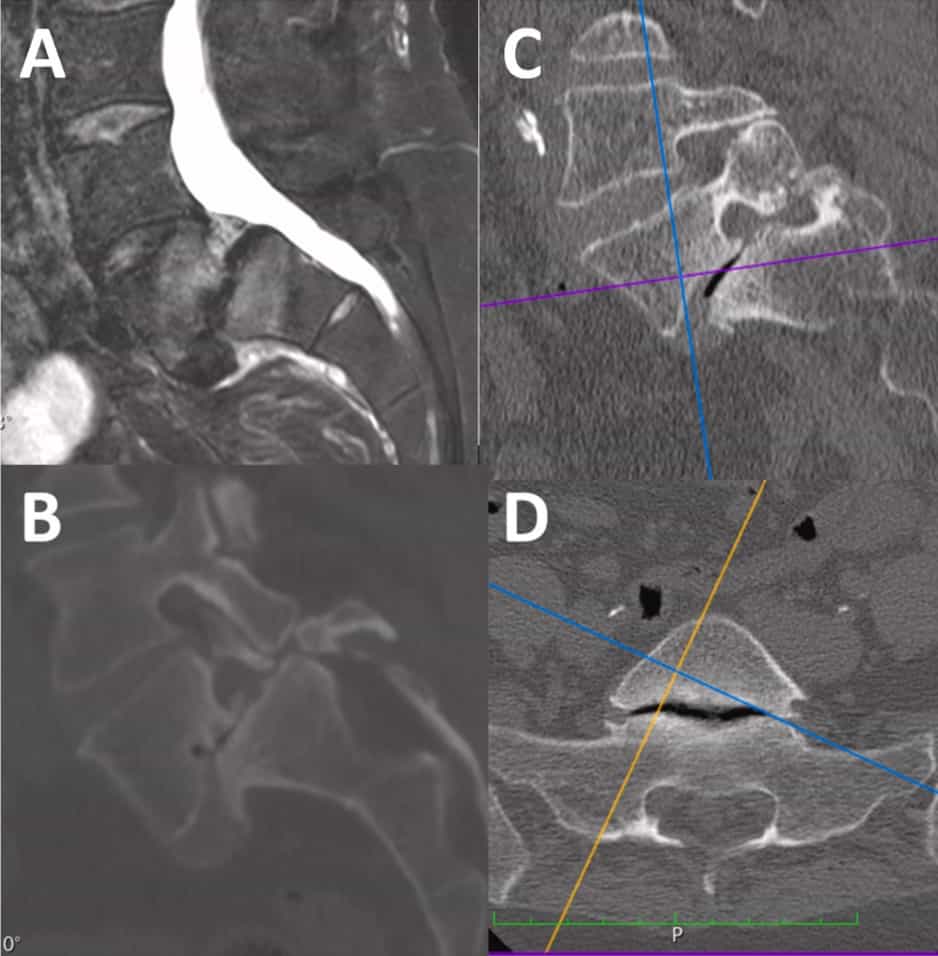
Radiological preoperative study included spine MRI and CT together with flexo-extension lateral lumbar spine X-ray in all cases. Standing X-rays were obtained preoperatively to analyse spinal balance according to parameters defined by Dubousset [19] for slip angle and Labelle et al.[20] for pelvic tilt, sacral slope and pelvic incidence. During surgery, intraoperative CT spine was performed to asses correct positioning of instrumentation screws. Postoperatively, almost all patients had a spine CT scan at least at 1-year follow-up, and in most of the cases later on to assess the degree of fusion. Fusion grade was determined following the Lenke classification [21] and Bridwell criteria [22] on these CT scans.
As a retrospective study there was no need for a specific consent form for patient enrollment in the study.
