Long-term results of posteromedial hypothalamic deep brain stimulation for patients with resistant aggressiveness
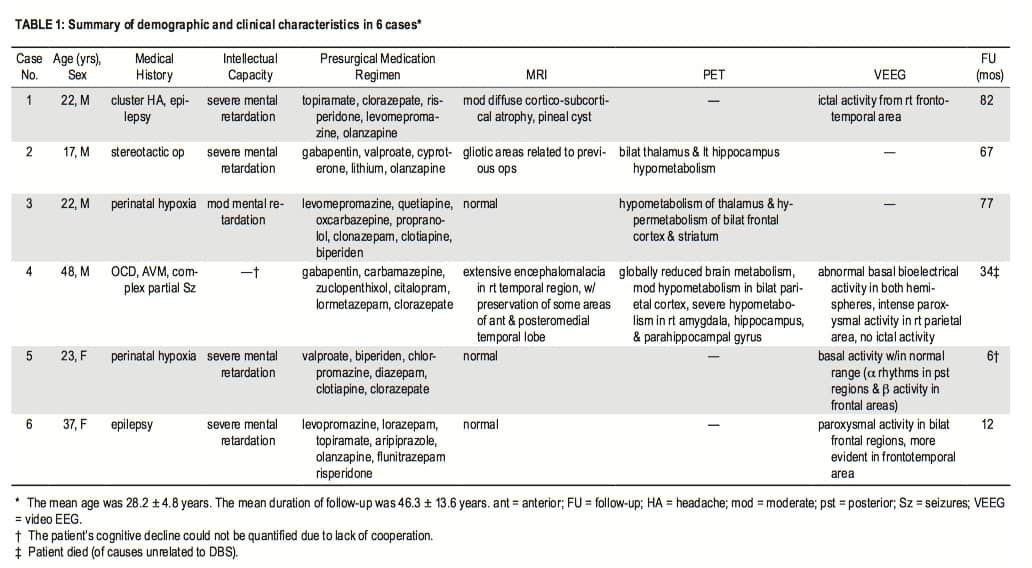
Posteromedial hypothalamic deep brain stimulation
Artículo escrito por la Dra. Cristina Torres Díaz y publicado por la revista JNS – Journal of Neurosurgery
Clinical article
See the corresponding editorial, DOI: 10.3171/2013.1.JNS122308.
DOI: 10.3171/2013.4.JNS121639. ©AANS, 2013
Cristina V. Torres, M.D., Ph.D.,1 Rafael G. Sola, M.D., Ph.D.,1
Jesús Pastor, M.D., Ph.D.,2 Manuel Pedrosa, M.D., Ph.D.,1 Marta Navas, M.D.,1 Eduardo García-Navarrete, M.D., Ph.D.,1 Elena Ezquiaga, M.D., Ph.D.,3 and Eduardo García-Camba, M.D., Ph.D.3
1 Departments of Neurosurgery, 2 Clinical Neurophysiology, and 3 Psychiatry, University Hospital La Princesa, UAM, Madrid, Spain
Posteromedial hypothalamic DBS for intractable erethism
Object. Erethism describes severe cases of unprovoked aggressive behavior, usually associated with some degree of mental impairment and gross brain damage. The etiology can be epileptic, postencephalitic, or posttraumatic, or the condition can be caused by brain malformations or perinatal insults. Erethism is often refractory to medication, and patients must often be interned in institutions, where they are managed with major restraining measures. The hypothalamus is a crucial group of nuclei that coordinate behavioral and autonomic responses and play a central role in the control of aggressive behavior. deep brain stimulation (DBS) of the posteromedial hypothalamus (PMH) has been proposed as a treatment for resistant erethism, although experience with this treatment around the world is scarce. The objective of this study was to examine the long-term outcome of PMH deep brain stimulation (DBS) in 6 patients with severe erethism treated at the authors’ institution.
Methods. Medical records of 6 patients treated with PMH DBS for intractable aggressiveness were reviewed. The therapeutic effect on behavior was assessed by the Inventory for Client and Agency Planning preoperatively and at the last follow-up visit.
Results. Two patients died during the follow-up period due to causes unrelated to the neurosurgical treatment. Five of 6 patients experienced a significant reduction in aggressiveness (the mean Inventory for Client and Agency Planning general aggressiveness score was -47 at baseline and -25 at the last follow-up; mean follow-up 3.5 years). Similar responses were obtained with lowand high-frequency stimulation. In 4 cases, the patients’ sleep patterns became more regular, and in 1 case, binge eating and polydipsia ceased. One of the 3 patients who had epilepsy noticed a 30% reduction in seizure frequency. Another patient experienced a marked sympathetic response with highfrequency stimulation during the first stimulation trial, but this subsided when stimulation was set at low frequency. A worsening of a previous headache was noted by 1 patient. There were no other side effects.
Conclusions. In this case series, 5 of 6 patients with pathological aggressiveness had a reduction of their outbursts of violence after PMH DBS, without significant adverse effects. Prospective controlled studies with a larger number of patients are needed to confirm these results.
(http://thejns.org/doi/abs/10.3171/2013.4.JNS121639)
Key Words • aggressiveness • deep brain stimulation • erethism • posteromedial hypothalamus • functional neurosurgery
Abbreviations used in this paper: AVM = arteriovenous malformation; DBS = deep brain stimulation; EEG = electroencephalography; ICAP = Inventory for Client and Agency Planning; OCD = obsessive-compulsive disorder; PMH = posteromedial hypothalamus.
Introduction
Erethism describes severe cases of unprovoked aggressive behavior, usually associated with some degree of mental impairment and gross brain damage.33 The etiology can be epileptic, postencephalitic, or posttraumatic, or the condition can be due to brain malformations or perinatal insults. Erethism is usually accompanied by hyperkinesia, destructiveness of objects, and self-aggressiveness; the condition is often refractory to medication and electroconvulsive therapy, and patients may need to be interned in institutions, where they are managed with major restraining measures.12,19.
