Surgical treatment of brainstem cavernous malformations: an international Delphi consensus
El Dr. Rafael García de Sola ha participado y trabajado junto a grandes neurocirujanos especialistas en el campo neurovascular.
Se trató de hacer una revisión sobre uno de los problemas más complejos de tratar hoy día: Los angiomas cavernosos a nivel del tronco cerebral.
Se adjunta el abstract y el trabajo publicado en el Journal of Neurosurgery.
Si lo desea, puede leer también el artículo del Dr. García de Sola de 2007, sobre este tema:
Surgical treatment of brainstem cavernous malformations: an international Delphi consensus
Philipp Dammann 1, Adib A Abla 2, Rustam Al-Shahi Salman 3, Hugo Andrade-Barazarte 4, Vladimir Benes 5, Marco Cenzato 6, E Sander Connolly 7, Jan F Cornelius 8, William T Couldwell 9, Rafael G Sola 10, Santiago Gomez-Paz 11, Erik Hauck 12, Juha Hernesniemi 4, Juri Kivelev 13, Giuseppe Lanzino 14, R Loch Macdonald 15, Jacques J Morcos 16, Christopher S Ogilvy 11, Hans-Jakob Steiger 8, Gary K Steinberg 17, Alejandro N Santos 1, Laurèl Rauschenbach 1, Marvin Darkwah Oppong 1, Börge Schmidt 18, Robert F Spetzler 19, Karl Schaller 20, Michael T Lawton 19, Ulrich Sure 1
Affiliations
• 1 Department of Neurosurgery and Spine Surgery, University Hospital Essen, Germany.
• 2 Department of Neurological Surgery, University of California, San Francisco, California.
• 3 Centre for Clinical Brain Sciences, University of Edinburgh, United Kingdom.
• 4 Department of Neurosurgery, Juha Hernesniemi International Center, Henan Provincial People’s Hospital, University of Zhengzhou, China.
• 5 Department of Neurosurgery and Neuro-oncology, Military University Hospital and Charles University, First Medical Faculty, Prague, Czech Republic.
• 6 Department of Neurosurgery, Niguarda Metropolitan Hospital, Milan, Italy.
• 7 Columbia University Medical Center Department of Neurological Surgery, New York, New York.
• 8 Department of Neurosurgery, Medical Faculty, Heinrich-Heine-University, Düsseldorf, Germany.
• 9 Department of Neurosurgery, University of Utah, Salt Lake City, Utah.
• 10 UAM Chair «Innovation in Neurosurgery,» Universidad Autónoma de Madrid, Spain.
• 11 Neurosurgical Service, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, Massachusetts.
• 12 Department of Neurosurgery, Duke University Medical Center, Durham, North Carolina.
• 13 Department of Neurosurgery, Turku University Hospital, Turku, Finland.
• 14 Neurosurgery, Mayo Clinic, Rochester, Minnesota.
• 15 Department of Neurological Surgery, University of California, San Francisco, Fresno Campus, Fresno, California.
• 16 Department of Neurological Surgery, University of Miami, Florida.
• 17 Department of Neurosurgery and Stanford Stroke Center, Stanford University School of Medicine, Stanford, California.
• 18 Institute for Medical Informatics, Biometry and Epidemiology, University Hospital of Essen, University of Duisburg-Essen, Essen, Germany; and.
• 19 Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona.
• 20 Department of Neurosurgery, Department of Clinical Neurosciences, Geneva University Hospital, Geneva, Switzerland.
• PMID: 34598135
• DOI: 10.3171/2021.3.JNS2156
ABSTRACT
Objective:
Indication for surgery in brainstem cavernous malformations (BSCMs) is based on many case series, few comparative studies, and no randomized controlled trials. The objective of this study was to seek consensus about surgical management aspects of BSCM.
Methods:
A total of 29 experts were invited to participate in a multistep Delphi consensus process on the surgical treatment of BSCM.
Results:
Twenty-two (76%) of 29 experts participated in the consensus. Qualitative analysis (content analysis) of an initial open-ended question survey resulted in 99 statements regarding surgical treatment of BSCM. By using a multistep survey with 100% participation in each round, consensus was reached on 52 (53%) of 99 statements.
These were grouped into 4 categories:
- 1) definitions and reporting standards (7/14, 50%);
- 2) general and patient-related aspects (11/16, 69%);
- 3) anatomical-, timing of surgery-, and BSCM-related aspects (22/37, 59%); and
- 4) clinical situation-based decision-making (12/32, 38%). Among other things, a consensus was reached for surgical timing, handling of associated developmental venous anomalies, handling of postoperative BSCM remnants, assessment of specific anatomical BSCM localizations, and treatment decisions in typical clinical BSCM scenarios.
Conclusions:
A summary of typical clinical scenarios and a catalog of various BSCM and patient-related aspects that influence the surgical treatment decision have been defined, rated, and interpreted.
Keywords:
Delphi consensus; brainstem cavernous malformations; surgery; vascular disorders.
Abbreviations
BSCM = brainstem cavernous malformation; CCM = cerebral cavernous malformation; DVA = developmental venous anomaly; SH = symptomatic hemorrhage.
ACCOMPANYING EDITORIAL 10.3171/2021.4.JNS21622.
SUBMITTED January 13, 2021. ACCEPTED March 9, 2021.
INCLUDE WHEN CITING Published online October 1, 2021; DOI: 10.3171/2021.3.JNS2156.