2.2. Surgical technique
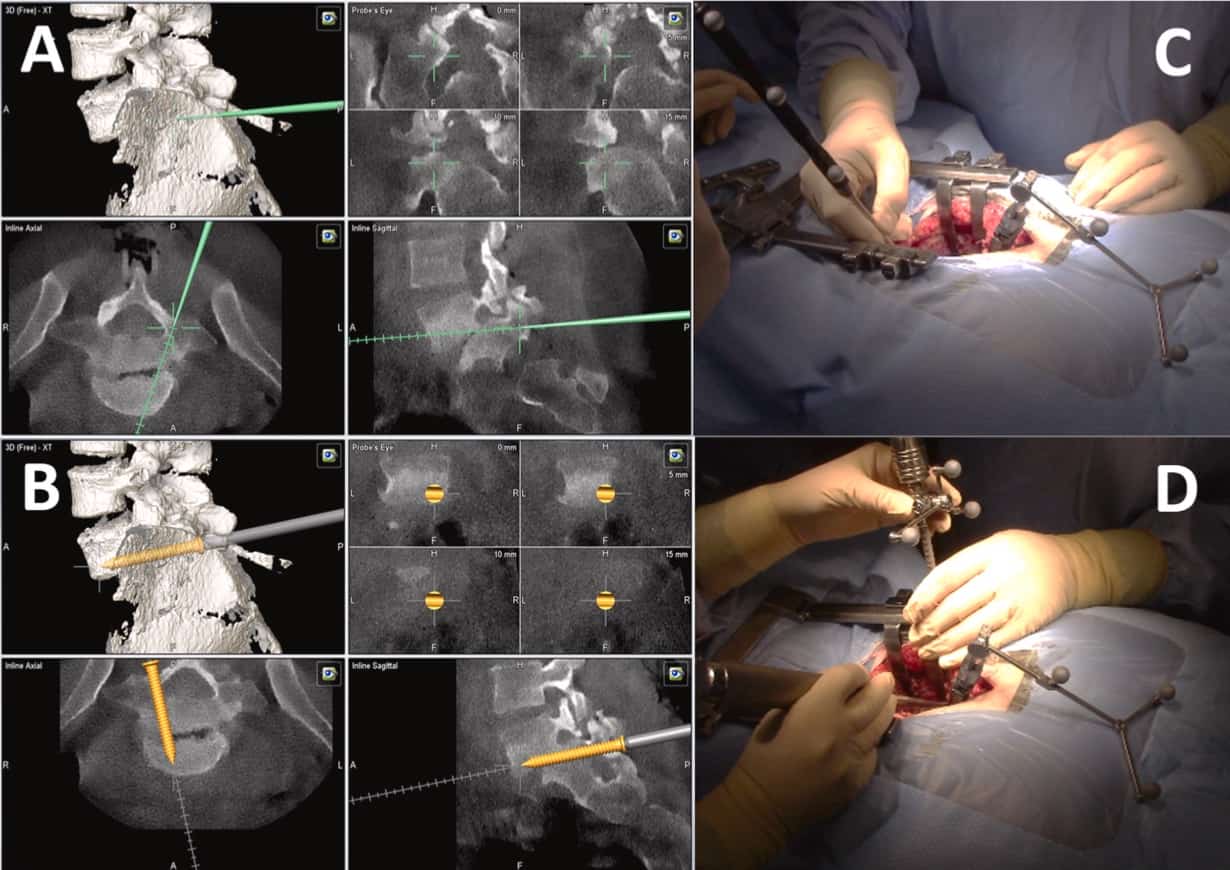
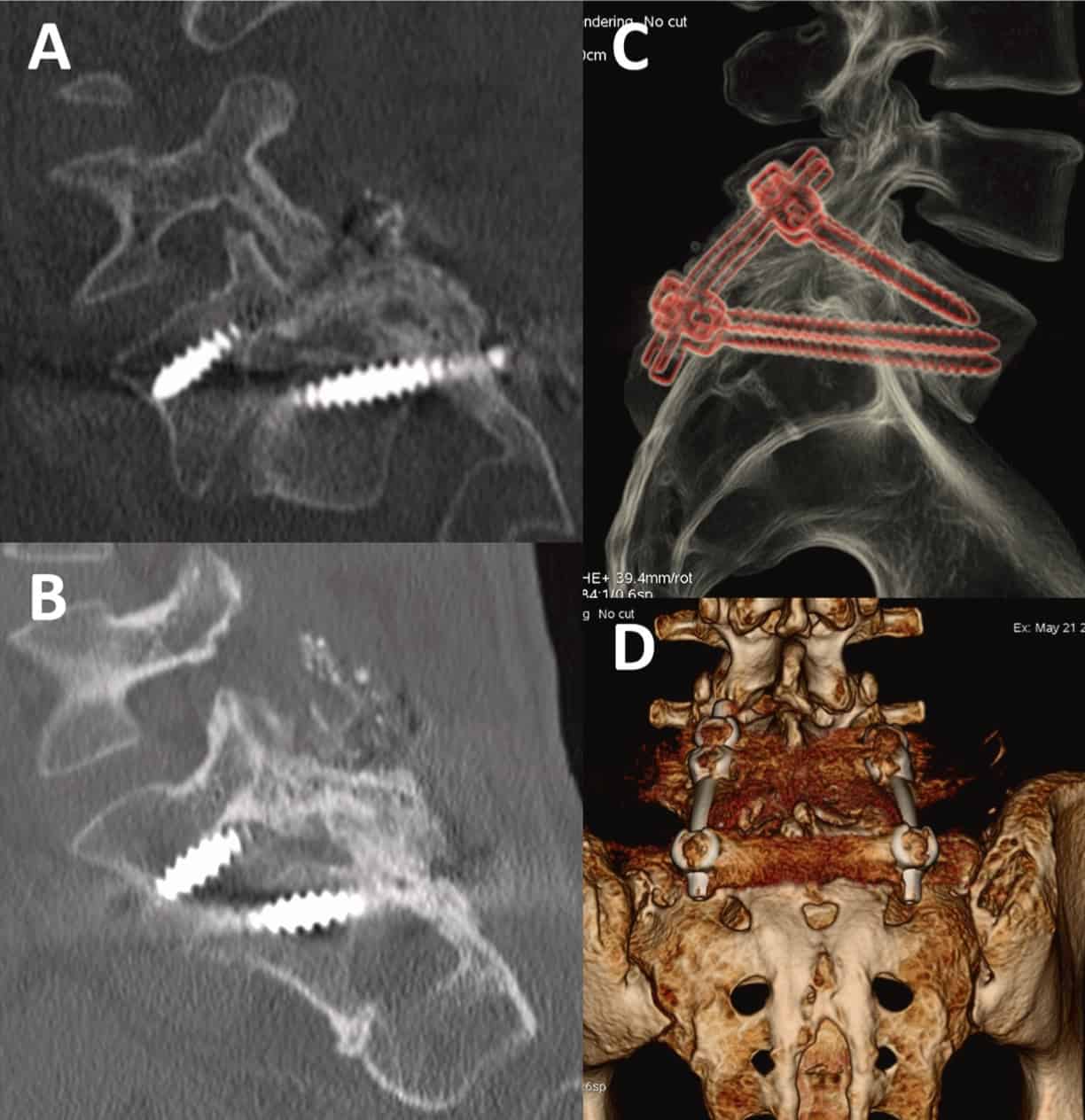
Preoperatively, patient images were analyzed with OsiriX DICOM Viewer to plan the correct insertion of the transdiscal screws, checking that they will have a correct positioning through S1 pedicles, interver-tebral disc and reaching L5 vertebral body (Fig. 2). In the operating room, patients were placed on a Jackson-type radiolucent surgical table, and in all cases spine neuronavigation was used. The surgical technique was performed as previously described [14,23] (Video). After radio-scopic location of L5-S1 space, a midline incision is made and sub-periosteal muscle dissection is carried out until exposing the transverse processes of the L5-S1 vertebrae. Once the sacrum is exposed, neuro-navigation array is fiXed to the spinous process of S2 or S3.