To alleviate these symptoms, a few stereotactic surgical procedures have been performed, including dorsomedial thalamotomy, fornicotomy, subfrontal gyrectomy, cingulectomy, and amygdalotomy, with different degrees of success.19,29 In 1970, Sano and coworkers33 published their first series of cases in which posteromedial hypothalamotomy was performed for the treatment of medication-resistant erethism. The lesions were located in an area 2 mm below the midcommisural point and 2 mm lateral to the wall of the third ventricle, where high-frequency stimulation caused marked sympathetic responses, diffuse delta waves in EEG and ipsilateral ocular deviation.32,33 The procedure was effective to some degree in 95% of the patients and yielded satisfactory results in 84% of them.32,33 These results have been replicated by other groups,3,29,36 and posteromedial hypothalamotomy has been performed in large numbers of patients.
Intelligence was not impaired, and no significant side effects were noticed after the operations.
The hypothalamus is a crucial group of nuclei that process external information from frontal areas and send output fibers to lower systems that coordinate behavioral and autonomic responses.19,39,46,48 A part of the mediobasal hypothalamus (known as the “aggression area”)14,38,48 plays a central role in the control of aggressive behavior;9,34 electrical stimulation of this area elicits biting attacks in cats and rodents.9,22,34,47,48 There are some recent studies suggesting that efferent and afferent connections overlap in many regions, in turn suggesting that reverberating circuits are important in the control of aggressiveness.47 The trophotropic system includes the preoptic area, the posterior orbital area, the insula, the uncus, and the amygdala complex, while the ergotropic system consists of the posterior part of the hypothalamus, the nucleus anterior thalami, the cingulum, and the hypocampus.15,16 It has been proposed that the integrated function of the ergotropic and trophotropic circuits is essential in the expression and experience of emotion.15,16 Some studies suggest that the limbic system is activated mainly by the hypothalamic activating system, the posterior hypothalamus. Accordingly, posteromedial hypothalamotomy would result in the elimination of the ergotropic zone and decreased activation of the limbic system.
Deep brain stimulation has widely replaced the ablative procedures as a therapy in neurological and psychiatric disorders. The clinical benefits of lesioning target structures appear to be similar to those achieved by chronic stimulation of these structures. Moreover, deep brain stimulation (DBS) provides the added benefit of reversibility and titratability, as well as a low risk of complications.
To our knowledge, there are only 9 published cases of erethism treated with PMH DBS.11,12,13,19,20 In 2008, Franzini et al.12 presented their results using high-frequency stimulation in 2 patients with intractable aggressiveness. In 2012, the same group reported on a series of 7 patient cases, in which 6 of 7 patients had derived benefit from stimulation.11,13 In 2008, our group published the longterm outcome of the first case of erethism successfully treated with low-frequency stimulation of the PMH. Preoperatively, the patient exhibited uncontrollable bouts of aggression on a daily basis and remained in chair or bed restraints most of the day. At 18 months of follow-up, he was returning to live with his family during weekends and was able to participate in a specialized community facility. He occasionally showed disruptive behavior that was easily managed.19 Kuhn et al.,21 in 2008, reported a complete cessation of the self-mutilation behavior in a patient with posttraumatic brain injury 4 months after the onset of high-frequency stimulation of the PMH.
In these studies, both highand low-frequency stimulation seemed beneficial in reducing the patients’ symptoms. Although this phenomenon has also been observed with DBS for other disorders, the number of patients treated with PMH DBS is too small to allow for any meaningful conclusions.
The objective of this study is to examine the longterm outcome PMH DBS in 6 patients with severe erethism treated in our institution. The effect of lowand high-frequency stimulation on behavior was assessed. Other domains of interest, such as changes in weight, sleep pattern, and seizure frequency in patients with epilepsy, were evaluated.
Methods
Patient Group
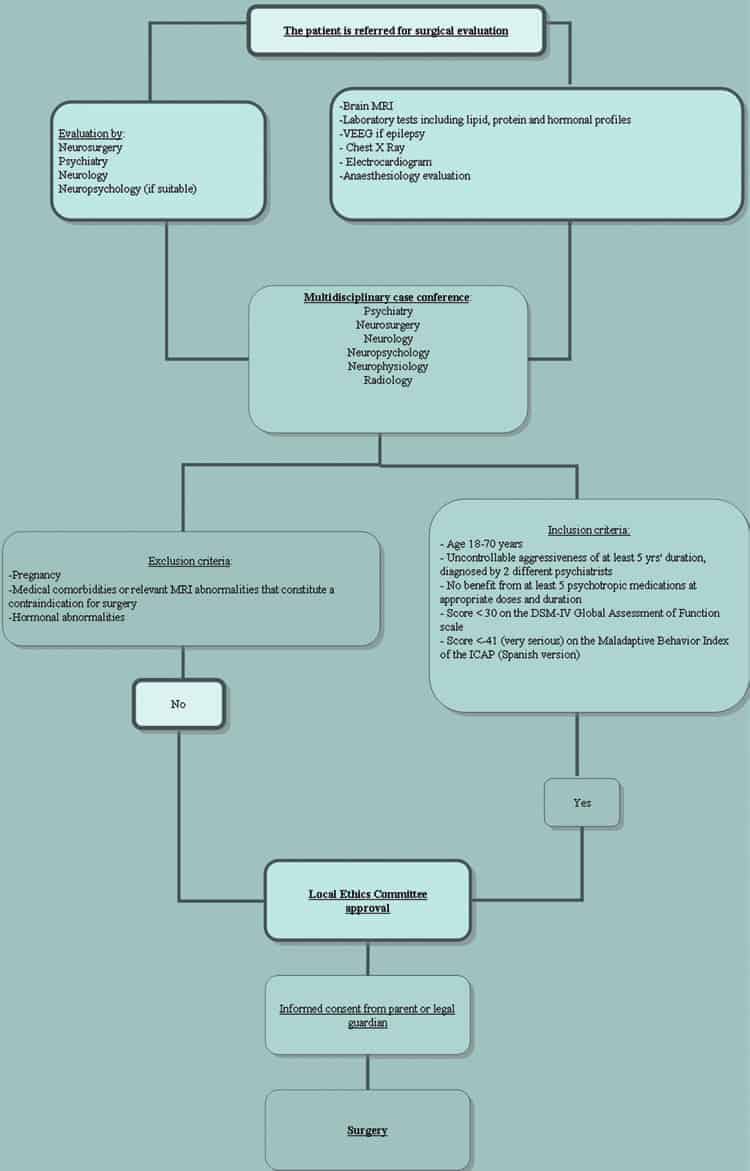
Medical records of 6 patients with medication-resistant erethism treated with PMH DBS at our institution since January 2005 were evaluated. In all cases, uncontrollable refractory aggressiveness of at least 5 years’ duration had been diagnosed by at least 2 different psychiatrists. In each case, at least 5 psychotropic medications had been tried (at appropriate doses and with appropriate treatment duration) without benefit, and all 6 patients were being managed in specialized institutions. Their scores on the DSM-IV Global Assessment of Functioning1 scale were below 21, and their scores on the Maladaptive Behavior Index of the Spanish version of the ICAP5,6 were below -40 (very serious). None of the patients had any medical comorbidities that constituted a contraindication for surgery (Fig. 1).