©AANS 2021, except where prohibited by US copyright law J Neurosurg October 1, 2021 1
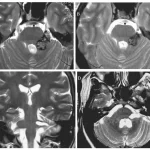
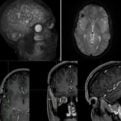
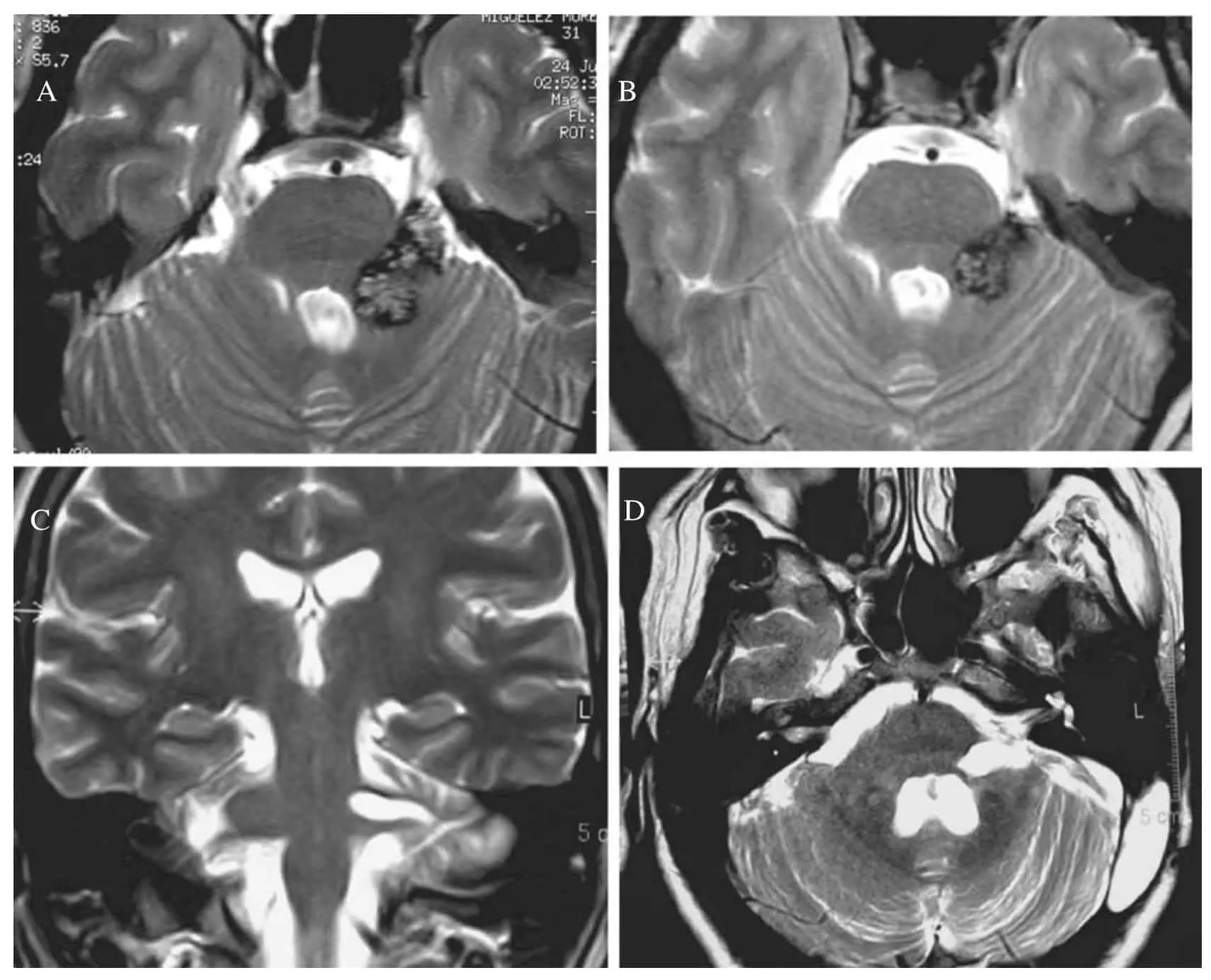
BRAINSTEM cavernous malformations (BSCMs)
BRAINSTEM cavernous malformations (BSCMs) are rare, accounting for approximately 30% of all cerebral cavernous malformations (CCMs),1 which have a prevalence of about 0.5% in the general population.2 Due to the eloquent location of BSCMs, symptomatic hemorrhages3 (SHs) of BSCMs are more frequent and cause more severe disability compared to supratentorial CCMs.1,4,5 These factors make patients with BSCMs potential candidates for neurosurgical treatment, which is performed in approximately 20%-25% of cases according to larger series.1 Because such treatment is associated with significant risks for shortand long-term morbidity and impairment of health-related quality of life,5-9 the indications for surgery remain controversial.10 The anatomical complexity of the brainstem and the clinical heterogeneity of BSCMs in terms of lesion characteristics (such as symptoms, specific location, past clinical course and frequency of SHs, size, time point of SH, etc.) in combination with the relative rarity of the disease make comparative clinical investigations with a randomized controlled approach in BSCM very challenging.
Current treatment decisions are therefore mainly based on the individual surgeonÕs experience, with medical evidence limited to many case series, few comparative studies, and no randomized controlled trials.5,11 This limited evidence may lead to high levels of variation in the management and outcome of BSCM among clinicians.1,5,12 Guidelines for clinical management of CCM10 only give very general recommendations in this regard: the authors of the guidelines believe offering a resection after a second SH is ÒreasonableÓ (class IIb evidence, level B13), while an indication for resection after a single disabling SH is seen as more controversial (class IIb evidence, level C13). Clinical decision-making and selection for BSCM surgery requires considering more variables than merely the history and frequency of SHs.
Therefore, we sought to perform a survey using the Delphi method on multiple aspects of surgical treatment and management of BSCM among international experts to investigate the extent of current clinical consensus that could be achieved and identify questions and ambiguities that may drive future (randomized controlled) trials. The Delphi method is a structured, multiround communication technique in which an expert panel identifies and evaluates key aspects of a complex problem to reach a consensus.14-16 This method, among others, is recommended as a tool to help develop guidelines for rare diseases with low medical evidence.17-20
Methods
Delphi Survey
An international group of experts in the surgical management of BSCM was selected by a steering committee (M.T.L., K.S., and U.S.). The selection process included the following steps: First, the number of potential panel members was set at 20-30 according to survey extent, number of survey rounds, and comparable surveys.21-24 Second, inclusion criteria for panel members were defined as follows: clinical experience with BSCM management > 10 years (leading to an assumed minimum of 50 BSCM consultations), and/or contribution in PubMed (NCBI)-listed cohort studies or clinical series on BSCM (2005-2018), and/or prominent participation in clinical CCM research. The steering committee finally unanimously designated 29 international candidates based on their assumed willingness and availability to participate. The selection process was therefore performed consistent with those in comparable Delphi surveys.21,24
All candidates were invited to take part in a basic Delphi survey20 to identify and evaluate crucial aspects of surgical decision-making and management of BSCM. Data were collected using web-based surveys (SurveyMonkey) that were electronically distributed in 2018 and 2019. The 3 authors who designed the survey rounds (P.D., M.T., and B.S.) did not participate in the consensus process. Three panel members confirmed the eligibility of the first survey (M.T.L., K.S., and U.S.) and participated in the consensus process. An overview of the survey process is given in Fig. 1.
In the first round, participants were invited to answer 47 open-ended questions (see Supplemental Digital Content 1). The first survey was segmented into 5 sections: patient-, symptom-, cavernoma-, and intervention-related, and general questions. Additionally, self-reported background characteristics of each panel member were evaluated and reviewed, including the number of consulted BSCM cases, number of BSCM surgeries performed, and proportion of cases in which an indication for surgery and/or postoperative outcome was uncertain or unclear. A content analysis of the answers to the 47 open-ended questions, consisting of 4 stages (decontextualization, recontextualization, categorization, and compilation),25 was performed (P.D., B.S., M.D.O., L.R., A.N.S.). Based on this analysis, 99 categorized statements were designed (see Supplemental Digital Content 2).