Then an intraoperative CT scan (Zhiem Vision RFD 3D, Zhiem Imaging, Nur-emberg, Germany) is performed and after obtaining images a 3D reconstruction is generated with the Brainlab spinal navigation system (Brainlab, Germany). Next, the drill guide, tap, and screwdriver are calibrated. At L5 and L4 levels, transpedicular screws are placed ac-cording to the usual technique. For the introduction of transdical screws, the entry point is located at the level of S1 and the drill-guide is placed with the help of the neuronavigator. Once placed, the trajectory is planned with an anteromedial direction through the pedicle of S1, S1 vertebral body, reaching the intervertebral disc that is traversed until the inferior endplate of the L5 vertebra, which is perforated, thus ac-commodating the screw in this vertebral body. Once the trajectory described has been planned and drilled with the bit, the next step is to insert the guided tap to increase the trajectory and secure it before inserting the screw. Afterwards, a previously calibrated screw (7.5 mm in diameter and 60-70 mm in length; Zodiac, Alphatec Spine) was placed with imaging guidance. When all the screws are placed, a new 3D fluoroscopic scan is performed to ensure that they are correctly posi-tioned. Finally, decompression of the L5-S1 space is performed through a hemilaminectomy and foraminotomy of the affected root. Posterolateral fusion with autologous iliac crest bone and heterologous bone graft substitute was carried out (Fig. 3).
2.3. Statistical analysis
IBM SPSS 21.0 (IBM Corp.) was used for performing the statistical analysis. Preoperative and postoperative clinical data were compared with a non-parametric WilcoXon signed-rank test. The threshold for statistical significance was set at p-values ≤ 0.05.
3. Results
Since the beginning of this retrospective study in 2014, 12 patients underwent surgery for HGS in our hospital. Two patients were male, and the other ten patients were female (1/5 ratio). Mean age of patients at the date of surgery was 48 years (range 37-63). Patients mainly presented with low-back pain during standing with radiation to the lower extremities. In half of the patients this pain was accompanied with unilateral radiculopathy of L5 or S1 root. Mean preoperative ODI score was 49.83 (range 15-71.1), mean preoperative VAS score was 6.58 (range 5-9) and mean preoperative EQ5D score was 0.307 (range 0.031-0.85).
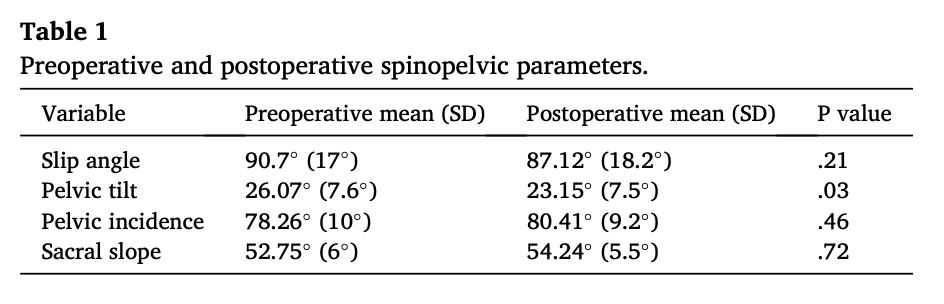
Preoperative MRI and CT demonstrated a Meyerding IV grade spondylolisthesis in 3 cases and grade III in 9 cases. As previously described, 5 patients were subjected to L4-S1 instrumented fusion due to instability signs in the preoperative MRI or flexo-extension X-ray im-ages. In the other 7 patients only the L5-S1 segment was fiXed. Pre and postoperative spinopelvic parameters at 1 year follow-up are described in Table 1.
Mean surgical time was 210 min (range 170-280) without any intraoperative complications. Intraoperative CT was performed in every case, as described previously in methods section. There was no need of repositioning any of the screws. Postoperative course of patients was uneventful, with the exception of a case of a urine infection. None of the patients needed transfusion. Mean time for hospital discharge was 5.58 days (range 4-7).