All patients underwent a complete preoperative evaluation, which involved 1.5-T MRI; laboratory tests, including lipid, protein, and hormonal profiles (measuring basal levels of luteinizing hormone, follicle-stimulating hormone, thyroid-stimulating hormone, testosterone, and cortisol); and psychiatric, neurosurgical, and neurological assessment. Neuropsychological evaluation was not possible due to the patients’ low IQs and lack of cooperation. Four patients were evaluated by video-EEG—because of suspicion of seizures in 2 cases and because of an established diagnosis of epilepsy in the other 2. Epilepsy was confirmed in 3 of these 4 patients. Recording was performed with scalp electrodes placed according to the International 10–20 System. Three of the 6 patients had undergone a previous positron emission tomography (Table 1).
Each case was discussed at a multidisciplinary conference, where the patients were evaluated as DBS candidates. The local ethics committee also evaluated each case, and surgical treatment was approved on the basis of chronicity and severity of the condition and refractoriness to conservative treatment. The patients’ parents and legal guardians were informed of the purpose of the treatment and its potential risks and benefits in detail. They were also informed about the possibility of unknown risks associated with an untested target and patient profile, in addition to the risks of DBS for other conditions. Moreover, the patients’ parents and legal guardians were made aware that the treatment might not produce any benefit and that a second procedure to remove the system might be necessary in case of treatment failure. Informed consent was obtained in all cases.
Five of the 6 patients had developmental delay since birth or demonstrated regression below a developmental age of 2. They showed hyperkinesis and progressive violent behavior and were diagnosed with infantile autism and pervasive developmental disorder. In 2 of these 5 cases, the patients had perinatal hypoxia due to complicated births, and in the remainder, the etiology of the disorder was unknown. The results of MRI were normal or showed nonspecific lesions unrelated to the clinical picture in these 5 cases. Two of these patients had comorbid generalized seizures. One of these patients had undergone 4 previous stereotactic operations for lesioning of the bilateral left stria terminalis, anterior cingulum, internal capsule, right PMH, and dorsomedial and intralaminar thalamic nuclei. This patient initially benefited from each of these operations, but the positive effect wore off a few months after every surgery.
The sixth patient showed normal development until the age of 14, when he started having progressive behavioral disturbances, consisting of obsessions, compulsions, and aggressiveness. When he was 18 years old, he had a hematoma. During evaluation of the hematoma, a temporal lobe AVM was identified, and he underwent resection of the AVM along with evacuation of the hematoma. His symptoms were significantly aggravated after the operation; he developed severe aggressiveness, hypersexuality, disinhibition, bradypsychia, and hyperphagia. Organic mental disorder and obsessive-compulsive disorder were diagnosed. This patient also had partial complex seizures in relation to his temporal lobe AVM.
All 6 patients had daily bouts of uncontrollable aggressiveness directed against objects, caregivers, and themselves. Four of them required restraint measures on a daily basis. Five had severe sleep disturbances in relation to restlessness. Two patients had hyperphagia, and one of them had polydipsia.
Surgery
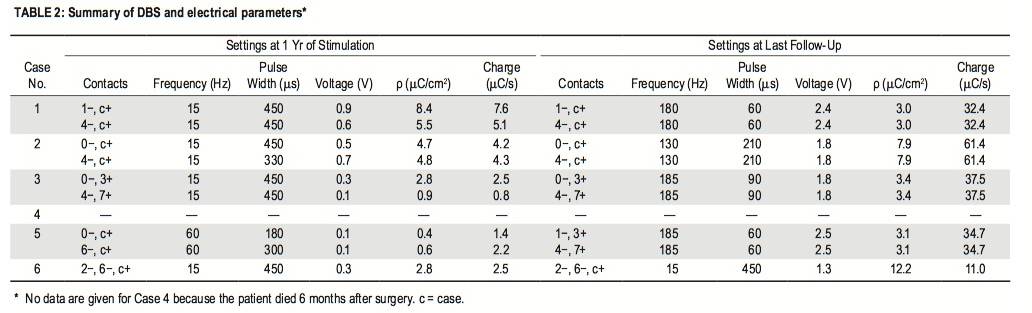
Surgery was performed as previously described.19 Preoperatively, mounting of the stereotactic frame (Leksell model G, Elekta Instruments) and subsequent CT examination were performed with the patients in a state of general anesthesia. The anterior and posterior commissures were identified using axial 3D T1-weighted inversion recovery axial MR images. Images were transferred to a BrainLAB neuronavigation station and reformatted. Target coordinates for the PMH were determined; the tentative coordinates were 2 mm lateral to the wall of the third ventricle, at the midcommissural point, and 2 mm inferior to the anterior commissure–posterior commissure line. Microelectrode recordings and stimulation were used to map the target and surrounding regions using 3 FHC semi-microelectrodes (impedances 0.9–2.1 × 106 W, separated by 2 mm) and the Leadpoint microelectrode recording system (Medtronic). Physiological response to electrical stimulation was assessed by means of electrocardiography, electromyography of cervical muscles (using 2 monopolar needle electrodes), and EEG (using subcutaneous electrodes placed in Fp1/Fp2, T3/ T4, and O1/O2; International 10–20 system). Electrical stimulation was performed using the reference contact of the microelectrode as a cathode and the insertion cannula as an anode. We used a train of 6 seconds at 60 Hz, with a pulse width of 60 ms and intensity of 3 mA.