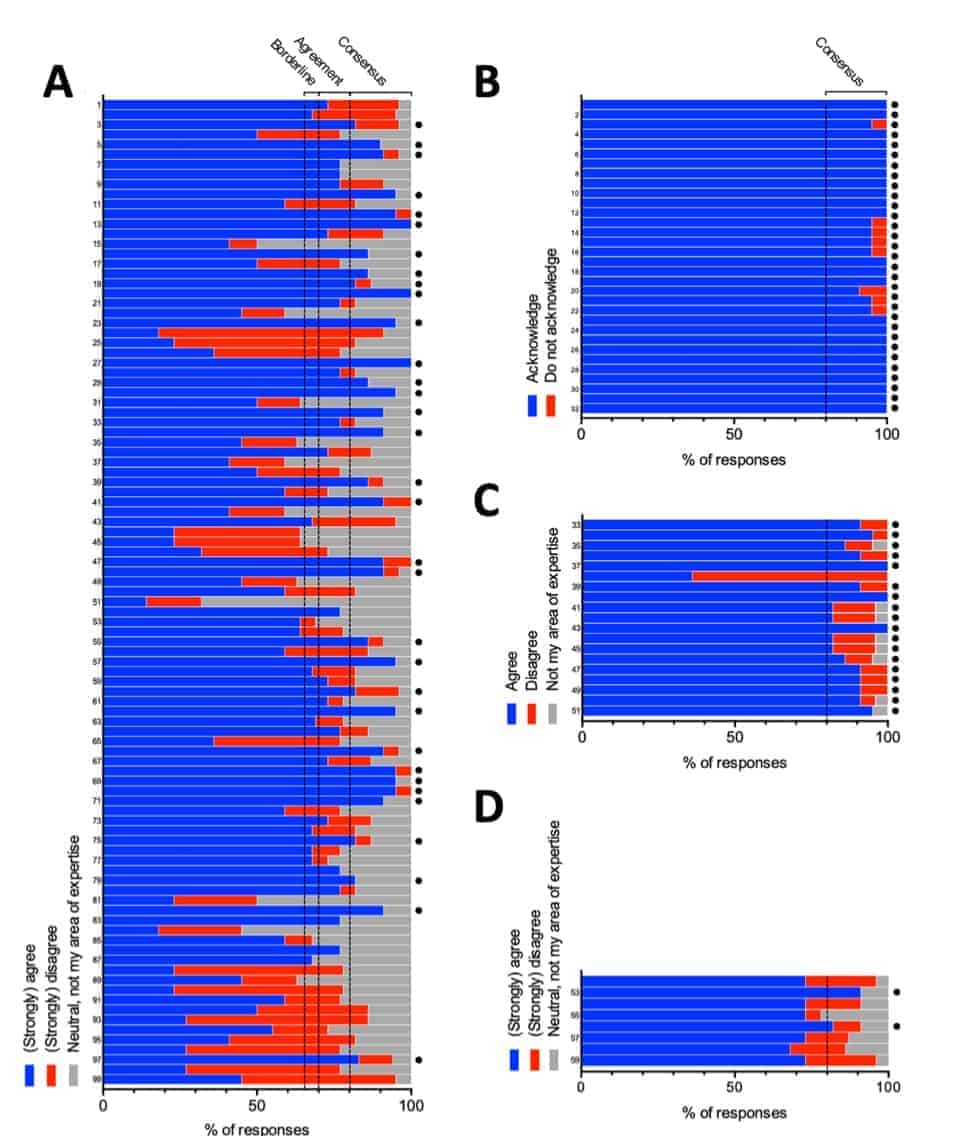
In the second round, these statements were grouped into 4 categories: 1) definitions and reporting standards (n = 14); 2) general and patient-related aspects (n = 16); 3) anatomical-, timing of surgery-, and BSCM-related aspects (n = 37); and 4) clinical situation-based decisionmaking (n = 32). These statements were then distributed to the group (see Supplemental Digital Content 3). Degree of agreement was rated according to a 5-point Likert scale (strongly agree, agree, neutral, disagree, strongly disagree, not my area of expertise [skip the statement]) and responses were anonymously collected (Fig. 2A). Reponses with 80%-100% agreement (strongly agree, agree) or disagreement (disagree, strongly disagree) were considered acceptable to reach consensus. Responses with 70%-79% agreement (strongly agree, agree) or disagreement (disagree, strongly disagree) were defined as agreement. Responses with 65%-69% agreement (strongly agree, agree) or disagreement (disagree, strongly disagree) were considered borderline agreement. Based on the answers, statements were modified and distilled into 59 revised statements that functioned as feedback to the panel members.
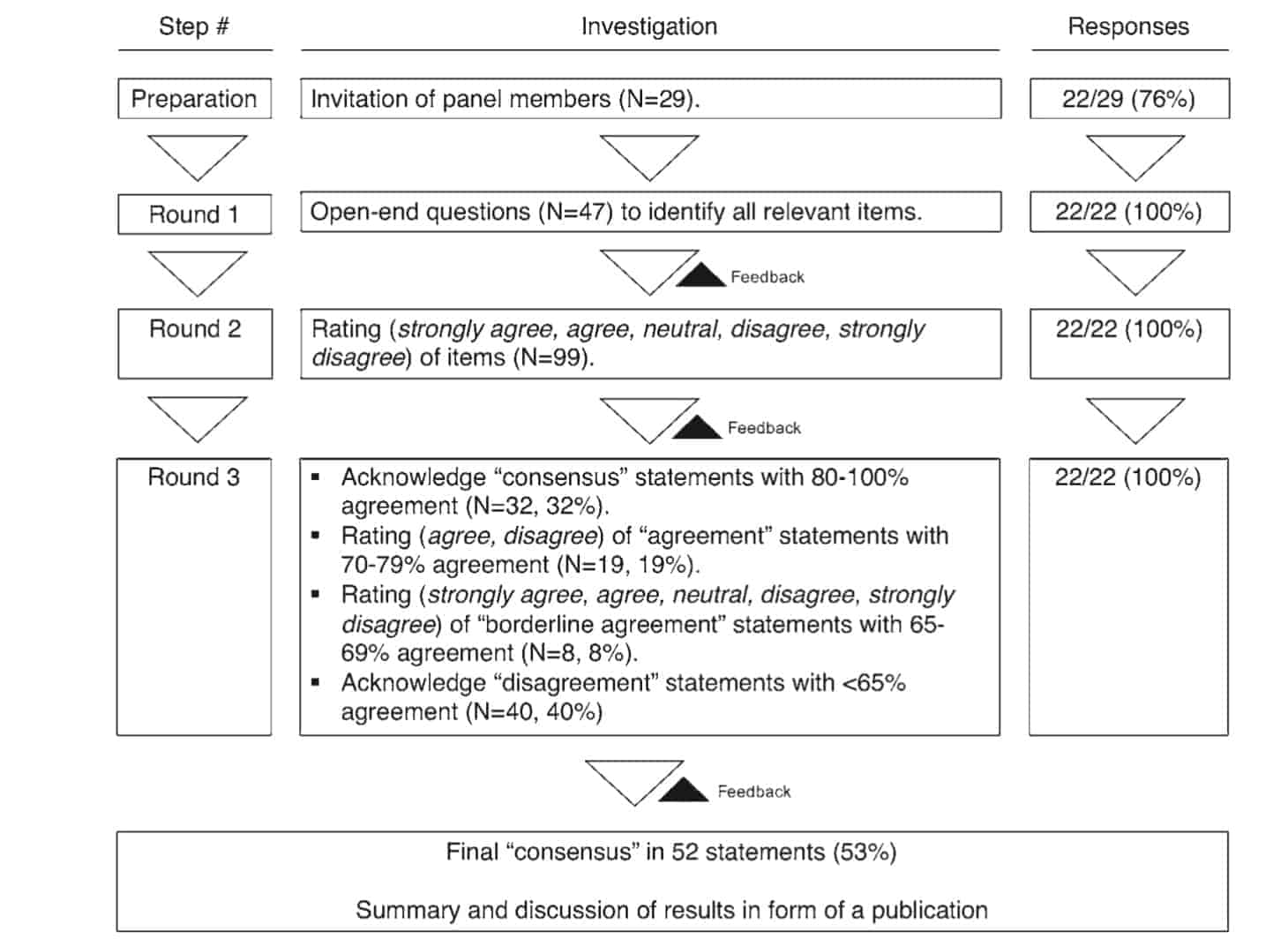
A third survey was conducted to meet further consensus and revised statements were distributed to the group (Fig. 2B-D). Statements that reached consensus in the second survey were again confirmed (acknowledge, do not acknowledge). Statements that reached agreement in the second round were distributed to the panel members using a 2-point Likert scale (agree, disagree, not my area of expertise). Statements that reached borderline agreement in the second survey were again rated with a 5-point Likert scale (strongly agree, agree, neutral, disagree, strongly disagree, not my area of expertise). A final document including consensus statements was generated, in which statements were grouped to specific items, commented on regarding the clinical implications (P.D., M.T.L., K.S., U.S.), and reviewed by all participants (see Supplemental Digital Content 4, Tables 1-4).
