Surgical treatment of symptomatic cavernous malformations of the brainstem
Surgical treatment of symptomatic cavernous malformations of the brainstem
R. G. Sola 1 ; 2 , P. Pulido 2 , J. Pastor 3 , M. Ochoa 4 , and J. Castedo 5
- 1 Department of Neurosurgery, Hospital de la Princesa, Universidad Autónoma de Madrid, Madrid, Spain
2 Service of Neurosurgery, Hospital Montepríncipe, Madrid, Spain
3 Neurophysiology Section, Hospital de la Princesa, Universidad Autónoma de Madrid, Madrid, Spain
4 Service of Neurology, Hospital Montepríncipe, Madrid, Spain 5 Service of Radiology, Hospital Montepríncipe, Madrid, Spain
Received April 5, 2006; accepted February 15, 2007; published online April 5, 2007 # Springer-Verlag 2007
ABSTRACT
Summary:
Introduction and objectives. Cavernous malformations (CM) at the level of the brainstem, continue to present a challenge in therapeutic terms and are an important source of controversy. Here we present our experience and the results obtained by adopting surgical treatment.
Materials and methods:
The results of a consecutive series of 17 patients were studied. The surgical intervention was designed after: 1. A neurological examination. 2. MRI and cerebral angiography. 3. Correlation with anatomical brainstem maps. The surgical intervention was approached from the most damaged zone or through a zone which was functionally least important.
Results:
Complete extirpation was achieved in 15 patients without mortality. In a few patients the surgical intervention temporarily aggravated the prior lesion of the cranial nerves (2 = 17) or damage new sensory tracts (2 = 17). The functional post-operative recovery was good, in terms of consciousness (4 = 5), cranial nerves (11 = 17), the pyramidal tract (3 = 5) and the cerebellum (2 = 4). Of the patients that were operated, 14 of 17 returned to their professional activities.
Conclusions:
The results of surgery can surpass the morbidity-mortality of the natural history or treatment with radiosurgery. There is a clear consensus in recommending surgical intervention for CMs that are superficially located, in young patients and in those with a risk of further bleeding. It is probably best that the surgery is performed during the subacute period, when the MRI offers a clear image confirming the presence of the CM.
Keywords:
Cavernous malformation; brainstem hemorrhage; intraoperative neurophysiological monitoring; neuronavigation; radiosurgery.
Introduction
The natural history of brainstem cavernous malformations (CMs) indicates a higher morbidity and mortality than CMs in other locations [13]. Radiosurgery appears to diminish the incidence of further haemorrhages but not remove the risk completely [5].
On the other hand, controversy exists regarding the alternative of surgical treatment. Kupersmith et al. [9] stated that the location in the brainstem is an impediment to performing surgical intervention without a significant postoperative morbidity. Meanwhile, neurosurgeons such as Samii et al. [17] have demonstrated a low surgical morbidity with no adverse repercussions on quality of life. In accordance with this latter line of thought, we present the experience and results of a surgical approach to remove brainstem CMs from a consecutive series of 17 patients.
Between December 1991 and January 2005, 20 patients were referred for surgical treatment, having been diagnosed by MRI with a CM in any region of the brainstem. All of them had recently suffered from the clinical effects of symptomatic haemorrhage. Of these, 17 were surgically excised by the senior surgeon (RGS), 9 in the University Hospital de la Princesa (HUP) and 8 in the Hospital Madrid-Montepr íncipe (HMMP). In the other 3 patients, surgical intervention was not considered as the lesion was very small and deep, with only one episode of bleeding and a practically normal neurological situation (2 patients), or due to the general condition of the patient that made such an intervention unwise.