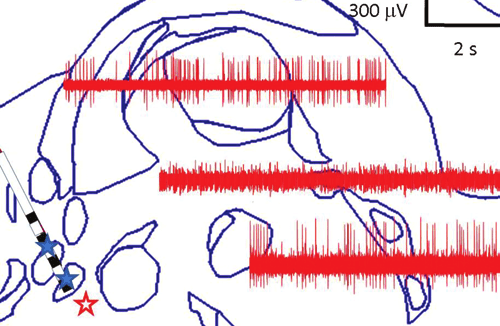
The area where we aimed to insert the electrodes showed neuronal activity suggestive of a nucleus, and if stimulation was applied to it, the patient had sympathetic responses, generalized theta-wave activity in the scalp electroencephalogram, ipsilateral cervical muscular tone increase, and downward deviation of the ipsilateral eye (Fig. 2). Once the target was localized, we implanted a DBS lead (Model 3389, Medtronic) at that site. On the same or different day, the leads were connected to battery-operated, programmable neurostimulators (Kinetra, Medtronic), implanted under general anesthesia in the infraclavicular chest wall above the pectoralis fascia.
Postoperative Management
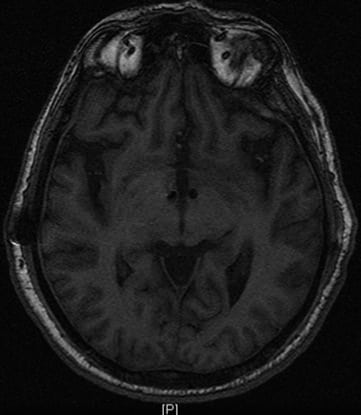
A postoperative MRI study was performed to verify the position of the electrodes and to rule out any complications (Fig. 3). The first trials of stimulation were performed during the first postoperative days, under continuous electrocardiographic and electroencephalographic monitoring, to detect and avoid adverse effects. Followup evaluations were planned for 1, 3, and every 6 months after the surgery, although the frequency was adjusted according to the patients’ clinical situation and accessibility.
All 6 patients had daily bouts of uncontrollable aggressiveness directed against objects, caregivers, and themselves. Four of them required restraint measures on a daily basis. Five had severe sleep disturbances in relation to restlessness. Two patients had hyperphagia, and one of them had polydipsia.
The primary outcome measure was the ICAP. This scale is one of the most widely used adaptive behavior assessments and can be used to assess children and adults with developmental disabilities. It measures the level of support that the patient needs and consists of 3 subscales, corresponding to self-aggressiveness, aggressiveness toward others, and asocial behavior. Scores are as follows: normal, +10 to –10; marginally serious, –11 to –20; moderately serious, –21 to –30; –serious, –31 to –40: and very serious –41 or lower.5 The effect of the surgery in reducing aggressiveness was assessed by comparing the patients’ presurgical baseline scores with their “on” stimulation scores at the last postoperative follow-up, as determined by a specialized psychiatrist.
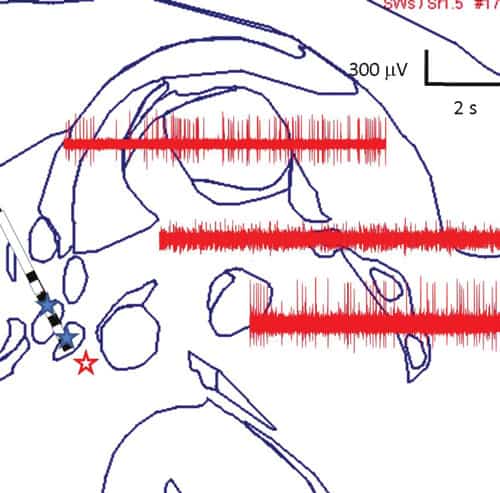
electrode lead, placed in the PMH in a patient with severe intractable
aggressiveness
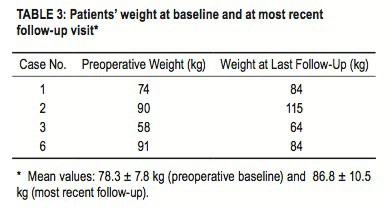
Changes in the postoperative medication were allowed. Laboratory tests, including lipid, protein, and hormone profiles, were performed every 3 months during the first 2 years of stimulation and every year thereafter. Weight measurements were performed at the preoperative baseline assessment and at the last follow-up.
Electrical Parameters
Electrical safety is a very important issue in DBS, because electricity can cause tissue injury when inappropriately applied.28 Although there is a debate about the limit
for safe stimulation, the manufacturer gives a threshold of 30 mC/cm2/phase. We have calculated the maximum density (r ) of charge according to the following equamax
tion: rmax = (Dt × V)/(S × R), where Dt represents the pulse width (msec), V the voltage (V), S the surface area of the active contact (0.0599 cm2 for lead model 3389), and R the resistance (W).
Another theoretical relevant parameter is the total (q, in mC/sec) charge applied per train, because the effect on the neurons depends on this magnitude. We have used the following expression for its calculation: q = rmax × S × n, where n is the frequency (in Hz)—that is, the number of pulses per second.
Statistical Analysis
Statistical comparisons between groups were performed using the paired Student t-test for normal distributions or the Mann-Whitney rank-sum test if normality was not demonstrated. Normality was evaluated using the Kolmogorov-Smirnov test. Statistical analysis was performed using SPSS 17.0 software. The significance level was set at p = 0.05. Results are shown as the mean ± SEM, except where otherwise indicated.
Results
We studied 6 consecutive cases involving 4 male and 2 female patients with medication-resistant erethism (Table 1). Their mean age at the time of surgery was 28 years (range 22–48 years).