Based on the consensus statements reached, a diagram covering typical clinical BSCM scenarios and their potential treatment options was designed (P.D., M.T.L., K.S.,U.S.).
Results
Of the 29 invited experts, 22 (76%) responded and agreed to participate (21/28 neurosurgeons [75%], 1/1 neurologist [100%]). All survey rounds were fully completed by all participants. Initial consensus (round 2) was reached on 33 statements. Initial agreement was reached on 19 statements, and initial borderline agreement on 9 statements. After reevaluation, final consensus (round 3) was reached on 52 statements (Fig. 1). Stratified by categories, consensus was reached on: 1) definitions and reporting standards statements (7/14, 50%); 2) general and patientrelated aspects statements (11/16, 69%); 3) anatomical-, timing of surgery-, and BSCM-related aspects statements (22/37, 59%); and 4) clinical situation-based decisionmaking statements (12/32, 38%). For the first round, no median duration to complete the survey was recorded, whereas for the second and third rounds, 45.32 and 39.21 minutes were spent to complete the survey, respectively.
Consensus Statements
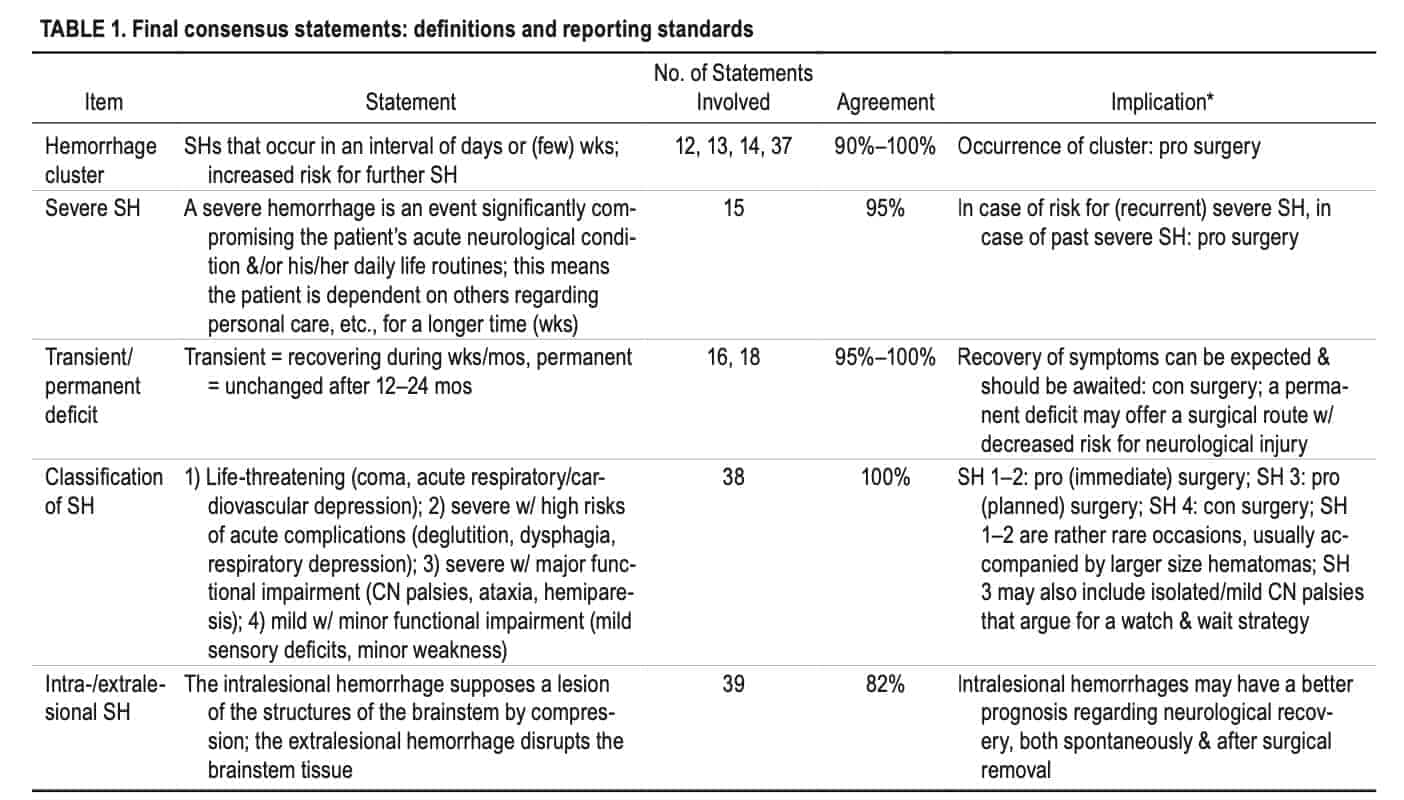
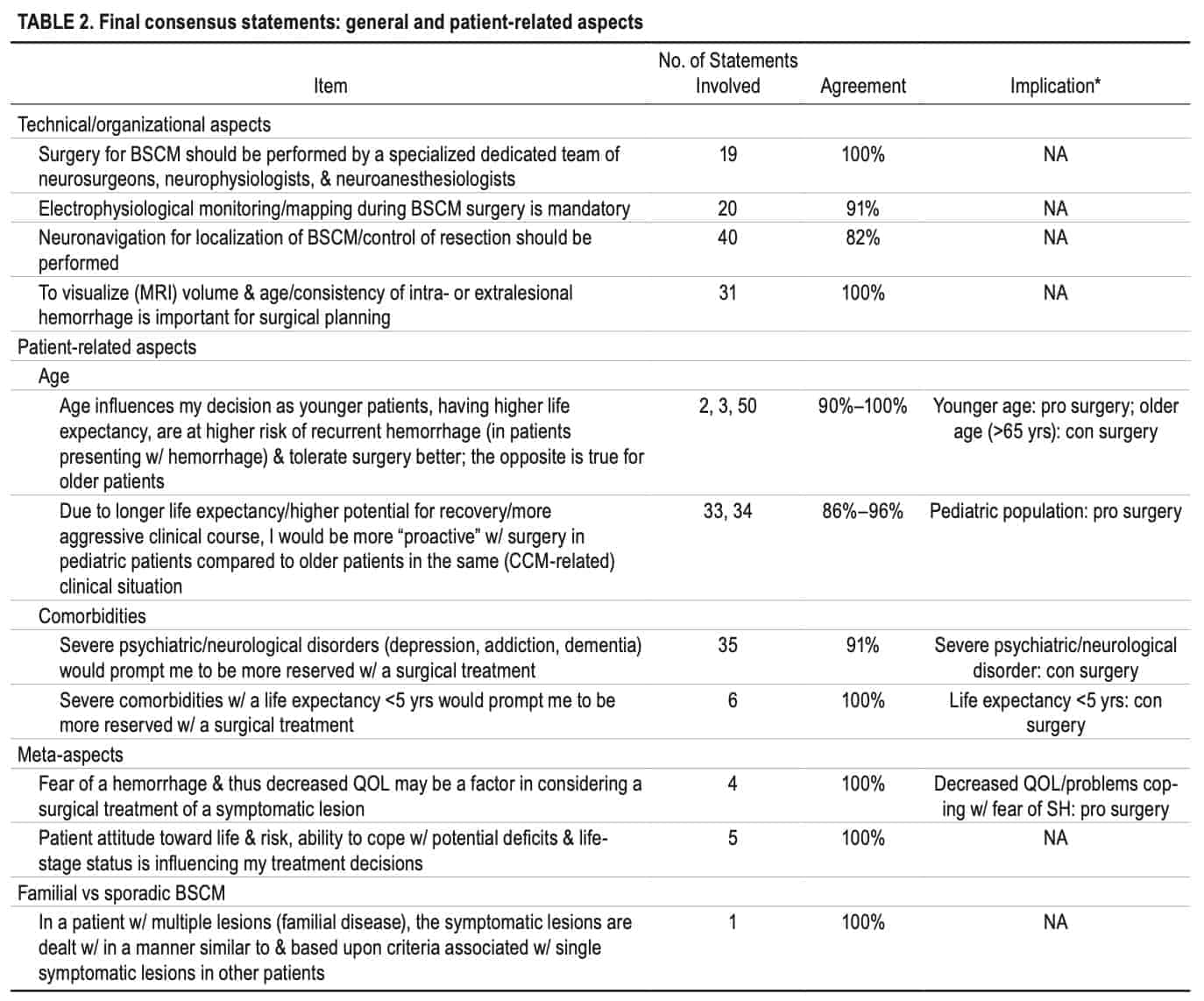
All 52 consensus statements were grouped into 4 categories, each consisting of several items: 1) definitions and reporting standards (5 items, Table 1); 2) general and patient-related aspects (11 items, Table 2); 3) anatomical-, timing of surgery-, and BSCM-related aspects (21 items, Table 3); and 4) clinical situation-based decision-making (9 items, Table 4). Information about several typical clinical BSCM scenarios merged into a diagram is found in Fig. 3.
