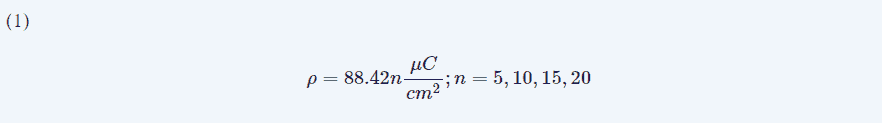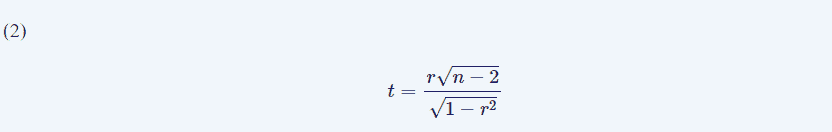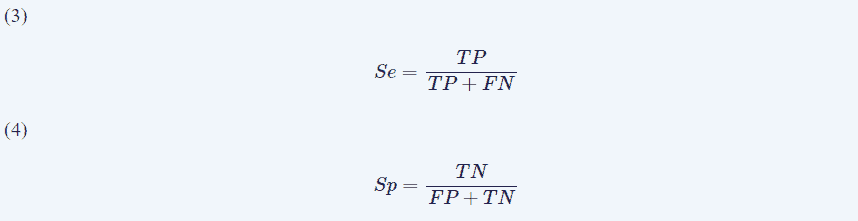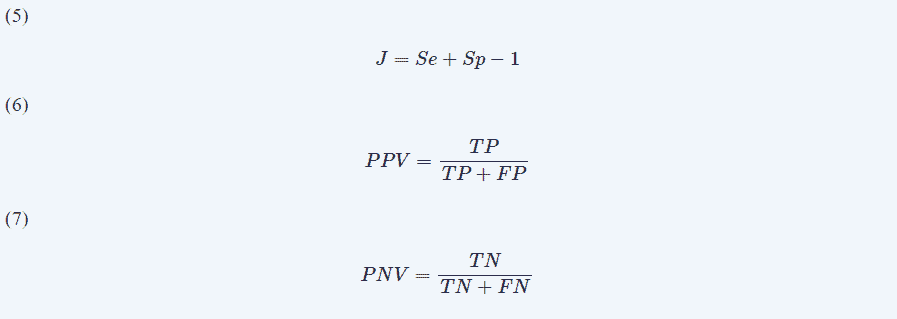Intraoperative Cortico-Cortical Evoked Potentials for Monitoring Language Function during Brain Tumor Resection in Anesthetized Patients
Técnica neurofisiológica fiable para mapear y monitorizar las regiones asociadas a la función del lenguaje
El Dr. Rafael García de Sola y su equipo analizan en este artículo como los potenciales evocados corticocorticales (CCEPs en inglés) han demostrado ser una técnica neurofisiológica fiable para mapear y monitorizar las regiones asociadas a la función del lenguaje en un pequeño grupo de pacientes anestesiados. La alta correlación entre los eventos de alerta y los resultados posquirúrgicos sugirió una alta sensibilidad y especificidad y los CCEPs pueden ser utilizados sistemáticamente en pacientes bajo anestesia general. No obstante, el pequeño número de pacientes estudiados sugiere considerar estos resultados con cautela.
Rafael García de Sola
Director de la Cátedra UAM “Innovación en Neurocirugía”
Jefe del Servicio de Neurocirugía
Hospital Ntra. Sra. del Rosario
Madrid
Lorena Vega-Zelaya 1,2, Paloma Pulido 2,3, Rafael G. Sola 4,5, Jesús Pastor 1,2,*
1 Clinical Neurophysiology, Hospital Universitario La Princesa, 28006 Madrid, Spain
2 Biomedical Research Institute, Hospital Universitario La Princesa, 28006 Madrid, Spain
3 Neurosurgery, Hospital Universitario La Princesa, 28006 Madrid, Spain
4 Neurosurgery, Clínica Nuestra Señora del Rosario, 28006 Madrid, Spain
5 Neurosurgery, Autonomous University of Madrid, 28006 Madrid, Spain
*Correspondence: jesus.pastor@salud.madrid.org (Jesús Pastor)
Academic Editor: Gernot Riedel
J. Integr. Neurosci. 2023, 22(1), 17; https://doi.org/10.31083/j.jin2201017
Submitted: 13 August 2022 | Revised: 29 September 2022 | Accepted: 9 October 2022 | Published: 13 January 2023
(This article belongs to the Special Issue Brain Stimulation and Neuroimaging)
Copyright: © 2023 The Author(s).
This is an open access article under the CC BY 4.0 license.