In designing the surgical intervention, several steps were followed: 1. Neurological examination. 2. High resolution MR images and an angiographic study was performed to rule out any other type of vascular malformation. 3. The neurological examination and the MRI were correlated with the anatomic maps of the brainstem [22, 24]
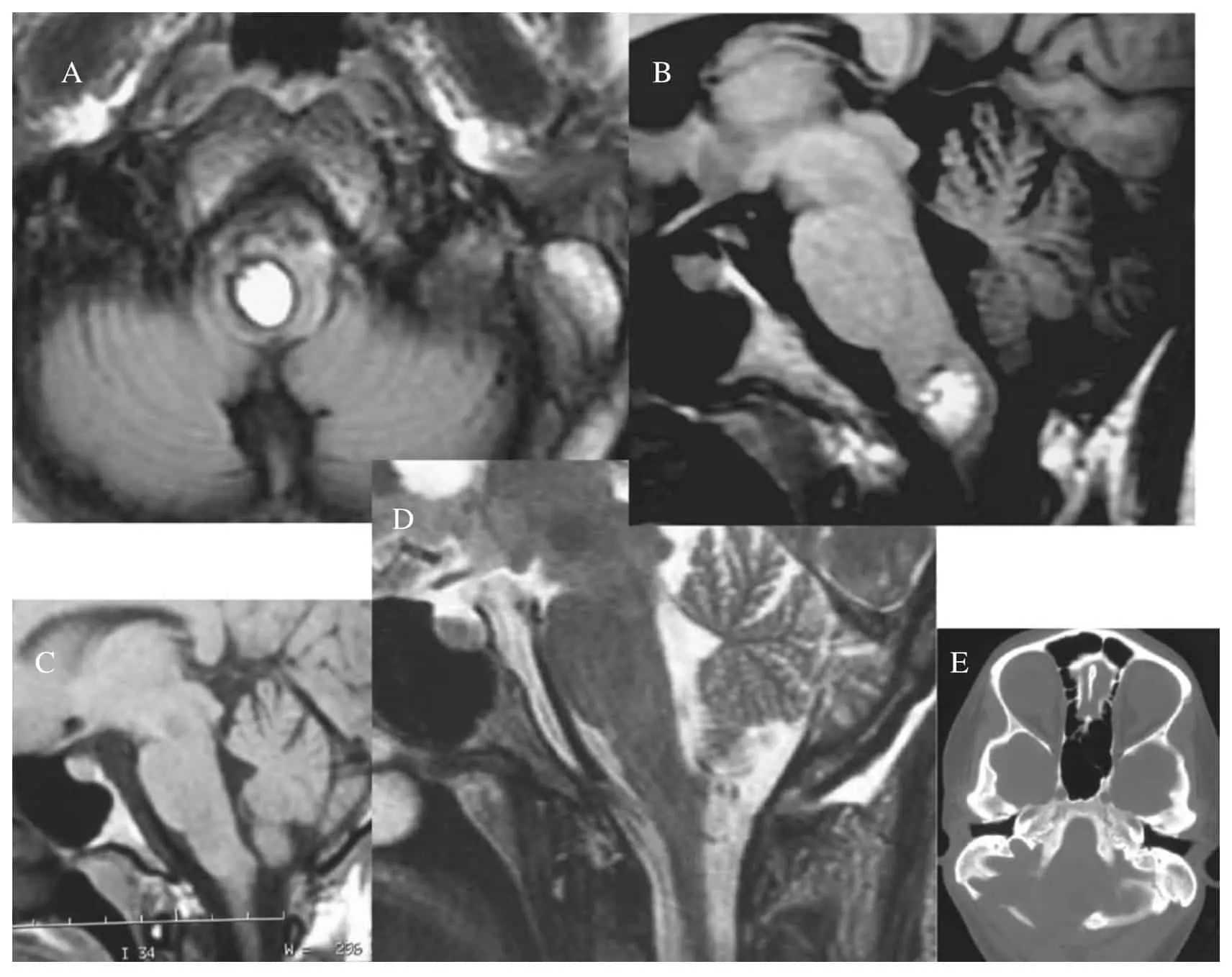
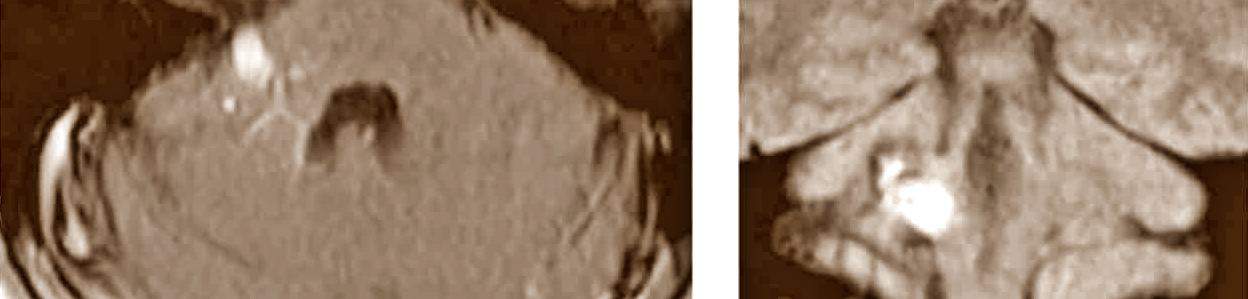
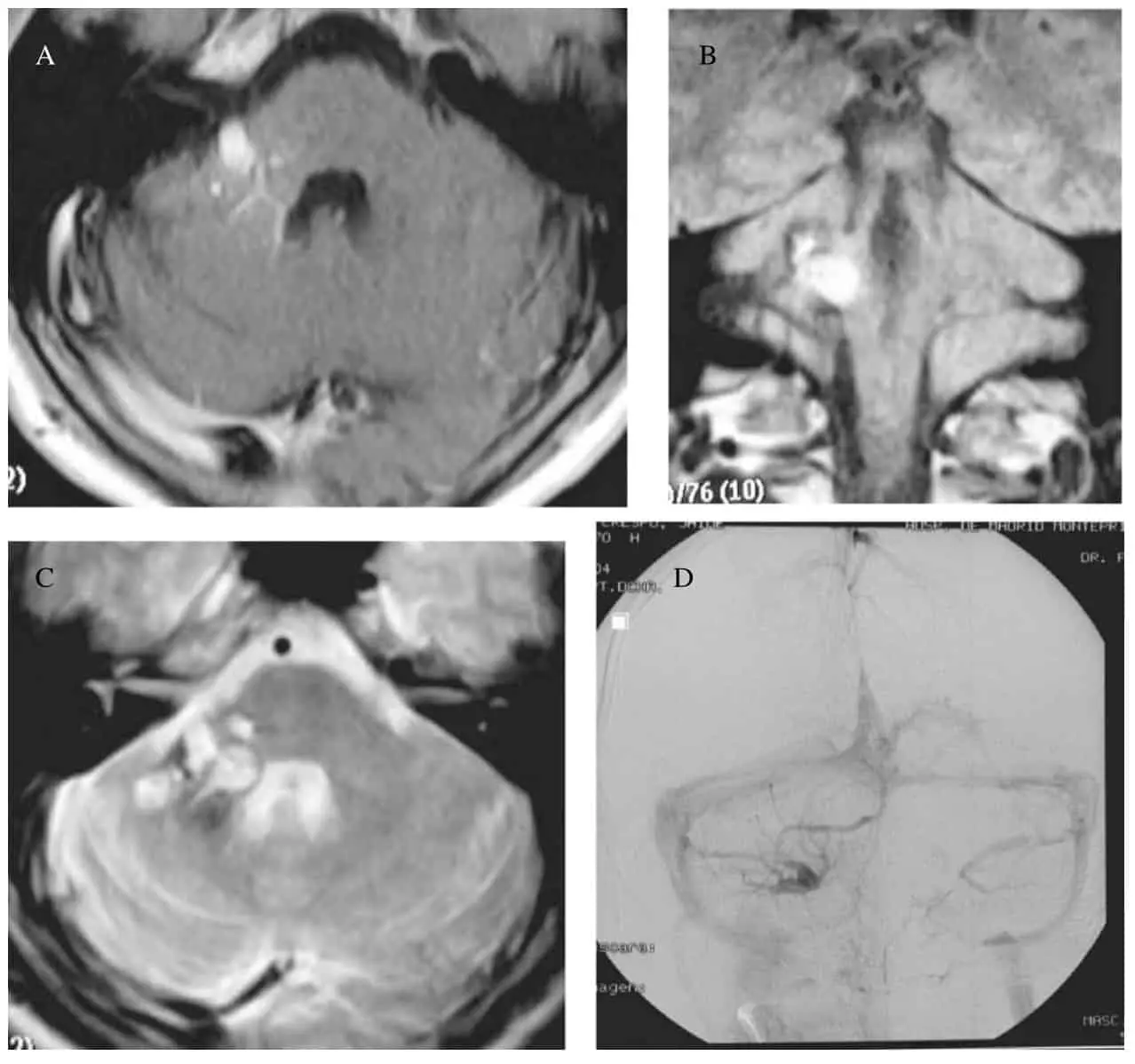
In order to facilitate this approach, our system of image integration was used in one of the 17 patients [15, 18] and neuronavigation techniques (BrainLab + ) were used in patients 16 and 17 (Table 1). The final 7 patients were monitored with neurophysiological techniques to examine somatosensorial and brainstem evoked potentials, as well as controlling other cranial nerves. In all patients the diagnosis of cavernous malformation was confirmed following a pathological study of the tissue removed at operation. Two patients (number 6 and 7), with a CM at the level of the cerebellopontine angle, needed two surgical interventions in order to achieve complete removal (Fig. 1).
CT scan and MRI controls were carried out immediately after the operation, and at 6 months and 1 year after the intervention.
The mean post-operative follow up period was 3.7 + -3 years (maximum of 12 years and a minimum of 1.5 years). The surgical results were analysed in terms of the level of consciousness, focal neurological damage (cranial nerves, pyramidal tracts, sensory tracts and cerebellar function), as well as the Karnofsky Scale [8].
Results
In this study, 10 patients were males and 7 females, aged between 15 and 65 years (mean of 38 years aprox 14.51) (Table 1). Of these, 8 had a clinical history of 2 or more haemorrhages and a syndrome of multiple cavernomatosis existed in 3 of these 8 patients.
The overall neurological situation at the time of surgical treatment was, Coma (2 patients), stupor (1 patient), bradypsychia (2 patients) and normal consciousness in 12 patients.