Main Statements That Reached Consensus
The following selected, more-general statements areconsidered to be clinically most important among the various statements upon which consensus was reached: symptomatic lesions are managed according to similar criteria in sporadic and familial cases; asymptomatic BSCM should not be treated, regardless of CCM or patient characteristics; a first severe SH with mass effect should undergo surgical treatment; recurrent SH with progressive neurological deficit and easy access of the BSCM should undergo surgical treatment; developmental venous anomalies (DVAs) should be spared during surgery and large DVAs can conflict the surgical approach and increase surgical risks; preferable timing for a resection is between 4 and 8 weeks after last SH event; and significant remnants identified on postoperative imaging should be directly resected, if accessible.
Controversies
Forty-seven items (47%) did not reach consensus, indicating controversial/neutral opinions among the panel members regarding these items (see Supplemental Digital Content 5). These items mainly concern the relevance of intraversus extralesional hemorrhage, follow-up imaging standards, interpretation of ÒgrowingÓ lesions (silent hemorrhages) on serial MRI, and the exact definition of a deep-seated lesion or difficult BSCM location.
Background Characteristics of Panel Members
Panel members reported consultations with an overall total of 5300 BSCM patients (median 200 patients). The number of reported surgeries was 1270 cases (unavailable in 3 panel members). The mean proportion of BSCMs treated was 27.4% (range 10%-55%). The proportion of uncertainty about expected postoperative outcome or indication for surgery in treated cases was 19.2% (range 5%-50%).
Discussion
This Delphi exercise achieved consensus (80%-100% agreement) for 52 (53%) of 99 statements relevant to surgery for BSCM, providing level III evidence13 (expert committee). Among other topics, panel members agreed on the definition of so-called hemorrhage clusters and a classification of SH severity. Necessary technical and organizational requirements for surgical treatment of BSCM were outlined. Patient characteristics such as age, comorbidities, psychological aspects, and familial disease, as well as various BSCM-specific aspects (MRI morphology and location), were evaluated regarding their possible influence on an operative indication. A preferable time point of surgery after the last SH (4-8 weeks) and handling of postoperative BSCM remnants was agreed upon. Finally, the clinically most relevant basic BSCM scenarios (frequency and severity of SHs, location of BSCM) were rated regarding indication for surgery.
CN = cranial nerve; con = against; pro = in favor of. * The item can influence the overall decision toward or against surgery.
How to Read the Results?
The results of this work are condensed expert opinions reflecting personal experiences with the treatment and understanding of the currently available body of literature on BSCM. Given that absolute indications for surgery in BSCM appear to be rare (e.g., emergency situations with SH level 1-2; Table 1), and most of the time a very complex balancing of risks and benefits is necessary, this detailed evaluation of relevant clinical aspects regarding an indication for surgery is believed to function as an information baseline to drive future (randomized controlled) trials. As long as recommendations with higher levels of evidence do not exist, the extensive details of this work on different aspects of surgical treatment and indications may also comprehensively support current individual decisionmaking. The authors have deliberately refrained from establishing or proposing a grading scale with specific cutoff values, because such a scale would not reach the necessary level of detail. Validated grading scales based on a more limited number of parameters to estimate postoperative outcome are described elsewhere.6,26,27 However, based on the results of the consensus, an overview of typical clinical scenarios and their potential treatment options is proposed (Fig. 3).
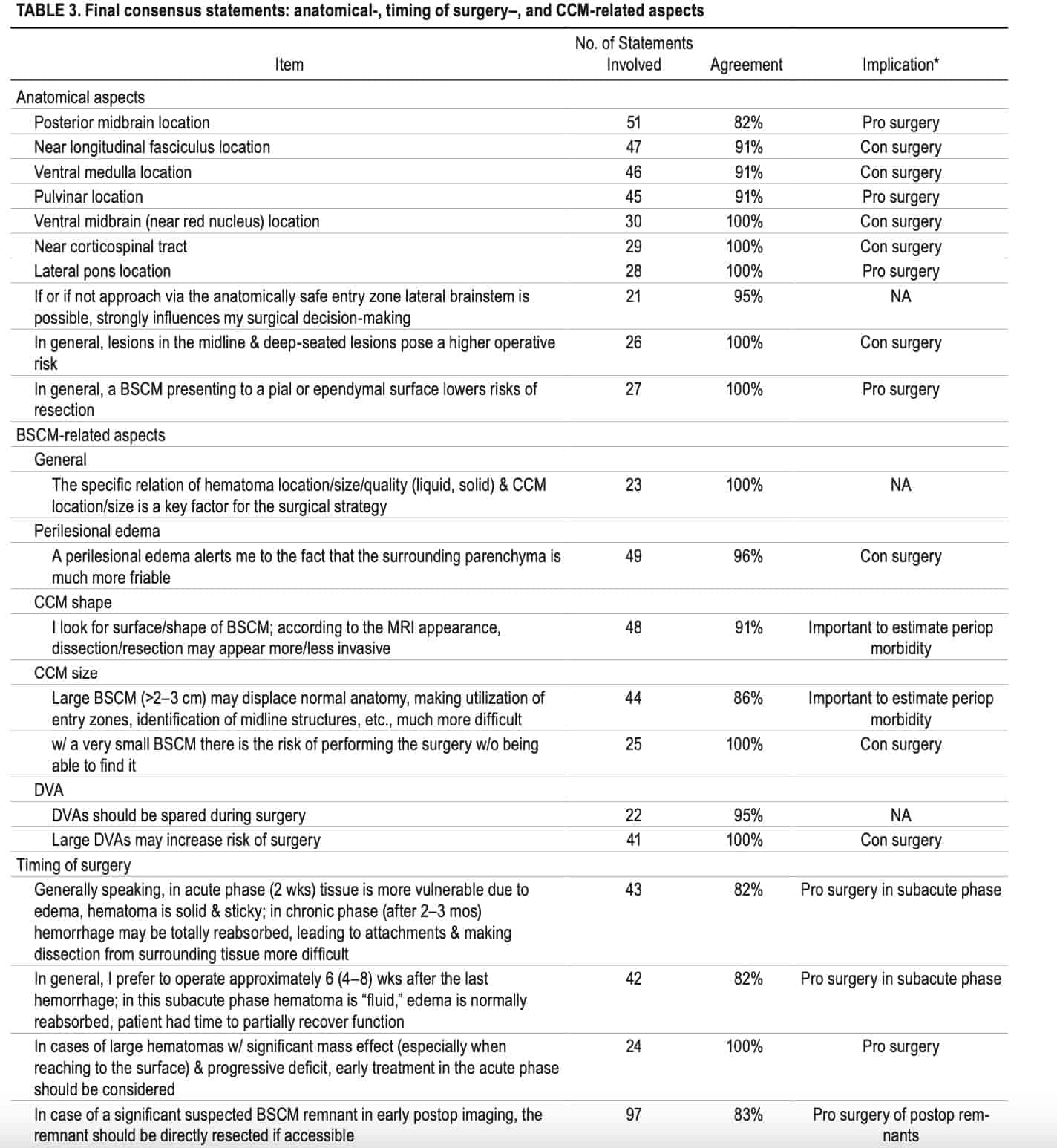
* The item can influence the overall decision toward or against surgery.