Abstract
Objective
Background: Cortico-cortical evoked potentials (CCEPs) have been used to map the frontal (FLA) and parietal (PLA) cortical regions related to language function. However, they have usually been employed as a complementary method during sleep-awake surgery. Methods: Five male and two female patients received surgery for tumors located near language areas. Six patients received general anesthesia and the sleep-awake method was used for patients with tumors located near the cortical language areas. We performed motor and somatosensory mapping with CCEPs to identify language areas and we monitored responses during surgery based on the mapping results. Electrocorticography was performed throughout the surgery. Single pulses of 1 ms duration at 5–20 mA were delivered by direct cortical stimulation using one grid at one region (e.g., FLA) and then recording using a second gird at another area (i.e., PLA). Next, reversed stimulation (from PLA to FLA) was performed. The charge density for electrical stimulation was computed. Sensibility, specificity, predictive positive values, and predicted negative values were also computed for warning alterations of CCEPs.
Results
Gross tumor resection was achieved in four cases. The first postsurgical day showed language alterations in three patients, but one year later six patients remained asymptomatic and one patient showed the same symptomatology as previously. Seizures were observed in two patients that were easily jugulated. CCEPs predicted warning events with high sensibility and specificity. Postsurgical language deficits were mostly transitory. Although the latency between frontal and parietal regions showed symmetry, the amplitude and the relationship between amplitude and latency were different for FLA than for PLA. The charge density elicited by CCEPs ranged from 442 to 1768 μC/cm 2.
Conclusions
Conclusions:
CCEPs have proven to be a reliable neurophysiological technique for mapping and monitoring the regions associated with language function in a small group of anesthetized patients. The high correlation between warning events and postsurgical outcomes suggested a high sensitivity and specificity and CCEPs can be used systematically in patients under general anesthesia. Nevertheless, the small number of studied patients suggests considering these results cautiously.
Keywords
: brain mapping, direct cortical stimulation, intraoperative neurophysiological monitoring, motor evoked potentials, somatosensory evoked potentials
Introduction
Cortico-cortical evoked potentials (CCEPs) are a promising technique for detecting functional connections in cortical networks, even under general anesthesia [1, 2]. This method uses single pulse electrical stimulation (SPES) applied directly to the cortex with subdural electrodes to record evoked waveforms from the remote cortex. This response has been shown to be robust and stable.
In an extraoperative setting, functional cortical networks were mapped including language networks between the frontal language area (FLA) and the temporal language area (PLA) [2, 3] and the motor-sensory system [4, 5], limbic system [6, 7], visual system [8], and seizure propagation networks [9, 10]. In addition, recent studies have demonstrated that intraoperative CCEPs are a feasible and reproducible tool for mapping language system during resection of brain tumors [2, 11, 12].
The main advantage of CCEPs is to gain access to functional connectivity in real time with good spatial resolution, unlike diffusion tensor imaging that depicts the connections between different areas, but cannot resolve the function nor direction of white matter tracts. In patients with structural lesions, the functional tract can be distorted or interrupted due to brain edema or infiltration of a tumor [13]. Also, functional magnetic resonance imaging (fMRI) provides dynamic information on cortical functions, but it does not indicate the actual dynamics of information flow.
In awake patients, the technique commonly used for functional mapping is direct cortical stimulation (DCS) with Ojemann’s stimulation or low-frequency stimulation, which consists of a 50–60 Hz train, 3–5 seconds in length, with a pulse-width as high as 400 μs , although a high frequency can be used as well [14, 15].
CCEPs were initially described as a neurophysiological monitoring technique for language areas in patients with brain tumors [2, 16]. In most cases, CCEPs were used during sleep-awake craniotomies. However, a good correlation between CCEPs and language function has been repeatedly observed [11] and confidence has developed for its use in patients that cannot be evaluated with an awake craniotomy.
In this report, we describe the utility of CCEPs for mapping and monitoring language associated areas in patients under general anesthesia during brain surgery performed for tumors located near language regions. Interestingly, one of the patients underwent surgery with the sleep-awake-sleep technique, which allowed us to correlate CCEPs and language functions. This patient was described in detail elsewhere [11].
Materials and Methods
Patients
We performed a prospective study on seven patients undergoing surgery for removal of an intraparenchymal brain lesion located within or close to language-related structures in the dominant cerebral hemisphere. The patients had a mean age of 53.4 ± 4.2 (range, 31–62) years. The clinical and demographic features are shown in Table 1. During the surgery, CCEPs were used to identify and monitor functional connectivity. The study observed the principles of the Helsinki’s Declaration and was approved by the Ethics Committee of the Hospital Universitario de la Princesa. Each patient provided wrote informed consent before surgery.
Patients were evaluated with neurological and neuropsychological assessments using the Boston Diagnostic Aphasia Examination (BDAE) [17] before surgery. Follow-up examinations were performed on the first postoperative day and the first and six months following surgery as well as one year after surgery.
All patients were right handed and the tumors were always located in the left hemisphere. Preoperatively, all patients had a history of epileptic seizures (ES). One presented with motor aphasia and another with dysarthria due to involvement of the motor language area.