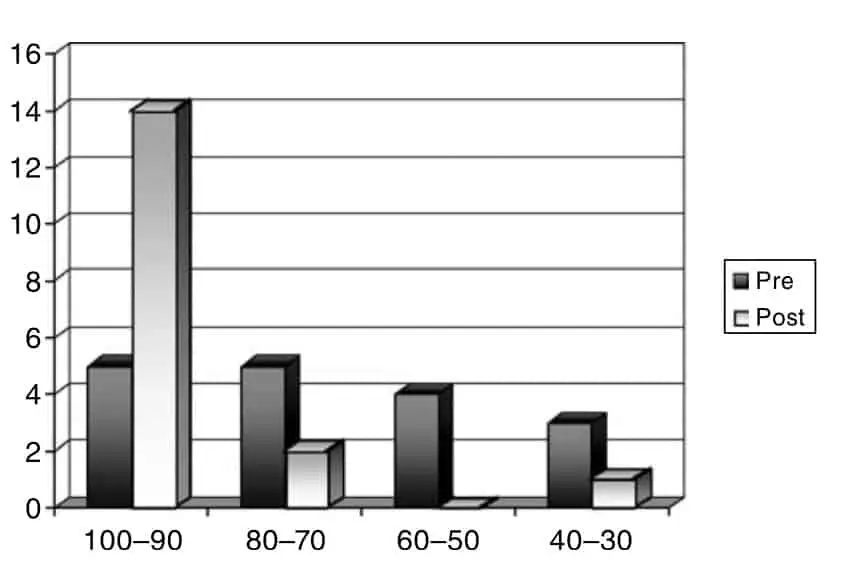
The pre-operative focal neurological deficits consisted of impairment of one or more cranial nerves (17 patients), injury to the pyramidal tract (5 patients), damage to sensory tracts (5 patients) and cerebellar disturbance in 4 patients. At the time of the operation, the Karnofsky Scale scores were 90-100 in 5 patients, 70-80 in 5 patients, 50-60 in 4 patients and 30–40 in 3 patients.
The localisation of the haemorrhage and the CM was in the Midbrain (affecting the III nerve) in 2 patients, Pons (affecting the V, VI, VII or VIII nerves) in 13 patients and the medulla oblongata (affecting lower nerves) in 2 patients.
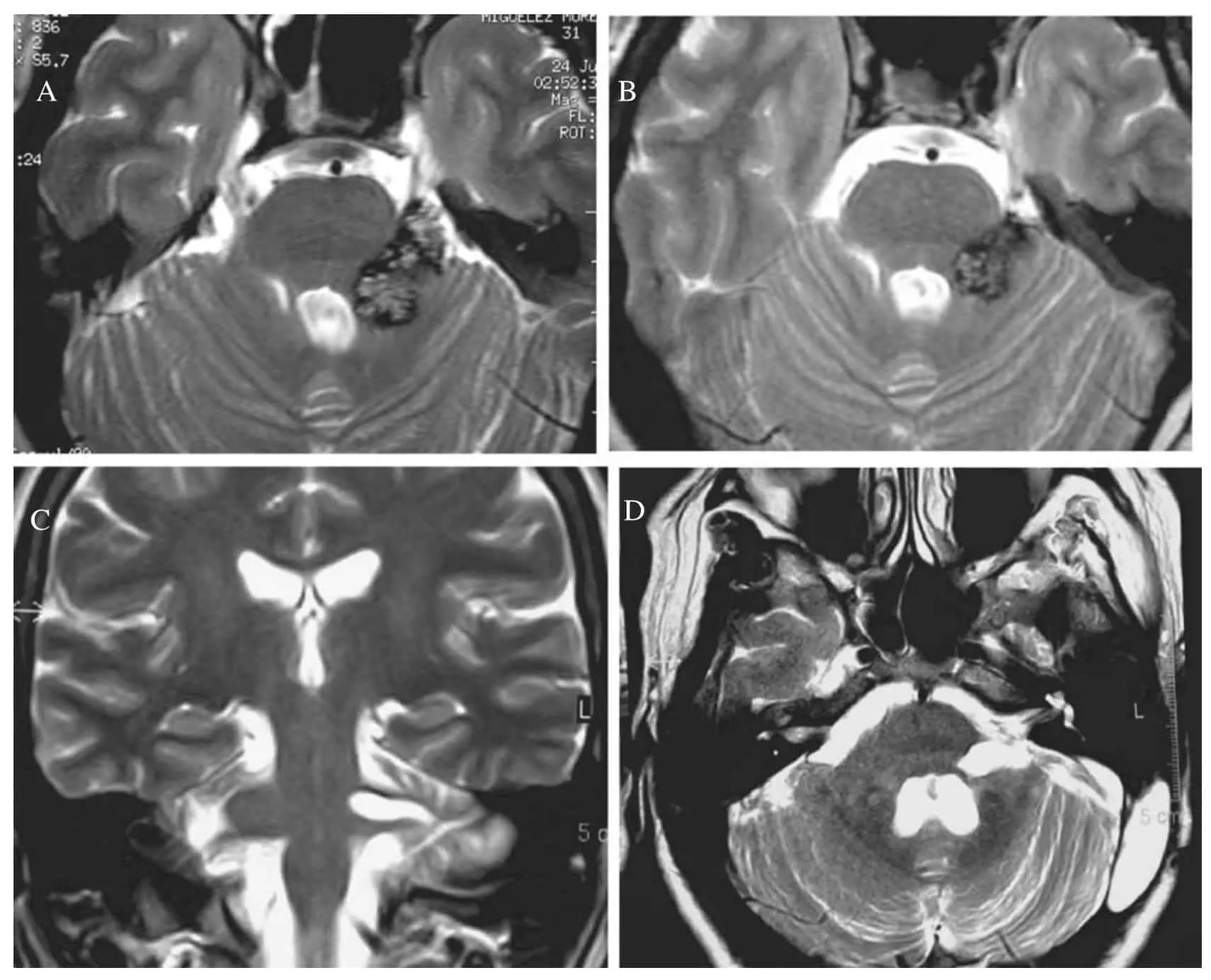
In 14 patients, the CM was superficially located, either erupting at the external surface in contact with the subarachnoid space (13 patients; Fig. 1), or erupting at the floor of the IV ventricle (1 patient). In three patients, the cavernoma was in the deeper regions of the mesencephalon, pons or the medulla oblongata (Fig. 2). The surgical approach was designed in accordance with the neurological examination, MR images and anatomical maps (Table 1). None of the operations were carried out in a seated position. Complete removal was achieved in 15 of the 17 patients, according to the delayed post-operative MRI control. In one patient (number 3), extirpation was not possible due to the presence of a large lesion within the mesencephalon, and surgical resection was interrupted. There were no clinical manifestations of further hemorrhage after 7 years follow up and the patient remains asymptomatic. Another patient (number 10) had a CM at the level of the union of the pons and the medulla oblongata and although during the intervention it appeared that the small CM had been removed, the MRI demonstrated a residual component. There was no surgical mortality, neither immediate nor long term, and there has been no case of post-operative bleeding. In 4 of the 5 patients in whom consciousness was affected post-operatively, a normal situation was reached within a time period of between days to months, in direct relation to the pre-operative level. One patient (number 5), still remained in coma vigil (chronic vegetative state) 7 years later, in a similar condition to the pre-operative state.