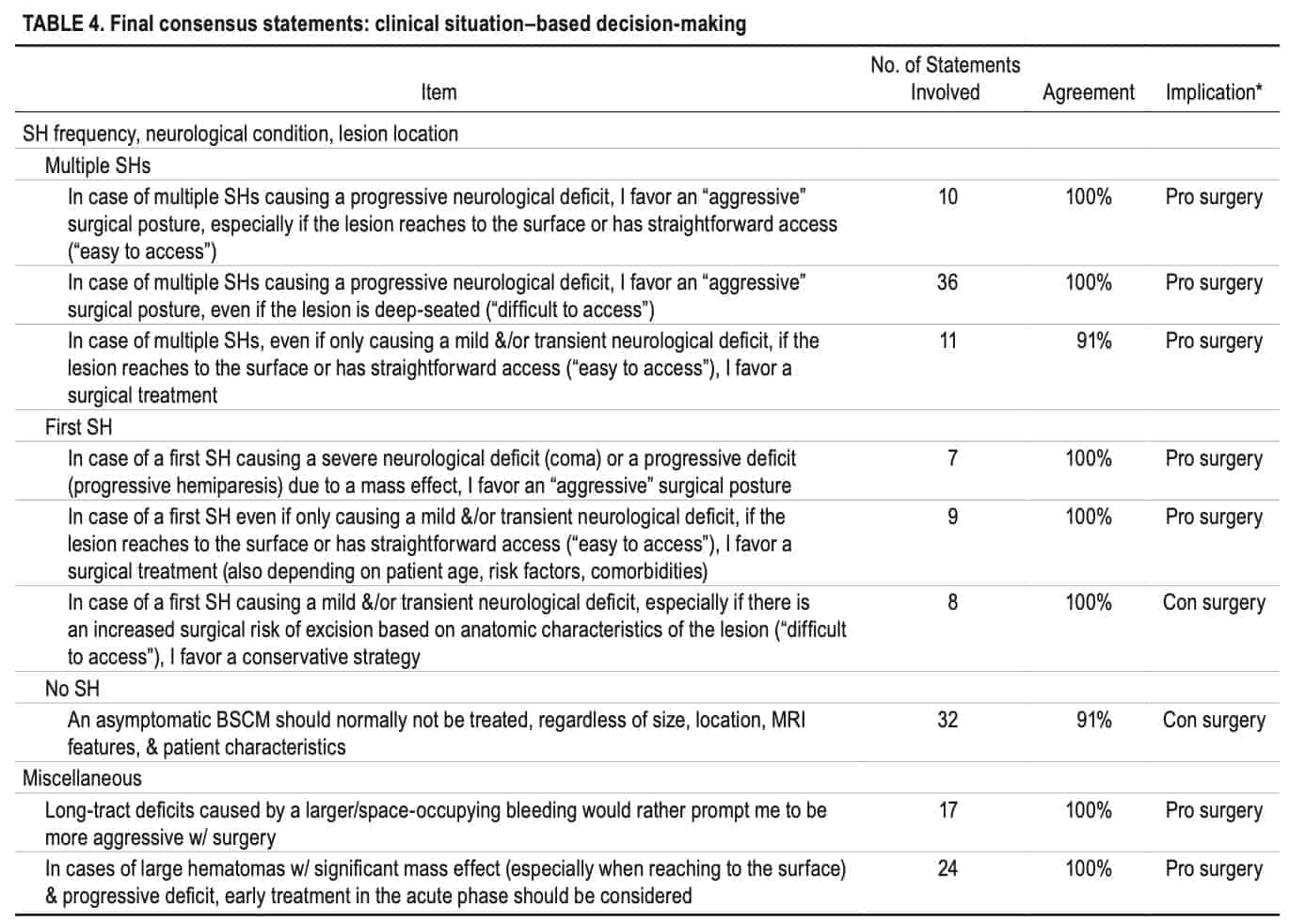
* The item can influence the overall decision toward or against surgery.
Generalizability to the Average Neurosurgical Practice
Surgical treatment of a rare and surgically challenging condition such as BSCM should be reserved for specialized teams that have already accumulated significant expertise and experience regarding surgical management and indications for treatment. Conversely, outcomes and indications of such experts should be continuously evaluated and reviewed, by establishing national and international registries and patient identifiers.28 Consequently, the generalizability of the consensus of this survey to the average neurosurgical practice is limited. We do not believe that the survey can replace higher-level evidence practice guidance, but it serves to condense currently available expert opinions. Of course, randomized controlled trials have the highest internal validity, providing the most reliable level of evidence guiding practice. However, these trials are limited regarding flexibility and generalizability to neurosurgical Òreal-worldÓ practice and their necessary level of detail, especially in rare, heterogenous, and complex diseases such as BSCM.
Recently, the National Institute of Neurological Disorders and Stroke of the NIH has published recommendations on how to address such challenges of clinical research in neurosurgery in the future.28
Strengths and Limitations
Among the strengths of this study is the extensive experience of the panel members (n = 22), reporting 5300 overall consultations and 1270 surgeries for BSCM. For comparison, the largest meta-analysis to date comprises 2493 surgical cases from 86 studies since 1986.5 The relatively typical reported mean proportion of patients with BSCM who underwent surgical treatment by panel members (27.4%) reflects the external validity of the panel membersÕ overall surgical Òposture.Ó However, a proportion ranging from 10% to 55% also indicate a wide variability in surgical management among the panel members. It is surprising that despite this variability, an agreement of 80%-100% was reached on 52 (53%) of 99 statements. Methodological strengths of our paper are the conception of a three-round survey with panel member feedback (circulation of previous responses) in between all rounds, and a clear definition of a desired consensus (80% agreement on a Likert scale).