Magnetic Resonance Imaging (MRI)
Preoperative and postoperative imaging included 1.5-T multimodal MRI (General Electric®, Fairfield, CT, USA), which included pre- and postcontrast T1-weighted, T2-weighted, FLAIR, diffusion-weighted, diffusion tensor, spectroscopic imaging, and tractography with a specific oncologic protocol.
Contrast MRI was performed after the surgery and compared with preoperative MRI results. The extent of tumor resection was defined as a gross total resection (GTR) if there was no residual enhancement in the postoperative MRI. Otherwise, the excision was classified as a subtotal resection (STR).
Intraoperative Neurophysiological Cortical Mapping and Monitoring
To identify eloquent areas, an intraoperative functional mapping was performed with multimodal neurophysiological monitoring equipment with 32 channels (Elite, Cadwell®, Kennewick, Washington, USA). The sampling rate was 10 kHz and the bandwidth was 1.5–1.000 Hz with the notch off. Electrocorticography (ECoG) was used to monitor the brain responses during electrical stimulation to identify the appearance of epileptiform patterns (post-discharges). Electrical stimulation was performed with DCS using a grid of 4 × 5 electrodes (Ad-Tech®, Racine, Wisconsin, USA) of 1.2 mm in diameter and 1 cm center-to-center by means of three monophasic single pulses that were 1 ms in length and separated by 1 s.
Before placing the grids onto the language-related areas, we identified the primary motor (PMC) and somatosensory (PSSC) cortices. After we identified the face/tongue primary motor region, we placed the grid onto what would be the Broca’s area or FLA. The grid that was placed onto supposedly the Wernicke’s area or PLA was located occipital to the primary somatosensory cortex. The pre- and postcentral gyri and the central sulcus (CS) were determined with a somatosensory evoked potentials (SSEPs) phase reversal technique [15]. Constant-current electrical stimulation was delivered at the right median nerve by trains of 300 pulses at 7.1 Hz, 200 μs in duration and 20 mA intensity. The band-pass filter was set at 10–1500 Hz, noch off. Recording through subdermal electrodes was performed at Erb’s point.
DCS for identifying the PMC was performed using paired grid electrodes. Stimulation was done using constant-current trains of 6 pulses at 500 Hz (high frequency technique) with bi-phasic pulses of 150–200 μs in duration/phase [15] and intensities between 4–30 mA. Motor-evoked potentials (MEPs) were recorded with a pair of subdermal electrodes (12/18 mm, SGM, Ljubiceva, Croatia) inserted into the muscles of the right side of the body. The filter bandwidth was 50–3.000 Hz, notch off, with a time base of 10 ms/division. It is extremely important to switch-off the notch filter because its presence induces an artifact similar to CCEPs and can mislead the neurophysiologist (Fig. 1). The appearance of post-discharge or ES was continuously monitored during all the processes.
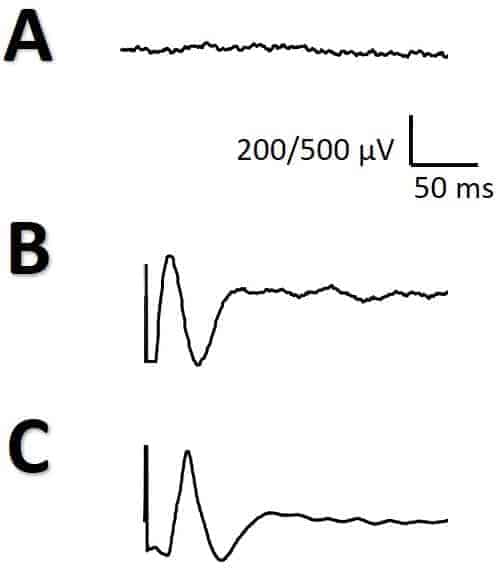
For CCEP recording, two grids of 4 × 5 electrodes were placed in the frontal and temporal language areas. The low-frequency filter was set at 10 Hz and the high frequency filter was set at 1500 Hz, notch off. DCS was applied used the SPES method on two adjacent electrodes (constant-current square pulse of 1 ms duration up to a maximum 20 mA). The stimulus intensity was increased steadily from 5 mA using stepwise increments of 5 mA until the effect was attained or post-discharges on ECoG were noted. A ground electrode was placed at the ipsilateral ear-lobe and a reference electrode was placed at the contralateral ear-lobe.
We started mapping by stimulating a pair of consecutive electrodes placed at the FLA that were in different orientations (parallel or orthogonal to the sulcus) and anterior to the PMC of the face and tongue, according to the results of motor mapping. To evaluate the dorsal language pathway, at least three trials were averaged separately to confirm the reproducibility of the responses on the grid located in the PLA. Recording was performed in a pseudomonopolar way, with all the grid electrodes referred to the contralateral ear-lobe. A positive response was recorded if a large N1 peak (upward directed) in the electrodes of the PLA was obtained [3, 18]. Next we used the electrodes of the PLA grid with positive responses to stimulate and record the grid located at the FLA.