In relation to focal neurological lesions, no patient suffered an added disabling neurological lesion. However, in 4 of 16 (25%), excluding patient number 5, new post-operative neurological deficits appeared, with some disturbance of the spinothalamic tract (2 patients) or worsening of the pre-operative damage of the cranial nerves (2 patients). Nevertheless, these deficits remitted in intensity over the following 6-12 months.
The post-operative state of the cranial nerve disturbance was similar to the pre-operative state in 4 of the 17 patients. Another two presented a clear improvement, while in the remaining 11 patients the recovery was practically complete. In relation to the pyramidal tract, a milder or similar lesion to the pre-operative deficit remained in 2 of the 4 patients. A disturbance of the sensory tracts persisted in 5 of the 7 patients, with pre or immediately post-operative damage. In 2 of the 5 patients the pre-operative cerebellar deficit improved but persisted. The majority of the patients have not been disabled and indeed 14 of the 17 patients returned to their previous activities with 90-100 points in the Karnofsky Scale (Table 1).
Discussion
Dandy [2] was the first to successfully operate on a brainstem CM. In 1976, Voigt and Yasargil [20] described a patient with a mesencephalic CM who was operated on successfully and reviewed a series of 164 cases published up to that time. Subsequently, although CMs at the level of the brainstem offered an important surgical challenge [6, 7, 12], several authors [3, 14, 25] presented series with good results and with an improvement of the natural history of this condition. In this context, the series published by Samii et al. [17] and Wang et al. [21] should be highlighted because there was no mortality and little incapacitating morbidity. However, in the most recent series of 52 patients of Ferroli et al. [4] the mortality was 1.9%. In our series we did not observe any mortality, nor did any patient worsen in terms of the level of consciousness after the surgical intervention. Furthermore, 4 of the 5 patients in whom the level of consciousness was affected recovered to a normal state.