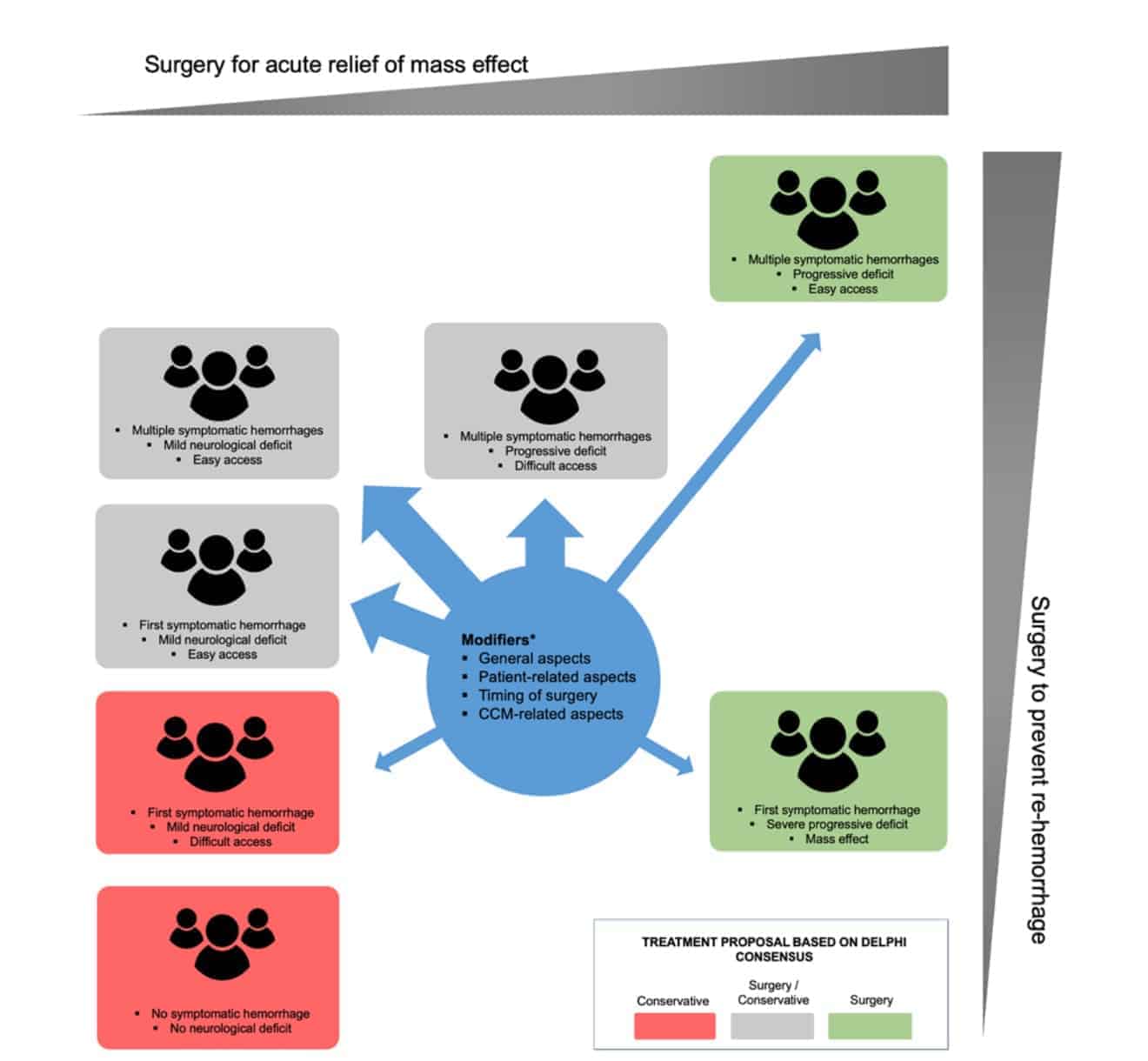
Although all these aspects are recommended for a Delphi survey, they are often not adhered to.20 The number of panel members was only slightly above the average of that in other Delphi surveys (n = 17) as reported in a meta-analysis.20 Because selection of panel members is crucial for the consensus technique to work properly,29 we followed recommendations in panel member selection (expertise and willingness and commitment to participate)30 and thus reached a response rate of 100% in each round. Overall, this Delphi consensus is in accordance with the Guidance on Conducting and Reporting Delphi Studies (CREDES),31 which is, however, a guideline for Delphi studies in palliative care.
Limitations of our work are the inclusion of terms that are only roughly defined and leave space for individual interpretation, such as ÒeasyÓ or ÒdifficultÓ surgical access. However, such a scope for interpretation is due to the anatomical and clinical complexity of BSCM and seems ultimately unavoidable. Another limitation is the overrepresentation of neurosurgeons (n = 21) versus neurologists (n = 1, with extremely high expertise in clinical CCM research) as panel members. This survey was, however, specifically planned to evaluate the core surgical aspects and indications of BSCM management, consequently demanding a neurosurgical background. Another limitation is the exclusion of radiotherapy as a treatment option for BSCM, which, although discussed with controversy, is established for the treatment of BSCM.32 However, the original aim of this work was to show consensus and controversy regarding selective surgical treatment in great detail. The balance between surgical, radiotherapeutic, and conservative treatment may also be a suitable topic for a Delphi consensus but would require a completely different structure of survey and panel members and should therefore be investigated in future studies.
Conclusions
We present a detailed summary and evaluation of relevant aspects for decision-making in the surgical treatment of BSCM. Due to lack of evidence from clinical trials, we performed a standardized multiround Delphi survey with a large number of highly experienced experts to identify consensus and ambiguities. As a result, a summary of typical clinical scenarios and a catalog of various BSCMand patient-related aspects that influence the surgical treatment decision have been defined and interpreted. We thus provide information to support BSCM-focused neurosurgeons in current clinical practice and inform clinical trial design, to ultimately improve the quality of the evidence for the management of BSCM.
Acknowledgments
We would like to thank Murray Turoff, BA, PhD (New Jersey Institute of Technology, Newark, New Jersey), for supervision of study concept and design.
References
1. Horne MA, Flemming KD, Su IC, Stapf C, Jeon JP, et al. Clinical course of untreated cerebral cavernous malformations: a meta-analysis of individual patient data. Lancet Neurol. 2016;15(2):166-173.
2. Flemming KD, Graff-Radford J, Aakre J, Kantarci K, Lanzino G, et al. Population-based prevalence of cerebral cavernous malformations in older adults: Mayo Clinic Study of Aging. JAMA Neurol. 2017;74(7):801-805.
3. Al-Shahi Salman R, Berg MJ, Morrison L, Awad IA. Hemorrhage from cavernous malformations of the brain: definition and reporting standards. Stroke. 2008;39(12):32223230.
4. Flemming KD, Link MJ, Christianson TJ, Brown RD Jr. Prospective hemorrhage risk of intracerebral cavernous malformations. Neurology. 2012;78(9):632-636.
5. Kearns KN, Chen CJ, Tvrdik P, Park MS, Kalani MYS. Outcomes of surgery for brainstem cavernous malformations: a systematic review. Stroke. 2019;50(10):2964-2966.
6. Garcia RM, Ivan ME, Lawton MT. Brainstem cavernous malformations: surgical results in 104 patients and a proposed grading system to predict neurological outcomes. Neurosurgery. 2015;76(3):265-278.
7. Abla AA, Lekovic GP, Turner JD, de Oliveira JG, Porter R, Spetzler RF. Advances in the treatment and outcome of
brainstem cavernous malformation surgery: a single-center case series of 300 surgically treated patients. Neurosurgery. 2011;68(2):403-415.