Intraoperative neurophysiological monitoring (IONM) of language areas was performed throughout the surgery using the electrodes for which the higher bidirectional responses were recorded. Also, motor or somatosensory functions were monitored when the tumor was close to a primary cortices or inner capsule.
Charge density (ρ, in μC/cm2) was computed according to this expression (Eqn. 1):
We offer the deduction of this formula in Appendix A.
Surgery
The patients with suspected malignant tumors receive preoperatory 20 mg/kg 5-ALA (Gliolan®). All patients were operated on under general anesthesia, either because awake surgery was contraindicated due to deficit in language tests or because the patient would not tolerate the awake procedure. The anesthesia was induced with a bolus of propofol and remifentanil. They were subsequently maintained with propofol (6.1 ± 0.9 mg/kg/h; range, 4–11) and remifentanil (0.07 ± 0.01 μg/kg/min; range 0.02–0.11). In one patient, after the functional mapping was complete, resection was performed while the patient was awake. During the entire procedure under a wakeful condition, both language function (counting, fluency, and comprehension) and CCEP were constantly monitored [11].
The craniotomy and surgery were guided with a neuronavigational system (Brainlab®, Feldkirchen, Germany). The tumor was removed using microsurgical techniques and ultrasonic aspiration guided by 5-ALA fluorescence and neurophysiology controls.
The warning criterion of CCEP was set at a reduction in amplitude greater than 20% [16]. When it appeared, the surgeon changed the resection area until a complete recovery was observed. When the changes were persistent or repeated in the same region, then the surgery was temporarily stopped and the area was irrigated with warm saline. If the recovery maneuvers did not work, the resection was stopped.
Statistics
To compare latencies and amplitudes for N1 CCEP picked up at the FLA and PLA, we averaged all the potentials obtained in one grid (e.g., FLA) after the stimulation at the contralateral grid (PLA). Then we used a paired Student’s t test to evaluate significance. The null hypothesis was H0: latency/amplitude_FLA →PLA = latency/amplitude PLA→FLA, and the alternative hypothesis was HA: latency/mplitude_FLA→PLA ≠ latency/amplitude PLA→FLA.
Pearson’s correlation coefficient was used to determine the linear dependence between variables. Linear regression was evaluated by means of the least-squared method and its significance was evaluated by means of a contrast hypothesis against the null hypothesis (H0: r = 0 and alternative hypothesis HA
: r≠ 0), using the formula (Eqn. 2):
This describes a t-Student distribution with n-2 degrees of freedom [19]. The significance level was set at p = 0.05, and the results are shown as the mean ± standard error of the mean.
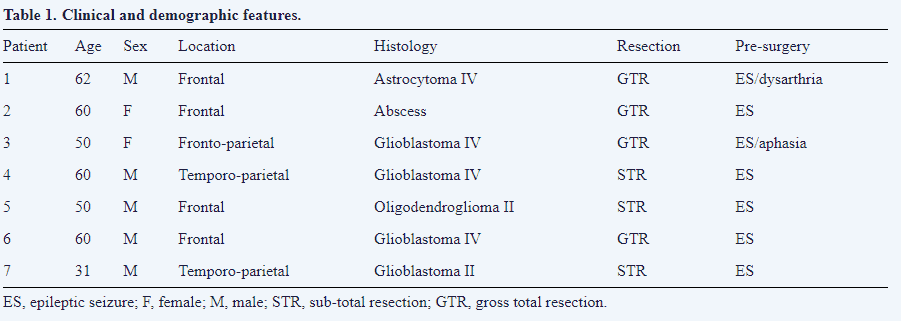
Although the number of patients was quite low, we could obtain the specificity (Sp) and sensibility (Se) by building a confusion matrix. It is important to build up these matrices for early outcomes (1 day) and longer period outcomes (1 year). For the first day, we used the following criteria: presence or absence of symptomatology and CCEP alteration or not. Therefore, we had these possible variables:
- • True positive (TP): CCEP alteration + symptomatic
• False negative (FN): no CCEP alteration + symptomatic
• True negative (TN): no CCEP alteration + asymptomatic
• False positive (FP): CCEP alteration + asymptomatic
where CCEP alteration can be reversible or not, because the relevance was the capacity to warn for possible lesions.