All patients were followed-up on clinics after surgery. At 1-year follow-up a CT was performed in all patients except 2. In these 2 cases
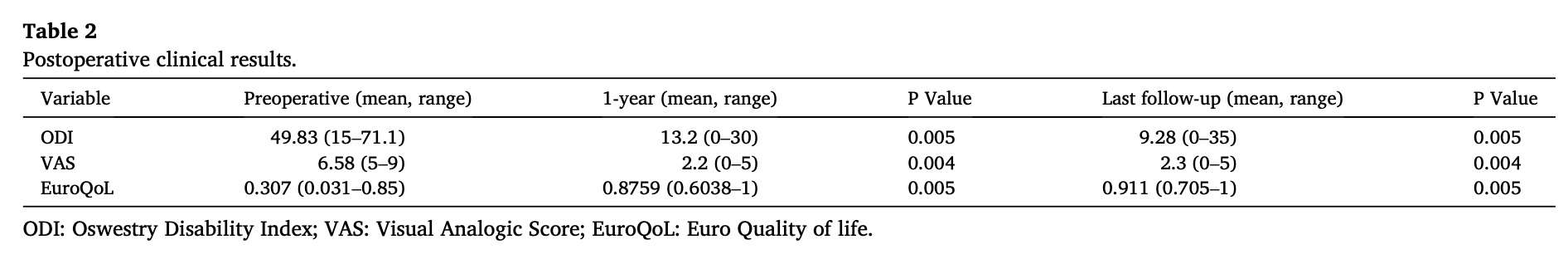
the last CT was performed 6 months after surgery. Overall mean follow-up time was 54.4 months (range 18.8-90.1 months). At 1-year of follow-up mean postoperative ODI score was 13.2 (range 0-30), mean post-operative VAS score was 2.2 (range 0-5) and mean postoperative EQ5D score was 0.8759 (range 0.6038-1). When we compared preoperative clinical data to postoperative 1-year clinical scores there was a clinical difference with an improvement in the three parameters 1 year post-operative ODI 13.2 (range 0-30) vs preoperative ODI 49.83 (range 15-71.1); p = .005; 1 year postoperative VAS 2.2 (range 0-5) vs pre-operative VAS 6.58 (range 5-9); p = .004; 1 year postoperative EQ5D 0.8759 (range 0.6038-1,) vs preoperative EQ5D 0.307 (range 0.031-0.85); p = .005. Mean follow-up at last telephone review of pa-tients was 49.44 months (range 12.8-84.1 months). At that moment, mean postoperative ODI score was 9.28 (range 0-35), mean post-operative VAS score was 2.3 (range 0-5) and mean postoperative EQ5D score was 0.911 (range 0.705-1). Also, when we compared preoperative and postoperative values of scores, we found a statistically significant difference in every score, last postoperative ODI 9.28 (range 0-35) vs preoperative ODI 49.83 (range 15-71.1); p = .005; last postoperative VAS 2.3 (range 0-5) vs preoperative VAS 6.58 (range 5-9); p = .004; last postoperative EQ5D 0.911 (range 0.705-1,) vs preoperative EQ5D 0.307 (range 0.031-0.85); p .005 (Table 2).
Clinically, 7 patients (58.3%) are pain free with no need of pain-killers, 3 patients have reduced their pain medication intake, and just two patients have not reduced their pain treatment. In one case, the patient needed further treatment from the pain clinic. Four patients have returned to their job and most of them (9 patients, 75%) have increased their physical exercise.
Mean time from surgery to postoperative CT scan to asses fusion was 19.73 months (range 6.33-53.23). Five patients were classified as Lenke grade A (41.6%), other 5 patients as grade B (41.6%), and only 2 pa-tients as grade C (16.6%). In the Birdwell criteria 4 patients were clas-
sified as grade I (33.3%), 7 patients were grade II (58.3%), and only 1 patient grade III (8.3%) (Table 3). None of the cases showed signs of radiolucency, hardware failure or breakage of the instrumentation sys-tem (Fig. 4).
4. Discussion
There are several studies addressing the treatment of HGS, but most of them have a limited number of patients and a limited prospective follow-up. Moreover, most of the literature that explores this topic miX many patients treated with different approaches and techniques, making it difficult to obtain a clear conclusion [1,24,25]. However, there is a general agreement about the need of surgical treatment for this condi-tion when conservative treatment has failed as patients can develop pain and neurological deficit over time [1,24]. Nevertheless, the adequate surgical technique and approach is still controversial and there is a lack of evidence-based knowledge about how to proceed in these patients [25,26]. Furthermore, the fusion obtained after surgical treatment of HGS has been ignored in many studies, and only the presence of pseu-doarthrosis has been more frequently described [10,27,28]. This lack of evidence led us to perform one of the longest retrospective reviews of posterior transdiscal fiXation to date, with a special interest in the fusion rate obtained with this procedure.