The mean duration of postoperative follow-up was 46 months (range 6–82 months). The first patient to undergo surgery experienced marked sympathetic responses, consisting of tachycardia and high blood pressure, when high-frequency stimulation was applied during the first stimulation trial, so the initial settings used were 15 Hz and 450 msec. At the 15-Hz frequency, no variations in his baseline measures were noted. We gradually switched to high-frequency stimulation over the first 2 years of follow-up. The slow rate of increase was due to concern about the patient’s chronic cluster headaches, which had worsened with the stimulation (Table 2).
High-frequency stimulation was eventually well tolerated. All of the remaining patients had the same initial settings, except for one, who had initial stimulation at 60 Hz and 180 msec. In general, the voltage and frequency were slowly increased over the course of the second year of stimulation, regardless of the obtained response, to explore the possibility of a greater improvement in behavior; the pulse width was decreased to maintain the maximum charge density below the limit established. There were no changes in the stimulating contacts, except in one case in which the patient’s condition did not improve with the stimulation.
Two patients died of causes unrelated to the implantation of the electrodes or the stimulation. In one of these 2 cases, death was due to a complication of orthopedic surgery, 2 years and 10 months after the implantation. The patient had OCD and had not derived any benefit from the stimulation or noticed any side effects. He was lost to follow-up 2 years after the implantation. The other death was a consequence of a stroke 6 months after the surgery. The patient did not experience any acute behavioral changes, but her family had noted a progressive reduction in the violent attacks and some improvement in her sleep pattern over the 6 months before her death.
Four patients continue to receive stimulation as of this writing. Three of them experienced an improvement in behavior from the first month of DBS, and the daily outbursts of anger and violence quickly became less frequent or disappeared. This reduction in the frequency and intensity of the aggressive spells was associated with a more regular sleep pattern, better sociability, and a decrease in the need for mechanical restraint so that family care became possible during weekends and holidays. These patients are all able to engage in social activities at the therapeutic community facility they are attending. The last patient’s general behavior and sleep disturbances have not been affected by the stimulation, but her selfaggression has stopped. This improvement was followed by the disappearance of severe skin lesions she had on her chest before the operation. After the first months of the stimulation, the patients condition became stable and has remained so since, except for discrete fluctuations that were mostly related to stressful events. Changes in frequency, pulse width, and amplitude were not followed by significant clinical effects.
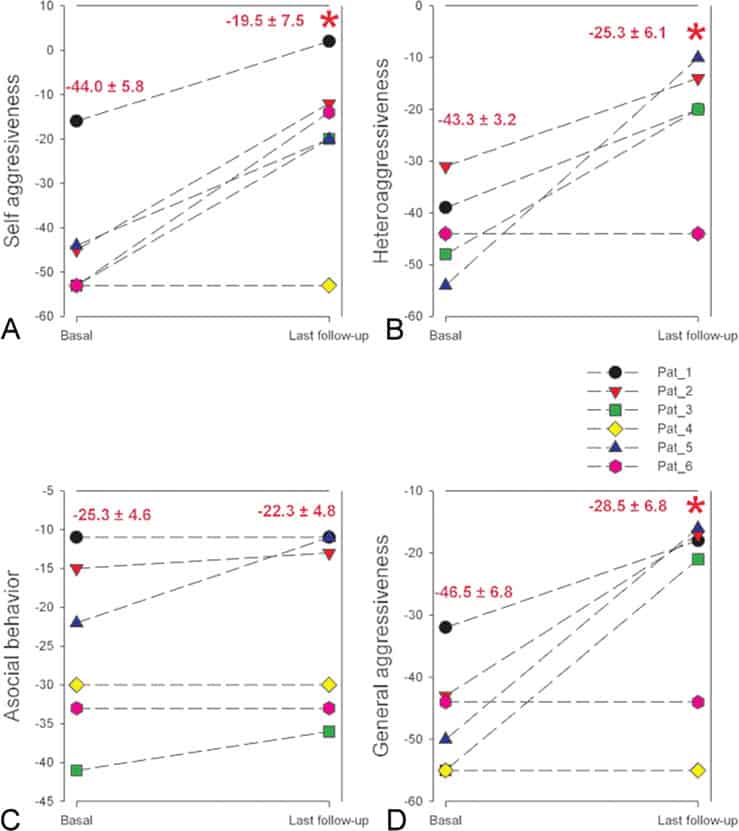
These observations have been supported by a reduction in the aggressiveness scores with respect to the baseline (Fig. 4) in 5 of the 6 patients. The differences between the mean preoperative scores and the mean onstimulation scores at the most recent follow-up were statistically significant, except for the asocial behavior subscores.
One of 3 patients with epilepsy experienced a 30% reduction in the frequency of his generalized seizures. The mean preoperative weight for the 4 patients who continue to receive stimulation was 78.3 ± 7.8 kg, whereas the mean weight at the longest follow-up was 86.8 ± 10.5 kg; the difference was not statistically significant. Three of the 4 patients gained weight (mean increase 13.7 kg, Table 3). According to caregivers, the patients’ physical activity decreased as hyperactivity was controlled. One patient had a reduction in food and water intake, which had been considered excessive before the stimulation. This patient had lost 7 kg over the course of follow-up.
One patient experienced a sympathetic response with high-frequency stimulation during the first stimulation trial. However, this response did not occur when stimulation was set at 15 Hz or when it was gradually increased during the follow-up. The same patient reported an increase in the frequency of his chronic cluster headaches, which could be controlled with medication. We noticed an increase in the free T4 level in one patient 4 months after the onset of stimulation, which remained in the high range of normality and did not cause any clinical effect (data not shown). No other side effects were noted.
Discussion
Erethism is a chronic disorder, imposing considerable suffering and significantly impairing affected individuals’ ability to interact socially. Behavioral improvement was observed in 5 of the 6 erethic patients who underwent PMH DBS at our institution. This reduction of the bursts of uncontrolled violence remained stable in all of the 4 patients who had long-term follow-up.