For the first year, we only considered when CCEP alterations were definite because we wanted to know the capacity for long term outcome predictions. We used the variables to obtain (Eqns. 3,4):
We used Youden’s index (J) J=Se+Sp-1 (Eqn. 5) to summarize the performance of CCEP monitoring, and the positive predictive value (PPV) and negative predictive value (NPV), which foresees the possibility of neurologic injury after CCEP alteration during IONM. To compute these values, we used the following formulas (Eqns. 6,7):
Results
One tumor was located in the fronto-parietal lobe (Fig. 2A), four tumors were located in the frontal lobe (Fig. 2B,E,F,G), and two were in the parieto-temporal (Fig. 2C,D) lobes.
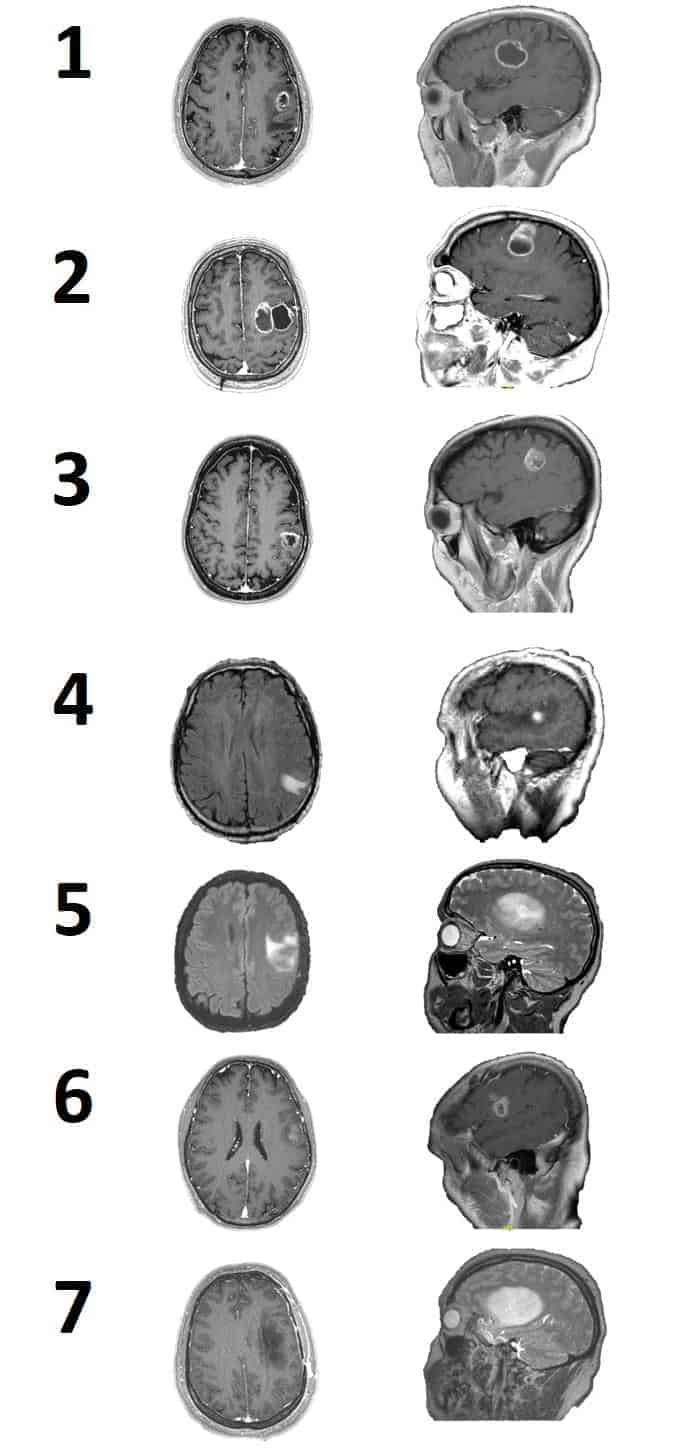
The patients with glioblastoma multiforme GBM were subsequently treated with radiotherapy and chemotherapy. Five of seven patients were asymptomatic one year after surgery (therefore, ES disappeared, although patients remained under pharmacological treatment), one persisted with ES, and one persisted with the same mild dysarthria previous to surgery but without ES. It is important to observe that none of the patients had aggravated symptomatology due to iatrogenic injury.
GTR was obtained in four of seven cases, meanwhile, in the remaining three patients, only STR was possible. In one case, the resection was stopped due to the presence of steady alterations of CCEPs; meanwhile, in the other two cases, the features of tumors (bleeding and malignity), out of IONM warnings, indicated the end of surgery. At one year of follow-up, no recurrence or tumor growth was observed (Table 2).
We estimated a charge density between 442 and 1768 μC/cm2. As stated above, ES were well controlled after surgery in five of seven patients and ES persisted in only one patient. Nevertheless, in this patient (#5), STR was achieved.
Cortical Mapping
Intraoperative mapping for peri-rolandic areas was carried out as stated above, identifying initially the CS, the PMC of the face, and the hand in all cases, and PSSC for the arm and hand (Fig. 3A). Then, the grids were moved and placed at putative FLA and PLA positions for monitoring identification of CCEPs. After extensive mapping, we selected the most robust and prominent potentials for monitoring during the surgery.