One of the largest studies about HGS management is the review published by Kasliwal et al. [6] of patients from the Scoliosis Research Society database. Although the sample size of this study was large (77 HGS patients) has the inconvenience of including different techniques and approaches, and the rate of pseudoarthrosis was not reported. Nevertheless, fusion rate was clearly addressed by Rindler et al. [29], in a systematic review of the evidence published about transsacral surgery approach to HGS. This study evaluated the technique, the reduction and the fusion obtained showing a low rate of pseudoarthrosis. All patients presented good fusion, except those cases that did not report the fusion status, and it should be highlighted that only around a quarter of patients underwent reduction of the slippage. Moreover, almost all pa-tients (94.7%) showed some improvement or at least average or better functional outcome after surgery [29].
Fusion analysis in our patients showed that 10/12 (83.3%) had a good consolidation assessed by Lenke and Bridwell criteria, showing at least a unilateral solid fusion stout or partial incorporation of the graft. In addition, we did not find any case of pseudoarthrosis in our series, even though there is some evidence that in situ fusion increases the risk of higher non-union incidence [1,11]. However, we decided to perform in situ fusion taking into account that there is a general consensus that in patients with a balanced spine is an adequate technique [30,31]. Furthermore, some studies have demonstrated that there is not a clear relation between reduction and clinical improvement [11,26], whereas a higher risk of neurological complications is associated with reduction, as shown by Kasliwal et al. [6] who described that these are the most common complication reaching an 11.5% incidence during HGS surgery.
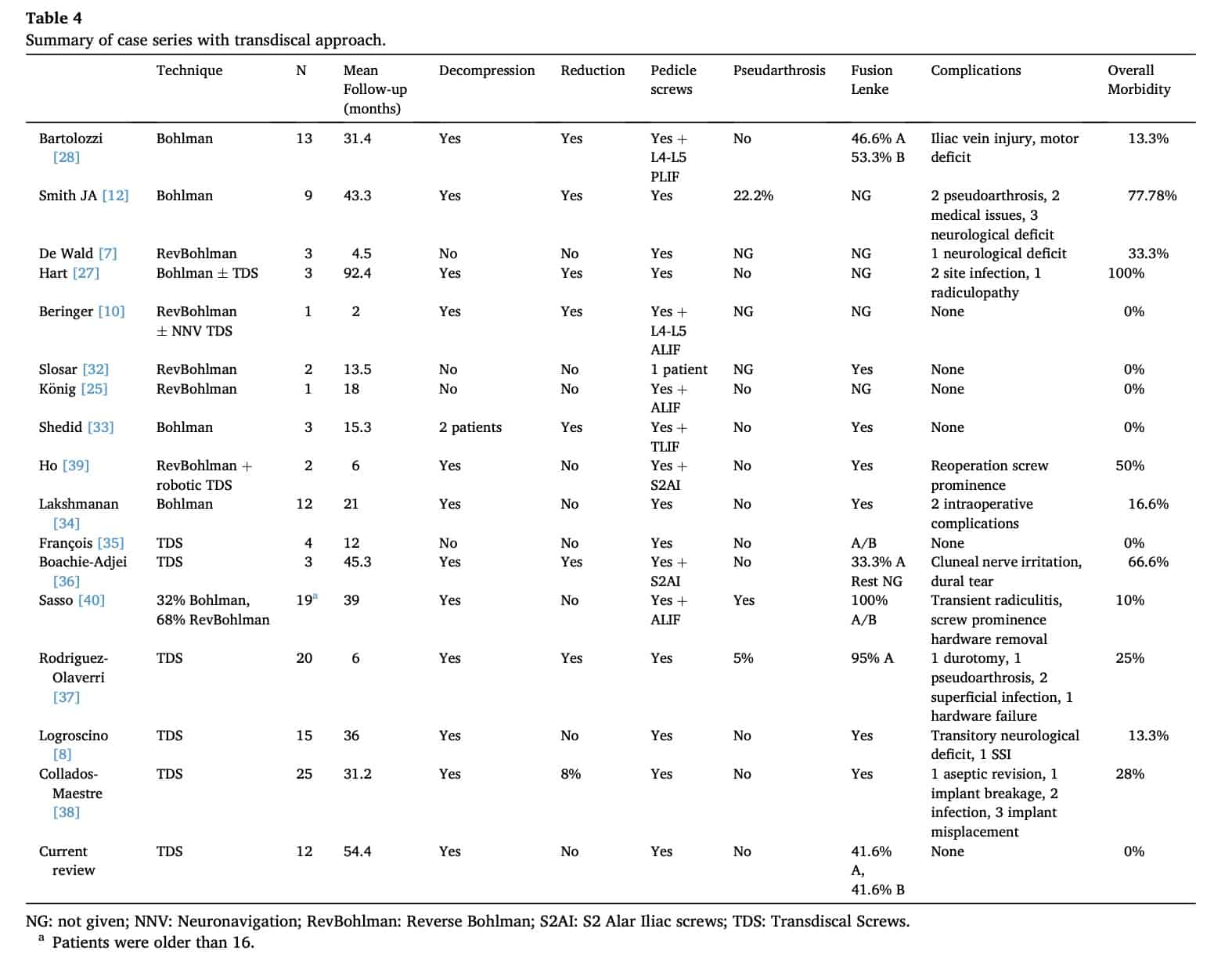
We have reviewed those case series of patients treated for HGS that included a transdiscal or transvertebral technique, with a cage or screws, in adult patients with both anterior or posterior approaches (Table 4 [7,8,10,12,25,27,28,32-39]). In some cases surgery was completed with
intervertebral cages such as PLIF [28], TLIF [33], or ALIF [10,25,40]; whereas in other cases with S2 alar iliac screws [36,39] were used. Compared to these series, our cohort has one of the longest follow-up.