A similar response was observed with lowand highfrequency stimulation of the PMH. Despite the therapeutic efficacy of Deep Brain Stimulation (DBS), there are still several controversies about its mechanism of action.19 A considerable amount of data has been produced experimentally to address this issue, and a large number of potential mechanisms have been proposed, including depolarizing block, energy depletion, synaptic failure, channel blocking, or anterograde or retrograde propagation.
The issue is made even more complex by the fact that clinical results are the sum of multiple independent effects produced by the current applied to the brain.26 The same phenomenon has been observed with DBS as a treatment for other disorders, such as dystonia, in which different parameters led to symptom control.19 In some cases, it is the advent of adverse effects that determines the stimulation frequency, as in DBS of the pedunculopontine area for gait disorders in Parkinson disease.2,4,8,20,37,42 Moreover, the beneficial effects of stimulation can take weeks or months to manifest, which makes unclear what stimulation amplitudes, pulse durations, and frequencies are most effective for new therapeutic indications. Finally, an insertion effect could not be discounted as the reason for the sustained improvement in some of our patients, since the pulse generators were turned on in the first days after the operation, and assessment was not performed during a postoperative nonstimulation period. Low-frequency stimulation was selected for our first patient because high frequencies elicited intolerable adverse effects in the initial testing. The setting was subsequently adjusted to higher frequencies, with good tolerance. We consider that performing an initial testing over frequencies from 10 to 130 Hz, under monitored conditions, is fundamental for avoiding potentially severe side effects.
The difference between the preoperative and last follow-up ICAP scores for aggressiveness (both selfdirected and outward-directed) was statistically significant, while scores for asocial behavior failed to show significant differences. In our series of patients, PMH DBS was undertaken to decrease aggressiveness. In contrast to the subscales corresponding to self-aggressiveness and outwardly directed aggressiveness, which are calculated based on the number and severity of the patients’ violent behaviors, many aspects of behavior that are rated by the ICAP asocial behavior subscores, such as refusing to share, or talking too loudly, were not expected to be modified by stimulation in this group of patients with baseline mental retardation.
Given the small number of patients who have undergone hypothalamic stimulation for erethism, it is not possible to define criteria for an optimal selection of candidates or specific diagnoses with a better or poorer prognosis. In our series, the patient who did not benefit from stimulation had specific clinical features, possibly related to his temporal lesion. Franzini et al. included in their series one case of posttraumatic damage of temporomesial structures, 5 cases of congenital or unknown origin and normal brain MRI, and one case of diffuse frontal cortex damage, secondary to heart arrest. All of the patients experienced a favorable outcome except for the patient with bifrontal brain damage secondary to cerebral hypoxia.13 It is likely that lesions in different structures within the limbic circuit might respond in different degrees to stimulation at the PMH.
Other beneficial effects were noticed, in addition to improvement in behavior. A more regular sleep pattern was generally observed, and one patient with epilepsy had a reduction in seizure frequency. The hypothalamus is a fundamental structure for generation of sleep-wakefulness rhythmicity. It is part of the “ascending reticular activating system,” which keeps the “thalamo-cerebral cortex unit” awake.40,41 Other structures involved in the maintenance of sleep-wakefulness cycles are located within the brainstem and basal forebrain and release neurotransmitters that activate the thalamus and cortex, producing the characteristic expressions of the awake state.41 A dysfunction in these circuits might be related to sleep disturbances, which could be appropriate for regulation by stimulation.
The therapeutic effect of hypothalamic stimulation on epileptic seizures has also been described by Franzini et al.12 They observed a 50% seizure reduction in one of their two erethic patients with comorbid epilepsy and a disappearance of the generalized seizures and persistence of complex seizures in the second patient. In a study published in 2005, van Rijckevorsel et al. described the electrophysiological features of the mammillary bodies and mammillothalamic tract and suggested that those nuclei might be potential targets for the treatment of refractory epilepsy.49 In another publication from that same year, Duprez et al.10 described a successful preliminary study to evaluate the efficacy and safety of DBS of the mammillary bodies of 3 patients with refractory epilepsy. Currently, there is an ongoing clinical trial to assess the role of DBS as a treatment for refractory epilepsy.
One patient (Case 6) demonstrated improvement in compulsive food and water intake with the stimulation. The lateral hypothalamus has long been implicated in feeding behavior and energy expenditure, and stereotactic lesioning of this area in obese humans resulted in significant, although transient, appetite suppression and slight weight reduction in 3 patients.7,17,18,27,30,31 Spreading currents from the stimulated area to the lateral hypothalamus might be responsible for the positive effect we observed in this patient. However, the other patients did not show significant variations in their weight, and the modest gain over the years may be explained by the control of hyperactivity. If DBS causes an overall modification in patterns of activity toward a more regular pattern, it may be possible that a preexisting circuit dysfunction is necessary for a modification in appetite and weight.19
This study was not controlled, so a placebo effect is possible. The persistence of benefit for 5 years in some patients argues against a placebo effect, however. Moreover, the intellectual level of these patients makes it difficult for them to retain the memory of these operations. We have to consider as well that medication regimens were not maintained over the follow-up period, and this constitutes another potential source of variability. Nevertheless, it seems unlikely that medication changes would account for the therapeutic effects observed, since exhaustive medical treatment had been attempted before the patients were referred for surgery. In any case, only a controlled trial under double-blind conditions and unchanged medication would adequately address this issue.
Finally, it is important to emphasize that all current approaches to neuromodulation in psychiatry must be framed in an ethical and regulatory context and that DBS treatment in mentally incompetent patients warrants extra ethical attention.23–25,35 While the special vulnerability of these patients is no reason to exclude them from beneficial treatment, it necessitates particularly careful observation of the highest ethical requirements, according to specific guidelines proposed by experts in the field.23–25,35 The following criteria are essential to ensure patient protection: involvement of an experienced multidisciplinary team, careful selection of severely afflicted patients with conditions that are refractory to other treatment, careful explanation of the purpose and risks of the procedures to parents and legal guardians, and approval from an institutional review board or a local ethics committee that oversees the observance of national and international regulations.