No post-discharges were observed in this group of patients. However, two focal (not propagated) ES were recorded and were jugulated by cold saline irrigation (Fig. 3B).
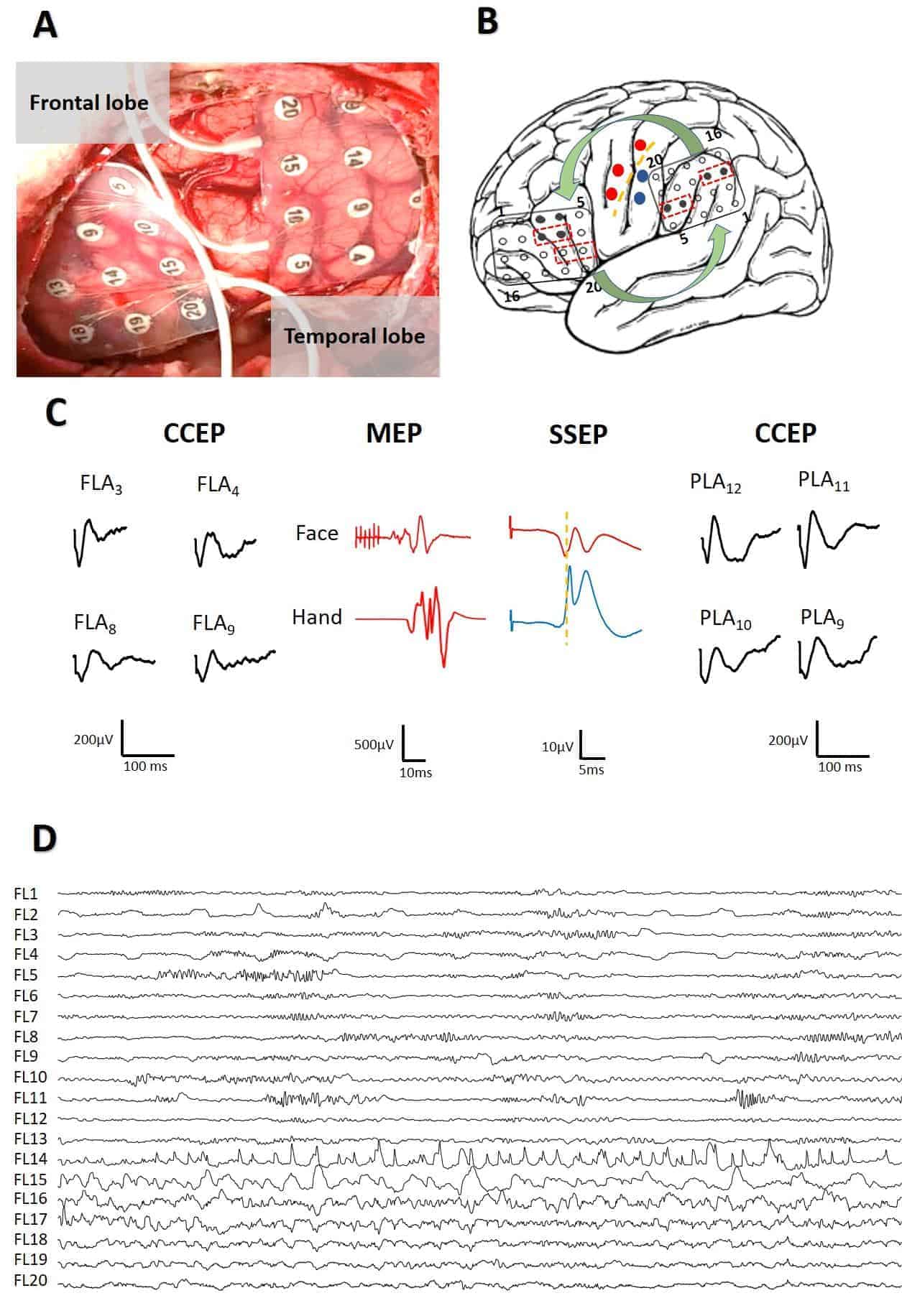
In all patients we obtained CCEPs, both from the FLA and the PLA, with a clear bidirectional response (Fig. 3B). On average, responses were obtained when stimulating 2.3 ± 0.6 (range 1–5) pairs of electrodes. The CCEPs were obtained with at least two electrodes. The average number of electrodes where responses were found in the FLA was 5.3 ± 1 and, in the PLA, 4.2 ± 1 responses were found (n.s., paired Student’s t test). In one patient, responses were obtained with 12 FLA electrodes. The different waveforms of CCEP found in the seven patients are illustrated in Fig. 4.