Moreover, it is the one of the largest and longest cohort analyzing and describing the fusion and consolidation obtained after surgery. This assessment was carried out by CT scan using a clear criteria such as the Lenke or the Birdwell classifications. Generally speaking, as we have previously said, the evidence about this concept is still limited. How-ever, in accordance with our results most of the case series demonstrate some degree of fusion in this type of patients [6,29]. This could be explained by the fiXation obtained with the transdiscal approach, as these screws have demonstrated previously a good behavior for axial flexion and compression loads, with better results than transpedicular screws in some series and at least similar in torsional loads [13,15].
However, other series such as that of Rodriguez-Olaverri et al. [37] showed a higher rate of pseudoarthrosis with transdiscal approach compared to transpedicular screws. According to these findings, we hypothesized that the use of intraoperative navigation in our cohort could have helped to avoid complications in relation to fusion and pseudoarthrosis as there is evidence that this tool decreases rates of misplacement and hardware failure [41,42].
Finally, we have seen a clear improvement in clinical outcome in our patients, with a significant decrease in postoperative ODI and VAS at 1 year, and at the last follow-up, as well as an improvement in their quality of life measured in EuroQoL score. This evidence is in accordance with other series that demonstrate an improvement in pain in all patients treated [28,29].
The main limitation of this study is the number of patients, added to the retrospective nature of the study. However, as we have previously explained the low incidence of HGS, make difficult to obtained a bigger cohort, but the results obtained are still valuable.
NG: not given; NNV: Neuronavigation; RevBohlman: Reverse Bohlman; S2AI: S2 Alar Iliac screws; TDS: Transdiscal Screws.
5. Conclusion
Guided transdiscal L5-S1 fiXation with neuronavigation is an adequate technique for HGS. His technique has the advantage of being a unique posterior approach, provide a suitable biomechanical support with a low rate of complications, and achieve a good and long-lasting clinical outcome with a robust fusion in the listhetic space.
Juan Delgado-Fernández: Conceptualization (lead); writing -original draft (lead); formal analysis (lead). Natalia Frade-Porto: Methodology (lead); formal analysis; writing -original draft (support-ing). Guillermo Blasco: Data curation (equal); Resources (equal); writing -review & editing. Patricia González-Tarno: Methodology; Data curation (equal); Resources (equal); writing -review & editing. Ricardo Gil-Simoes: Visualization; Resources (equal); writing -review & editing. Paloma Pulido Rivas: Supervision (equal); writing -review & editing. Rafael García de Sola: Supervision(equal); writing -review & editing.
Declaration of interest
None.
References
[1] M.K. Kasliwal, J.S. Smith, A. Kanter, C.J. Chen, P.V. Mummaneni, R.A. Hart, C.
I. Shaffrey, Management of high-grade spondylolisthesis, Neurosurg. Clin. N. Am. 24 (2013) 275-291, https://doi.org/10.1016/j.nec.2012.12.002.
[2] L. Kalichman, D.H. Kim, L. Li, A. Guermazi, V. Berkin, D.J. Hunter, Spondylolysis and spondylolisthesis prevalence and association with low back pain in the adult
community-based population, Spine 34 (2009) 199-205.
[3] S. Ko, S. Lee, Prevalence of spondylolysis and its relationship with low back pain in
selected population, Clin. Orthop. Surg. 3 (2011) 34-38.
[4] M. Roche, G. Rowe, The incidence of separate neural arch and coincident bone
variations: a survey of 4,200 skeletons, Anat. Rec. 109 (1951) 233-252.
[5] J.M. Hire, J.M. Jacobs, J.V. Bundy, J.G. Devine, A modified Bohlman technique using a novel implant for treatment of high-grade spondylolisthesis, J. Neurosurg.
Spine 22 (2015) 80-83, https://doi.org/10.3171/2014.10.SPINE14138.
[6] M.K. Kasliwal, J.S. Smith, C.I. Shaffrey, D. Saulle, L.G. Lenke, Jr Polly DW, C.
P. Ames, J.H. Perra, Short-term complications associated with surgery for high-grade spondylolisthesis in adults and pediatric patients: a report from the scoliosis
research society morbidity and mortality database, Neurosurgery 71 (2012) 109-116, https://doi.org/10.1227/NEU.0b013e3182535881.
[7] C.J. DeWald, J.E. Vartabedian, M.F. Rodts, K.W. Hammerberg, Evaluation and
management of high-grade spondylolisthesis in adults, Spine 30 (2005) 49-59, https://doi.org/10.1097/01.brs.0000155573.34179.7e.
[8] C.A. Logroscino, F.C. Tamburrelli, L. Scaramuzzo, G.R. Schiro`, S. Sessa, L. Proietti, Transdiscal L5-S1 screws for the treatment of adult spondylolisthesis, Eur. Spine J. 21 (2012) 128-133, https://doi.org/10.1007/s00586-012-2229-8.