Erethism is a rare but devastating syndrome with a tremendous individual, familial, and social impact. In 5 of our 6 patients, PMH DBS alleviated symptoms of erethism. It also produced positive effects in other areas. The ideal criteria for patient selection, as well as optimal stimulation parameters, remain uncertain. Prospective controlled studies on PMH DBS, under blinded conditions with a larger number of patients, are necessary to confirm these results.
Conclusions
Five of our 6 patients with pathological aggressiveness had a reduction of their outbursts of violence after PMH DBS without significant adverse effects. Lowfrequency and high-frequency stimulation were found equally beneficial in controlling their dysfunctional behavior. Improvement in other domains of interest was observed in some patients, including regulation of sleep patterns, reduction of seizure frequency, and resolution of compulsive eating and drinking. This long-term study suggests that PMH DBS might have therapeutic effects for patients with intractable aggressiveness. A larger DBS study using a controlled design would be necessary to confirm the efficacy and safety of DBS for erethism and would possibly help define clinical predictors of responsiveness and optimal stimulation parameters.
Disclosure
This work was partially supported by a grant from the Plan Nacional de Investigación Científica, Desarrollo e Innovación Tecnológica, Instituto de Salud Carlos III, Subdirección General de Evaluación y Fomento de la Investigación PS09/02116.
Author contributions to the study and manuscript preparation include the following. Conception and design: Torres. Acquisition of data: Torres, Pedrosa. Analysis and interpretation of data: Torres, Ezquiaga. Drafting the article: Torres. Critically revising the article: all authors. Reviewed submitted version of manuscript: all authors. Approved the final version of the manuscript on behalf of all authors: Torres. Administrative/technical/material support: Torres, Pastor. Study supervision: Sola.
BIBLIOGRAFÍA
1. American Psychiatric Association: Diagnostic and Statisti cal Manual of Mental Disorders (DSM-IV). Washington, DC: American Psychiatric Publishing, 1994
2. Androulidakis AG, Mazzone P, Litvak V, Penny W, Dileone M, Gaynor LM, et al: Oscillatory activity in the pedunculopontine area of patients with Parkinson’s disease. Exp Neurol 211: 59 –66, 2008
3. Arjona VE: Sterotactic hypothalamotomy in erethic children. Acta Neurochir Suppl (Wien) 21: 185 –191, 1974
4. Ballanger B, Lozano AM, Moro E, van Eimeren T, Hamani C, Chen R, et al: Cerebral blood flow changes induced by pe dunculopontine nucleus stimulation in patients with advanced Parkinson’s disease: a [(15)O] H2O PET study. Hum Brain Mapp 30: 3901–3909, 2009
5. Bruininks RH, Gilman CJ: La observación y el currículum de las destrezas de conducta adaptativa: dos instrumentos de evaluación y ayuda en la intervención, in Bruininks RH (ed): Conducta Adaptativa y Discapacidad. Bilbao: ICE de la Universidad de Deusto-Mensajero, 1993
6. Bruininks RH, Hill B, Weatherman RF, Woodcock RW: ICAP: Inventory for Client and Agency Planning. Itasca, IL: Riverside Publishing, 1986
7. Carron R, Chabardes S, Hammond C: [Mechanisms of action of high-frequency deep brain stimulation. A review of the literature and current concepts.] Neurochirurgie 58: 209 –217, 2012 (Fr)
8. Costa A, Carlesimo GA, Caltagirone C, Mazzone P, Pierantozzi M, Stefani A, et al: Effects of deep brain stimulation of the peduncolopontine area on working memory tasks in patients with Parkinson’s disease. Parkinsonism Relat Disord 16: 64 –67, 2010
9. Duncan GE, Inada K, Farrington JS, Koller BH, Moy SS: Neural activation deficits in a mouse genetic model of NMDA receptor hypofunction in tests of social aggression and swim stress. Brain Res 1265: 186 –195, 2009
10. Duprez TP, Serieh BA, Raftopoulos C: Absence of memory dysfunction after bilateral mammillary body and mammillothalamic tract electrode implantation: preliminary experience in three patients. AJNR Am J Neuroradiol 26: 195 –198, 2005 (Letter)
11. Franzini A, Broggi G, Cordella R, Dones I, Messina G: Deep brain stimulation for aggressive and disruptive behavior. World Neurosurg [epub ahead of print], 2012
12. Franzini A, Marras C, Ferroli P, Bugiani O, Broggi G: Stimulation of the posterior hypothalamus for medically intractable impulsive and violent behavior. Stereotact Funct Neurosurg 83: 63 –66, 2005
13. Franzini A, Messina G, Cordella R, Marras C, Broggi G: Deep brain stimulation of the posteromedial hypothalamus: indications, long-term results, and neurophysiological considerations. Neurosurg Focus 29(2): E13, 2010
14. Gaul C, Diener HC, Müller OM: Cluster headache: clinical features and therapeutic options. Dtsch Arztebl Int 108: 543 – 549, 2011
15. Gellhorn E: Conditioning, sensations, and the ergotropic-trophotropic balance. Cond Reflex 3: 34 –44, 1968
16. Gellhorn E: The emotions and the ergotropic and trophotropic systems. Psychol Forsch 34: 48 –94, 1970
17. Halpern CH, Torres N, Hurtig HI, Wolf JA, Stephen J, Oh MY, et al: Expanding applications of deep brain stimulation: a potential therapeutic role in obesity and addiction management. Acta Neurochir (Wien) 153: 2293 –2306, 2011
18. Halpern CH, Wolf JA, Bale TL, Stunkard AJ, Danish SF, Grossman M, et al: Deep brain stimulation in the treatment of obesity. A review. J Neurosurg 109: 625 –634, 2008