8. Cornelius JF, KŸrten K, Fischer I, HŠnggi D, Steiger HJ. Quality of life after surgery for cerebral cavernoma: brainstem versus nonbrainstem location. World Neurosurg. 2016; 95:315-321.
9. Dammann P, Herten A, Santos AN, Rauschenbach L, Chen B, et al. Multimodal outcome assessment after surgery for brainstem cavernous malformations. J Neurosurg. Published online October 16, 2020. doi:10.3171/2020.6.JNS201823.
10. Akers A, Al-Shahi Salman R, A Awad I, Dahlem K, Flemming K, et al. Synopsis of guidelines for the clinical management of cerebral cavernous malformations: consensus recommendations based on systematic literature review by the Angioma Alliance Scientific Advisory Board Clinical Experts Panel. Neurosurgery. 2017;80(5):665-680.
11. Poorthuis M, Samarasekera N, Kontoh K, Stuart I, Cope B, et al. Comparative studies of the diagnosis and treatment of cerebral cavernous malformations in adults: systematic review. Acta Neurochir (Wien). 2013;155(4):643-649.
12. Gross BA, Batjer HH, Awad IA, Bendok BR, Du R. Brainstem cavernous malformations: 1390 surgical cases from the literature. World Neurosurg. 2013;80(1-2):89-93.
13. Guyatt GH, Sinclair J, Cook DJ, Glasziou P. UsersÕ guides to the medical literature: XVI. How to use a treatment recommendation. JAMA. 1999;281(19):1836-1843.
14. Milholland AV, Wheeler SG, Heieck JJ. Medical assessment by a Delphi group opinion technic. N Engl J Med. 1973; 288(24):1272-1275.
15. Kumaran KM, Morin F, Rowe M. Consensus taking in a Delphi study. Hosp Adm Can. 1976;18(10):70-72.
16. Kumaran KM, Lemieux M, Satchell G. Problem solving with the Delphi technique. Dimens Health Serv. 1976;53(8):34-35.
17. Wang X, Chen Y, Yang N, Deng W, Wang Q, et al. Methodology and reporting quality of reporting guidelines: systematic review. BMC Med Res Methodol. 2015;15:74.
18. Moher D, Schulz KF, Simera I, Altman DG. Guidance for developers of health research reporting guidelines. PLoS Med. 2010;7(2):e1000217.
19. Khodyakov D, Kinnett K, Grant S, Lucas A, Martin A, et al. Engaging patients and caregivers managing rare diseases to improve the methods of clinical guideline development: a research protocol. JMIR Res Protoc. 2017;6(4):e57.
20. Boulkedid R, Abdoul H, Loustau M, Sibony O, Alberti C. Using and reporting the Delphi method for selecting healthcare quality indicators: a systematic review. PLoS One. 2011; 6(6):e20476.
21. Ospel JM, Brouwer P, Dorn F, Arthur A, Jensen ME, et al. Antiplatelet management for stent-assisted coiling and flow diversion of ruptured intracranial aneurysms: a DELPHI Consensus statement. AJNR Am J Neuroradiol. 2020;41(10): 1856-1862.
22. Zhai XD, Wang CX, Ma YJ, Yu JX, Xiang SS, et al. Using the modified Delphi method to research the influencing factors of long-term health-related quality of life in patients with unruptured intracranial aneurysms after endovascular treatment. Chin Neurosurg J. 2020;6:7.
23. Marone EM, Freyrie A, Ruotolo C, Michelagnoli S, Antonello M, et al. Expert opinion on hostile neck definition in endovascular treatment of abdominal aortic aneurysms (a Delphi Consensus). Ann Vasc Surg. 2020;62:173-182.
24. Etminan N, Brown RD Jr, Beseoglu K, Juvela S, Raymond J, et al. The unruptured intracranial aneurysm treatment score: a multidisciplinary consensus. Neurology. 2015;85(10):881889.
25. Downe-Wamboldt B. Content analysis: method, applications, and issues. Health Care Women Int. 1992;13(3):313-321.
26. Dammann P, Wrede K, Jabbarli R, MŸller O, Mšnninghoff C, et al. Of bubbles and layers: which cerebral cavernous malformations are most difficult to dissect from surrounding eloquent brain tissue? Neurosurgery. 2017;81(3):498-503.
27. Santos AN, Rauschenbach L, Darkwah Oppong M, Chen B, Herten A, et al. Assessment and validation of proposed classification tools for brainstem cavernous malformations. J Neurosurg. Published online October 16, 2020. doi: 10.3171/2020.6.JNS201585.
28. Walicke P, Abosch A, Asher A, Barker FG II, Ghogawala Z, et al. Launching effectiveness research to guide practice in neurosurgery: a National Institute Neurological Disorders and Stroke Workshop Report. Neurosurgery. 2017;80(4):505514.
29. Duffield C. The Delphi technique: a comparison of results obtained using two expert panels. Int J Nurs Stud. 1993;30(3): 227-237.
30. Goodman CM. The Delphi technique: a critique. J Adv Nurs. 1987;12(6):729-734.
31. JŸnger S, Payne SA, Brine J, Radbruch L, Brearley SG. Guidance on Conducting and REporting DElphi Studies (CREDES) in palliative care: recommendations based on a methodological systematic review. Palliat Med. 2017;31(8): 684-706.
32. Poorthuis MH, Klijn CJ, Algra A, Rinkel GJ, Al-Shahi Salman R. Treatment of cerebral cavernous malformations: a systematic review and meta-regression analysis. J Neurol Neurosurg Psychiatry. 2014;85(12):1319-1323.
Disclosures
Dr. Couldwell reports receiving royalties from Bioplate. Dr. Lanzino reports being a consultant for Superior Medical Editors.
Author Contributions
Conception and design: Dammann, Schaller, Lawton, Sure. Acquisition of data: Dammann, Darkwah Oppong. Analysis and interpretation of data: Dammann, Santos, Rauschenbach, Darkwah Oppong, Schmidt, Schaller, Lawton, Sure. Drafting the article: Dammann. Critically revising the article: Dammann, Al-Shahi Salman, Santos, Rauschenbach, Darkwah Oppong, Schaller, Lawton, Sure. Reviewed submitted version of manuscript: all authors. Approved the final version of the manuscript on behalf of all authors: Dammann. Statistical analysis: Schmidt. Administrative/technical/material support: Dammann, Santos, Rauschenbach. Study supervision: Dammann, Couldwell, Ogilvy, Lawton, Sure. Delphi Consensus expert participant: Abla, Al-Shahi Salman, Andrade-Barazarte, Bene?, Cenzato, Connolly, Cornelius, Couldwell, Sola, Gomez-Paz, Hauck, Hernesniemi, Kivelev, Lanzino, Macdonald, Morcos, Ogilvy, Steiger, Steinberg, Spetzler, Schaller, Lawton, Sure.
Supplemental Information
Online-Only Content
Supplemental material is available with the online version of the article.
Supplemental Digital Content 1-5. https://thejns.org/doi/ suppl/10.3171/2021.3.JNS2156.
Previous Presentations
These data were previously presented as an oral (online) presentation at the Annual Meeting of the Deutsche Gesellschaft fŸr Neurochirurgie (DGNC), June 9, 2021, in Erfurt, Germany.
Correspondence
Philipp Dammann: University Hospital Essen, Germany. philipp.dammann@uk-essen.de. J Neurosurg October 1, 2021 11