[9] F. Lombardi, V.M. Custodi, R. Pugliese, A. Risso, P. Gaetani, G. Butti, Treatment of
high-grade spondylolisthesis with Schanz recoil screws: our experience, Eur. Spine J. 22 (2013) 914-918, https://doi.org/10.1007/s00586-013-3013-0.
[10] W.F. Beringer, J.-P. Mobasser, D. Karahalios, E.A. Potts, Anterior transvertebral interbody cage with posterior transdiscal pedicle screw instrumentation for high-grade spondylolisthesis, Neurosurg. Focus 20 (2006) 7, https://doi.org/10.3171/ foc.2006.20.3.8.
[11] A. Palejwala, J. Fridley, A. Jea, Transsacral transdiscal L5-S1 screws for the
management of high-grade spondylolisthesis in an adolescent, J. Neurosurg. Pediatr. 17 (2016) 645-650, https://doi.org/10.3171/2015.12.PEDS15535.
[12] J.A. Smith, V. Deviren, S. Berven, F. Kleinstueck, D.S. Bradford, Clinical outcome of
trans-sacral interbody fusion after partial reduction for high-grade l5-s1 spondylolisthesis, Spine 26 (2001) 2227-2234, https://doi.org/10.1097/
00007632-200110150-00014.
[13] A. Minamide, T. Akamaru, S. Yoon, T. Tamaki, J. Rhee, W. Hutton, Transdiscal L5-S1 screws for the fiXation of isthmic spondylolisthesis: a biomechanical evaluation, J. Spinal Disord. Tech. 16 (2003) 144-149.
[14] W.A. Abdu, R.G. Wilber, S.E. Emery, Pedicular transvertebral screw fiXation of the
lumbosacral spine in spondylolisthesis. A new technique for stabilization, Spine 19 (1994) 710-715.
[15] S. Ozalp, M. Ozkaya, O. Yaman, T. Demir, Biomechanical comparison of transdiscal fiXation and posterior fiXation with and without transforaminal lumbar interbody fusion in the treatment of L5 -S1 lumbosacral joint, Proc. Inst. Mech. Eng. H 232 (2018) 371-377, https://doi.org/10.1177/0954411918760959.
[16] K. Aghayev, S.A. Gonzalez-blohm, J.J. Doulgeris, W.E. Lee, J.K. Waddell, F. D. Vrionis, Feasibility and biomechanical performance of a novel transdiscal screw system for one level in non-spondylolisthetic lumbar fusion: an in vitro investigation, Spine J. 14 (2014) 705-713, https://doi.org/10.1016/j.
spinee.2013.08.033.
[17] J. Fairbank, P. Pynsent, The oswestry disability index, Spine J. 25 (2000) 2940-2953.
[18] B. Mueller, L.Y. Carreon, S.D. Glassman, Comparison of the EuroQOL-5D with the oswestry disability indez, back and leg pain scores in patients with degenerative lumbar spine pathology, Spine 38 (2013) 757-761, https://doi.org/10.1097/ BRS.0b013e31827ab803.
[19] J. Dubousset, Treatment of spondylolysis and spondylolisthesis in children and adolescents, Clin. Orthop. Relat. Res. 337 (1997) 77-85.
[20] H. Labelle, P. Roussouly, E. Berthonnaud, E. Transfeldt, M. O’brien, D. Chopin, T. Hresko, J. Dimnet, Spondylolisthesis, pelvic incidence, and spinopelvic balance: a correlation study, Spine 29 (2004) 2049-2054, https://doi.org/10.1097/01. brs.0000138279.53439.cc.
[21] L. Lenke, K. Bridwell, D. Bullis, R. Betz, C. Baldus, P. Schoenecker, Results of in situ fusion for isthmic spondylolisthesis, J. Spinal Disord. Tech. 5 (1992) 433-442.
[22] K. Bridwell, L. Lenke, K. McEnery, C. Baldus, K. Blanke, Anterior fresh frozen structural allografts in the thoracic and lumbar spine. Do they work if combined with posterior fusion and instrumentation in adult patients with kyphosis or
anterior column defects Spine 20 (1995) 1410-1418.
[23] J. Delgado-Fernández, P. Pulido, M.Á. García-Pallero, G. Blasco, N. Frade-Porto, R.G. Sola, Image guidance in transdiscal fiXation for high-grade spondylolisthesis in adults with correct spinal balance, Neurosurg. Focus 44 (2018) 9, https://doi.org/ 10.3171/2017.10.FOCUS17557.
[24] A.W. Beck, A.K. Simpson, High-grade lumbar spondylolisthesis, Neurosurg. Clin. N. Am. 30 (2019) 291-298, https://doi.org/10.1016/j.nec.2019.02.002.
[25] M. Ko¨nig, B. Boszczyk, Limited access surgery for 360 degrees in-situ fusion in a
dysraphic patient with high-grade spondylolisthesis, Eur. Spine J. 21 (2012) 390-395, https://doi.org/10.1007/s00586-011-1994-0.
[26] E.E. Transfeldt, A.A. Mehbod, Evidence-based medicine analysis of isthmic spondylolisthesis treatment including reduction versus fusion in situ for high-grade slips, Spine 32 (2007) 126-129.