The focal neurological deficits were divided this into 4 categories: 1. Cranial Nerves: Lesions were evident in all the patients either objectively or as a symptom(diplopia). The surgical intervention worsened the severity in 2 patients but none developed a new cranial nerve lesion. After intervention a large proportion of patients (11 of 17) recovered the function of the cranial nerves to practically normal levels. 2. Pyramidal tracts: The functional recovery of this tract was apparent in a high percentage of patients (4 of 6). 3. Sensory tracts: It should be emphasised that de novo post-operative lesions appeared in 2 patients, in relation with the chosen path of surgical access (Fig. 3), and recovery also occurred with only 2 patients persisting with a clear lesion of these tracts. 4. Cerebellar: This was the lesion that responded best postoperatively, with a marked recovery in all patients, and a discrete lesion persisting in only 2 patients. As already stated, none of these sequelae have been severe and the majority of the patients (14 of 17) returned to their previous activities (Fig. 4).
These results are similar to those published by Samii et al. [17], with the exception of the percentage of patients suffering cranial nerve disturbance, which was only 72% in that series. This difference is probably due to the fact that we considered the symptom of diplopia as a lesion of the cranial nerves, when it was well correlated with the location of the CM and the anatomical maps. Given the results in the different series, many authors are in favour of surgery as the best alternative to the natural progression of the condition [4, 11, 14, 16, 17, 19, 21]. In the surgical treatment of these lesions, one of the keys is the selection of an appropriate surgical approach [23] and to have up to date equipment available in the operating theatre. All of our patients were operated in the lateral or prone decubitus position, without the complication or discomfort of pneumoencephalus that may occur in the seated position [4]. In general, the approach adopted was based on the modern concepts applied to skull base surgery [14].
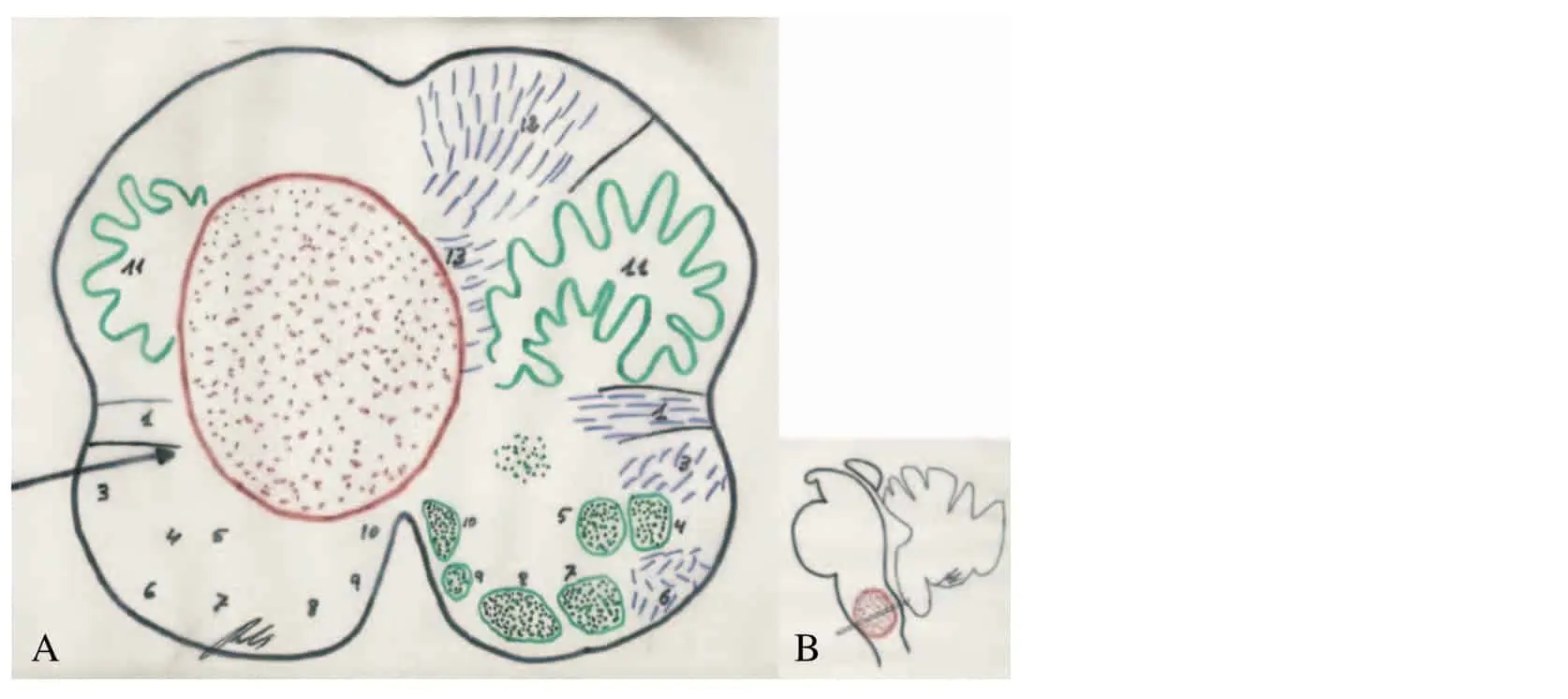
In the patients where the CM did not clearly erupt through the surface, the approach was designed in accordance with the neuroanatomical correlations based on the neurological exampatients). No patients became worse after surgery ination and anatomical maps of the brainstem (Fig. 3). As advised by several authors [11, 19, 25], the techniques of intraoperative neurophyisiological monitoring were used in the last 7 patients and this may have helped to reduce the proportion of post-operative morbidity [16].