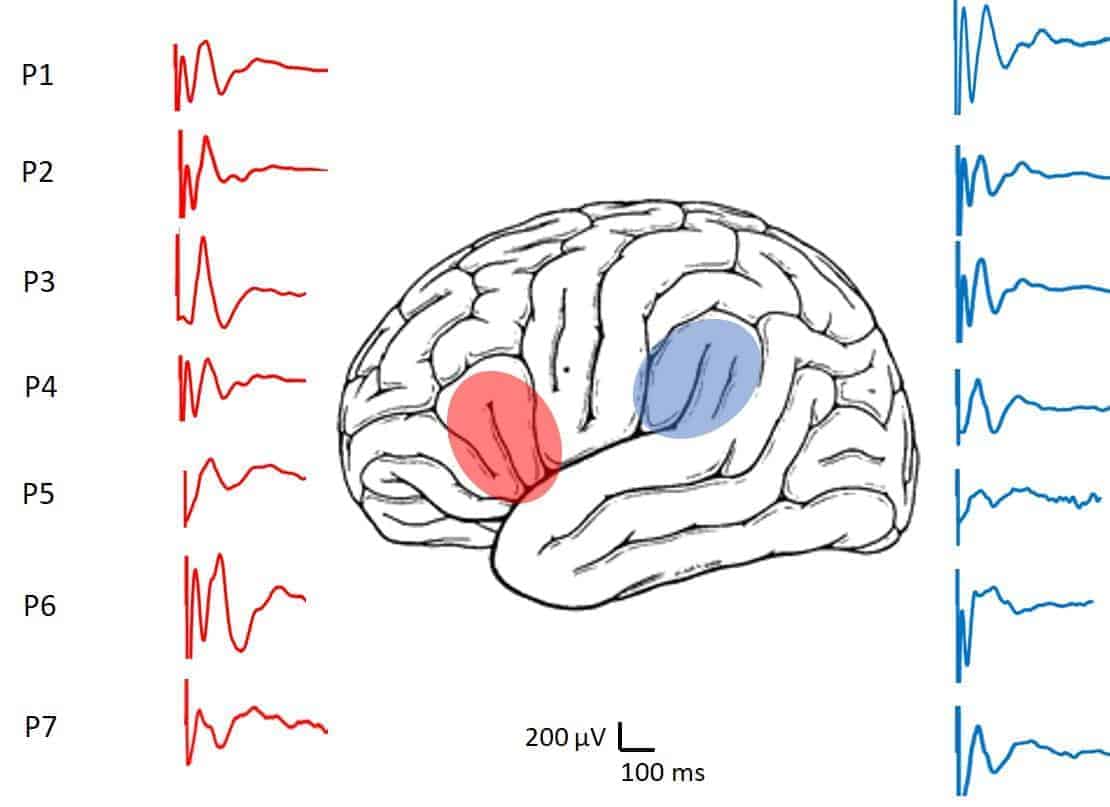
In the FLA we found responses with an average latency for N1 of 39.0 ± 0.7 ms (range 32.1–48.9 ms) and an amplitude of 204.0 ± 15.4 μV (range 48.0–400.0 μV). For the PLA, the average latency was 37.7 ± 0.8 ms (range 24.8–46.4 ms) and the amplitude was 184.1 ± 17.0 μV (range 30.6–540.2μV). Comparison between latencies in the FLA and PLA values were not significant (p = 0.274, paired Student’s t test) but they were significantly different for amplitudes (p = 0.017, paired Student’s t test).
We addressed the relationship between latencies and amplitudes for CCEPs at the FLA and PLA. Plots of these results are shown in Fig. 5.