[27] R.A. Hart, C.M. Domes, B. Goodwin, C.R. D’amato, J.U. Yoo, R.J. Turker, M. F. Halsey, High-grade spondylolisthesis treated using a modified Bohlman technique: results among multiple surgeons, J. Neurosurg. Spine 20 (2014)
523-530.
[28] P. Bartolozzi, A. Sandri, M. Cassini, M. Ricci, One-stage posterior decompression-stabilization and trans-sacral interbody fusion after partial reduction for severe L5-S1 spondylolisthesis, Spine 28 (2003) 1135-1141.
[29] R.S. Rindler, B.A. Miller, S.R. Eshraghi, G. Pradilla, D. Refai, G. Rodts, F.U. Ahmad,
Efficacy of transsacral instrumentation for high-grade spondylolisthesis at L5-S1: a systematic review of the literature, World Neurosurg. 95 (2016) 623.e11-623.e19, https://doi.org/10.1016/j.wneu.2016.05.030.
[30] U.G. Longo, M. Loppini, G. Romeo, N. Maffulli, V. Denaro, Evidence-based surgical management of spondylolisthesis: reduction or arthrodesis in situ, J. Bone Jt. Surg. 96 (2014) 53-58, https://doi.org/10.1016/S0021-9355(14)74000-7.
[31] F.L. Acosta, C.P. Ames, D. Chou, Operative management of adult high-grade
lumbosacral spondylolisthesis, Neurosurg. Clin. N. Am. 18 (2017) 249-254, https://doi.org/10.1016/j.nec.2007.01.001.
[32] P.J. Slosar, J.B. Reynolds, M. Koestler, The axial cage: a pilot study for interbody fusion in higher-grade spondylolisthesis, Spine J. 1 (2001) 115-120.
[33] D. Shedid, C. Frcs, A.G. Weil, I. Lieberman, C. Frcs, A novel minimally invasive
technique for the treatment of high-grade isthmic spondylolisthesis using a posterior transsacral rod, J. Spinal Disord. Tech. 27 (2014) 41-48.
[34] P. Lakshmanan, S. Ahuja, M. Lewis, J. Howes, P.R. Davies, Transsacral screw fiXation for high-grade spondylolisthesis, Spine J. 9 (2009) 1024-1029, https:// doi.org/10.1016/j.spinee.2009.08.456.
[35] J. François, P. Lauweryns, G. Fabry, Treatment of high-grade spondylolisthesis by posterior lumbosacral transfiXation with transdiscal screws: surgical technique and preliminary results in four cases, Acta Orthop. Belg. 71 (2005) 334-341.
[36] O. Boachie-Adjei, T. Do, B. a Rawlins, Partial lumbosacral kyphosis reduction, decompression, and posterior lumbosacral transfiXation in high-grade isthmic spondylolisthesis: clinical and radiographic results in siX patients, Spine 27 (2002) E161-E168, https://doi.org/10.1097/00007632-200203150-00019.
[37] J.C. Rodriguez-Olaverri, N.C. Zimick, A. Merola, J. Vicente, J. Rodriguez,
A. Tabuenca, A. Loste, E. Sun˜en, J. Burgos, E. Hevia, G. Piza-Vallespir, Comparing the clinical and radiological outcomes of pedicular transvertebral screw fiXation of the lumbosacral spine in spondylolisthesis versus unilateral transforaminal lumbar
interbody fusion (TLIF) with posterior fiXation using anterior cages, Spine 33 (2008) 1977-1981, https://doi.org/10.1097/BRS.0b013e31817ecc01.
[38] I. Collados-Maestre, A. Lizaur-Utrilla, T. Bas-Hermida, E. Pastor-Fernández, V. Gil-Guillén, Transdiscal screw versus pedicle screw fiXation for high-grade L5-S1 isthmic spondylolisthesis in patients younger than 60years: a casecontrol study, Eur. Spine J. 25 (2016) 1806-1812, https://doi.org/10.1007/s00586-016-4550-0.
[39] A.L. Ho, K. Varshneya, Z.A. Medress, A.V. Pendharkar, E.S. Sussman, I. Cheng, A. Veeravagu, Grade II Spondylolisthesis: reverse bohlman procedure with transdiscal S1-L5 and S2 alar iliac screws placed with robotic guidance, World Neurosurg. 132 (2019) 421-428, https://doi.org/10.1016/j.wneu.2019.07.229.
[40] R.C. Sasso, K.D. Shively, T.M. Reilly, Transvertebral transsacral strut grafting for high-grade isthmic spondylolisthesis L5-S1 with fibular allograft, J. Spinal Disord. Tech. 21 (2008) 328-333.
[41] R. Xiao, J.A. Miller, N.C. Sabharwal, D. Lubelski, V.J. Alentado, A.T. Healy, T.
E. Mroz, E.C. Benzel, Clinical outcomes following spinal fusion using an intraoperative computed tomographic 3D imaging system, J. Neurosurg. Spine 26
(2017) 628-637, https://doi.org/10.3171/2016.10.SPINE16373.
[42] N.F. Tian, Q.S. Huang, P. Zhou, Y. Zhou, R.K. Wu, Y. Lou, H.Z. Xu, Pedicle screw insertion accuracy with different assisted methods: a systematic review and meta-analysis of comparative studies, Eur. Spine J. 20 (2011) 846-859.