The techniques of image integration [15, 18] or neuronavigation were very useful, especially in patient number 16 (Fig. 2), where there were no signs of hemosiderin at the surface of the brainstem, nor clinico-anatomical data from the cranial nerves that could guide us with absolute certainty.
In this patient, as in others, we introduced the end of a very narrow and long Merocel + patty into the hemorrhagic cavity, without using spatulas with the objective of maximal protection of the normal tissue. The aspiration cannula, dissector and the bipolar forceps were rested on the Merocel sponge in the manipulations to remove the CM, which was performed by fragmentation. The microscope used had a long focal length and a high zoom lens (Neuro 4 + , Zeiss). As we experienced in patients 6 and 10 it is important to exert caution and leave behind tissue in the peri-gliotic area which cannot be clearly identified as abnormal (Fig. 1). We believe that it is preferable to reoperate than to continue manipulating a tissue that might be functionally important. Two of our patients (14 and 16) presented a clearly associated venous angioma (Fig. 5), as has been described in up to 16% of patients [25]. In accordance with the policy adopted by other authors [1, 14, 23], we have limited ourselves to resecting the CM only but not the venous angioma.
A controversial study of high risk patients performed by a group of radiosurgery experts with a Gamma Knife presents an alternative to surgical excision which needs to be taken into account [5]. These patients were treated with radiosurgery and an appreciable decrease in the risk of further haemorrhages was observed although, there was a high morbidity associated with the actual radiosurgery [10]. Regarding this point, Samii et al. [17] were very clear in not advising radiosurgery as a first option, claiming that, depending on the exact location, CMs can be successfully extirpated.
There are two other aspects that require further comment. Firstly, when should surgery be undertaken in relation to the last episode of bleeding ? Some authors [3, 11, 19] believe that surgery should be performed before the first month because the post-bleed gliosis can make the extirpation of the CM difficult without damaging the surrounding tissue [25]. Samii et al. [19] found no difference by operating in the subacute period after more than 3 months. In our limited experience, we also favour performing the intervention in the subacute phase once the patient has been stabilized.
Secondly, the criteria for the selection of patients for operation are relevant. Wascher and Spetzler [23] recommend surgery for patients with progressive symptoms, with superficially located CMs where a surgical approach is possible. Samii et al. [17] have made their opinions even clearer and go further in their recommendations. On the one hand they recommend intervention in the case of superficial CMs if the patient is young, even though it might be a serendipitous finding without haemorrhage. On the other, they recommend operating in cases of progressive deterioration, with further haemorrhage, even though the CM may not be superficial. The possibility of reaching deeply located CMs and removing them with a very low morbidity, such as in patients 3, 12, 17 in our series (Figs. 2 and 3), has already been referred to by Mathiesen et al. [11].
Conclusions
The surgical approach to the brainstem in order to remove CMs does not necessarily cause severe morbidity or disability and the global results may be better than natural history or treatment with radiosurgery. Appropriate selection of patients must be made and, in this respect, there is a clear consensus among authors in recommending surgery for superficially located CMs in young patients, and in those at risk of further haemorrhage. It is probably better to operate during the subacute period, assuming that the MRI demonstrates a clear image indicating the presence of a CM. The surgical treatment should be designed in accordance with the neuro-anatomical correlations based on the neurological examination and anatomical maps of the brainstem. The surgical approach essentially uses the concepts and techniques applicable to the skull base and using intraoperative neurophysiological monitoring. New imaging and neuronavigation techniques help to locate and approach CMs in deeper positions.