19. Hernando V, Pastor J, Pedrosa M, Peña E, Sola RG: Lowfrequency bilateral hypothalamic stimulation for treatment of drug-resistant aggressiveness in a young man with mental retardation. Stereotact Funct Neurosurg 86: 219 –223, 2008 20. Karimi M, Golchin N, Tabbal SD, Hershey T, Videen TO, Wu J, et al: Subthalamic nucleus stimulation-induced regional blood flow responses correlate with improvement of motor signs in Parkinson disease. Brain 131: 2710 –2719, 2008
21. Kuhn J, Lenartz D, Mai JK, Huff W, Klosterkoetter J, Sturm V: Disappearance of self-aggressive behavior in a brain-injured patient after deep brain stimulation of the hypothalamus: technical case report. Neurosurgery 62: E1182, 2008
22. Lin D, Boyle MP, Dollar P, Lee H, Lein ES, Perona P, et al: Functional identification of an aggression locus in the mouse hypothalamus. Nature 470: 221–226, 2011
23. Lipsman N, Bernstein M, Lozano AM: Ethical conduct of psychiatric neurosurgery clinical trials. Neurosurg Focus 29(2): E9, 2010
24. Lipsman N, Giacobbe P, Bernstein M, Lozano AM: Informed consent for clinical trials of deep brain stimulation in psychiatric disease: challenges and implications for trial design. J Med Ethics 38: 107–111, 2012
25. Lozano AJ: A historical perspective of casuistry and its application to contemporary biomedical ethics. Linacre Q 70: 37–45, 2003
26. Lozano AM, Eltahawy H: How does DBS work? Suppl ClinNeurophysiol 57: 733 –736, 2004
27. Melega WP, Lacan G, Gorgulho AA, Behnke EJ, De Salles AA: Hypothalamic deep brain stimulation reduces weight gain in an obesity-animal model. PLoS ONE 7: e30672, 2012 28. Merrill DR, Bikson M, Jefferys JGR: Electrical stimulation of excitable tissue: design of efficacious and safe protocols. J Neurosci Methods 141: 171–198, 2005
29. Ramamurthi B: Stereotactic operation in behaviour disorders. Amygdalotomy and hypothalamotomy. Acta Neuro chir Suppl (Wien) 44: 152 –157, 1988
30. Rieu I, Derost P, Ulla M, Marques A, Debilly B, De Chazeron I, et al: Body weight gain and deep brain stimulation. J Neu rol Sci 310: 267–270, 2011
31. Sani S, Jobe K, Smith A, Kordower JH, Bakay RA: Deep brain stimulation for treatment of obesity in rats. J Neurosurg 107: 809 –813, 2007
32. Sano K, Mayanagi Y: Posteromedial hypothalamotomy in the treatment of violent, aggressive behaviour. Acta Neurochir Suppl (Wien) 44: 145 –151, 1988
33. Sano K, Mayanagi Y, Sekino H, Ogashiwa M, Ishijima B: Results of stimulation and destruction of the posterior hypothalamus in man. J Neurosurg 33: 689 –707, 1970
34. Saper CB: Animal behaviour: the nexus of sex and violence. Nature 470: 179 –181, 2011
35. Schermer M: Ethical issues in deep brain stimulation. Front Integr Neurosci 5: 17, 2011
36. Schvarcz JR, Driollet R, Rios E, Betti O: Stereotactic hypothalamotomy for behaviour disorders. J Neurol Neurosurg Psychiatry 35: 356 –359, 1972
37. Schweder PM, Joint C, Hansen PC, Green AL, Quaghebeur G, Aziz TZ: Chronic pedunculopontine nucleus stimulation restores functional connectivity. Neuroreport 21: 1065 –1068, 2010
38. Seijo F, Saiz A, Lozano B, Santamarta E, Alvarez-Vega M, Seijo E, et al: Neuromodulation of the posterolateral hypothalamus for the treatment of chronic refractory cluster headache: experience in five patients with a modified anatomical target. Cephalalgia 31: 1634 –1641, 2011
39 Sokolowski K, Corbin JG: Wired for behaviors: from development to function of innate limbic system circuitry. Front Mol Neurosci 5: 55, 2012
40. Szymusiak R: Hypothalamic versus neocortical control of sleep. Curr Opin Pulm Med 16: 530 –535, 2010
41. Szymusiak R, McGinty D: Hypothalamic regulation of sleep and arousal. Ann N Y Acad Sci 1129: 275 –286, 2008
42. Temel Y, Visser-Vandewalle V: Targets for deep brain stimulation in Parkinson’s disease. Expert Opin Ther Targets 10: 355 –362, 2006
43. Tokizane T, Kawakami M, Gellhorn E: On the relation between the activating and the recruiting systems. Arch Int Physiol Biochim 65: 415 –432, 1957
44. Tokizane T, Kawamura H, Imamura G: [Effect of hypothalamic stimulation on the activity of the paleocortex and archicortex.] Prensa Med Argent 51:283–285, 1964 (Spain)
45. Torres CV, Pastor J, Navarrete EG, Sola RG: [Deep brain stimulation for refractory epilepsy: extrathalamic targets.]. Rev Neurol 53:153–164, 2011 (Spain)
46. Torres N, Chabardès S, Benabid AL: Rationale for hypothalamus deep brain stimulation in food intake disorders and obesity. Adv Tech Stand Neurosurg 36:17–30, 2011
47. Toth M, Fuzesi T, Halasz J, Tulogdi A, Haller J: Neural inputs of the hypothalamic “aggression area” in the rat. Behav Brain
Res 215:7–20, 2010
48. Tulogdi A, Toth M, Halasz J, Mikics E, Fuzesi T, Haller J: Brain mechanisms involved in predatory aggression are activated in a laboratory model of violent intra-specific aggression. Eur J Neurosci 32:1744–1753, 2010
49. van Rijckevorsel K, Abu Serieh B, de Tourtchaninoff M, Raftopoulos C: Deep EEG recordings of the mammillary body in epilepsy patients. Epilepsia 46:781–785, 2005
Manuscript submitted August 29, 2012. Accepted April 15, 2013.
Accepted April 15, 2013.
Please include this information when citing this paper: published online June 7, 2013; DOI: 10.3171/2013.4.JNS121639.
Address correspondence to: Cristina V. Torres Díaz, M.D., Ph.D., Department of Neurosurgery, University Hospital La Princesa, C/ Diego de León 32, 28006 Madrid, Spain. email: cristinatorresdiaz@ yahoo.es.