References
1. Barrow DL, Krisht A (1993) Cavernous malformations and hemorrhage. In: Awad IA, Barrow DL (eds) Cavernous malformations. American Association of Neurological Surgeons, Park Ridge, Illinois, pp 65-80
2. Dandy WE (1928)Venous abnormalities and angiomas of the brain. Arch Surg 17: 715-793
3. Fahlbusch R, Strauss C, Huk W (1990) Surgical removal of pontomesencephalic cavernous haemangiomas. Neurosurgery 26: 449-457
4. Ferroli P, Sinisi M, Franzini A, Giombini S, Solero CL, Broggi G (2005) Brainstem Cavernomas: Long-term results of microsurgical resection in 52 patients. Neurosurgery 56: 1203-1214
5. Hasegawa T, McInerney J, Kondziolka D, Lee JY, Flickinger JC, Lunsford LD (2002) Long-term results after stereotactic radiosurgery for patients with cavernous malformations. Neurosurgery 50: 1190-1198
6. Huhn SL, Rigamonti D, Hsu F (1993) Indications for surgical intervention. In: Awad IA, Barrow DL (eds) Cavernous malformations. American Association of Neurological Surgeons, Park Ridge, Illinois, pp 87-99
7. Isamat F, Conesa G (1993) Cavernous angiomas of the brainstem. Neurosurg Clin N Am 4: 507-518
8. Karnofsky DA, Burchenal JH (1949) The clinical evaluation of chemotherapeutic agents in cancer. In: MacLeod CM (ed) Evaluation of chemotherapeutic agents. Columbia University Press, New York, pp 191-205
9. Kupersmith MJ, Kalish H, Epstein F, Yu G, Berenstein A, Woo H, Jafar J, Mandel G, De Lara F (2001) Natural history of brain stem cavernous malformations. Neurosurgery 48: 47-53
10. Lisa´k R, Vladyka V, Simonova´ G, Vymazai J, Novotny J Jr (2005) Gamma knife surgery of brain cavernous haemangiomas. J Neurosurg Suppl 102: 207-213
11. Mathiesen T, Edner G, Kihlstr € o m L (2003) Deep and brainstem cavernomas: a consecutive 8-year series. J Neurosurg 99: 31-37 12. McCormick PC, Michelsen WJ (1991) Management of intracranial cavernous and venous malformations. In: Barrow DL (ed) Intracranial vascular malformations. American Association of Neurological Surgeons, Park Ridge, Illinois, pp 197-217
13. Moriarity JL, Clatterbuch RE, Rigamonti D (1999) The natural history of cavernous malformations. Neurosurg Clin N Am 10: 411-417
14. Porter RW, Detwiler PW, Spetzler RF, Lawton MT, Baskin JJ, Derksen PT, Zabramski JM (1999) Cavernous malformations of the brain stem: experience with 100 patients. J Neurosurg 90: 50-58 15. Pulido P, Sola RG (1996) Localización anatomo-funcional en la corteza cerebral. Aplicación de un sistema de integración de ima´genes como gu ´ ı a para la resección de lesiones cerebrales corticales. Rev Neurol Suppl 1: S5-S61, www.neurorgs.com
16. Robinson JR, Awad IA, Little JR (1991) Natural history of the cavernous angioma. J Neurosurg 75: 709-714
17. Samii M, Eghbal R, Carvalho GA, Matthies C (2001) Surgical management of brainstem cavernomas. J Neurosurg 95: 825-832 18. Sola RG, Pulido P (1991) Trans-fissural or trans-sulcal approach versus combined stereotactic-microsurgical approach. Acta Neurochir Suppl 52: 22-25
19. Steinberg GK, Chang SD, Gewirtz RJ, Lopez JR (2000) Microsurgical resection of brainstem, thalamic and basal ganglia angiographically occult vascular malformations. Neurosurgery 46: 260-271
20. Voigt K, Yasargil MG (1976) Cerebral cavernous haemangiomas or cavernomas: incidence, pathology, localization, diagnosis, clinical features and treatment. Review of the literature and report of an unusual case. Neurochirurgia (Stuttg) 19: 59-68
21. Wang CC, Liu A, Zhang JT, Sun B, Zhao YL (2003) Surgical management of brain-stem cavernous malformations. Report of 137 cases. Surg Neurol 59: 444-454
22. Warner JJ (ed) (2001) Atlas of neuroanatomy. Butterworth Heinemann, Boston
23. Wascher ThM, Spetzler RF (1993) Microsurgical treatment of infratentorial cavernous malformations. In: Awad IA, Barrow DL (eds) Cavernous malformations. American Association of Neurological Surgeons, Park Ridge, Illinois, pp 117-132
24. Williams PL, Warwick R (ed) (1975) Functional neuroanatomy of man. Churchill Livingstone, Edinburgh
25. Zimmerman RS, Spetzler RF, Lee KS, Zabramski JM, Hargraves RW (1991) Cavernous malformations of the brain stem. J Neurosurg 75: 32-39
Correspondence: Rafael G. Sola, Department of Neurosurgery, Hospital de la Princesa, Diego de León 62, 28006 Madrid, Spain. e-mail: rgsola@neurorgs.com
Volver a Investigación
Volver a Patología Vascular
Ver documento Surgical treatment of symptomatic cavernous malformations of the brainstem