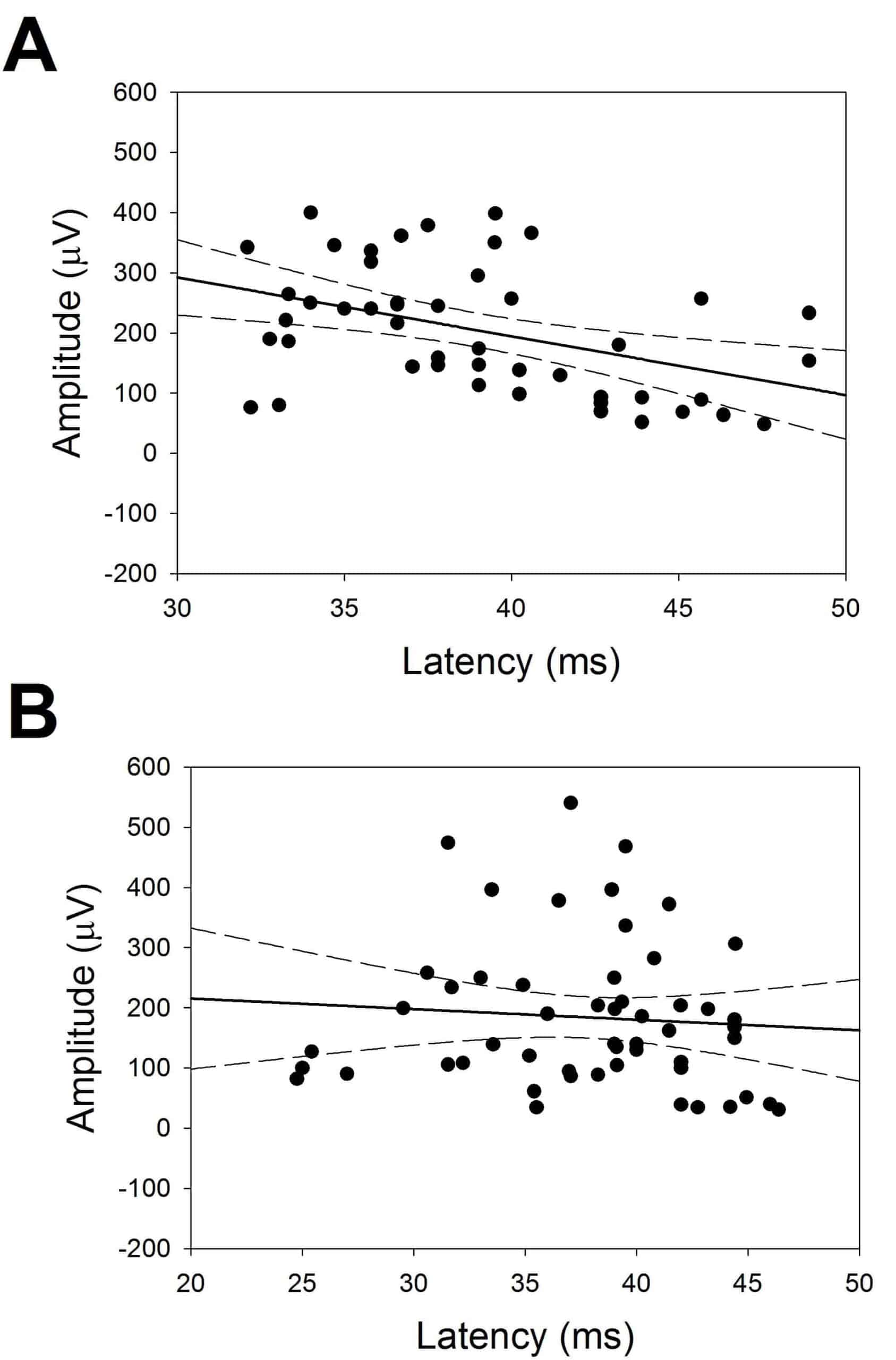
In Fig. 5, we can observe that, for FLA there was a significant linear relationship between latency and amplitude (Amp (µV ) = −9.8 × lat(ms) + 585.2, p < 0.001, Student’s t test). Nonetheless, no relationship was observed in the case of PLA (Amp (µV ) = −1.8 × lat(ms) + 250.6, p = 0.75, Student’s t test).
Intraoperative CCEP Monitoring
CCEPs were monitored in all cases during surgery, even in the patient that was awake [11]. In two cases, monitoring of the motor area of the face and tongue was also carried out through the electrodes of the FLA grid.
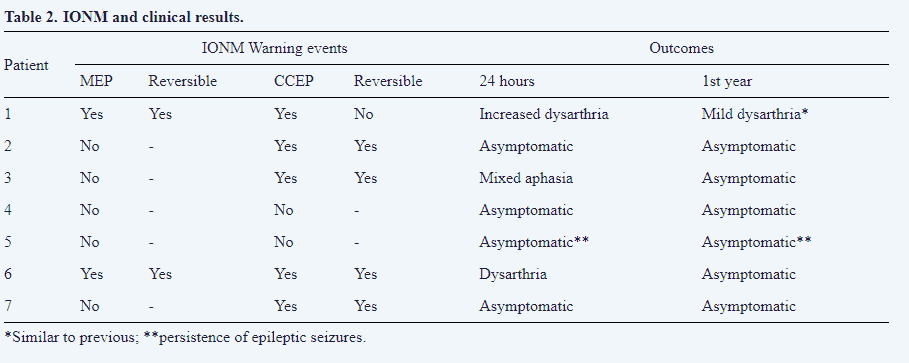
We had alarm criteria for five of the seven patients with a transitory alteration of CCEPs (Fig. 6A). Reversible alteration of tongue MEPs was also observed in one patient. In only one patient a definitely alteration in CCEPs was observed (Fig. 6B). This patient was the only one in which symptomatology persisted one year afther the surgery. In another patient, involvement of the CCEPs and MEPs of the face and mouth were also observed, occurring at different times during surgery, and both changes (Fig. 6C) were reversible. The patient presented with transient dysarthria in the early postoperative period.
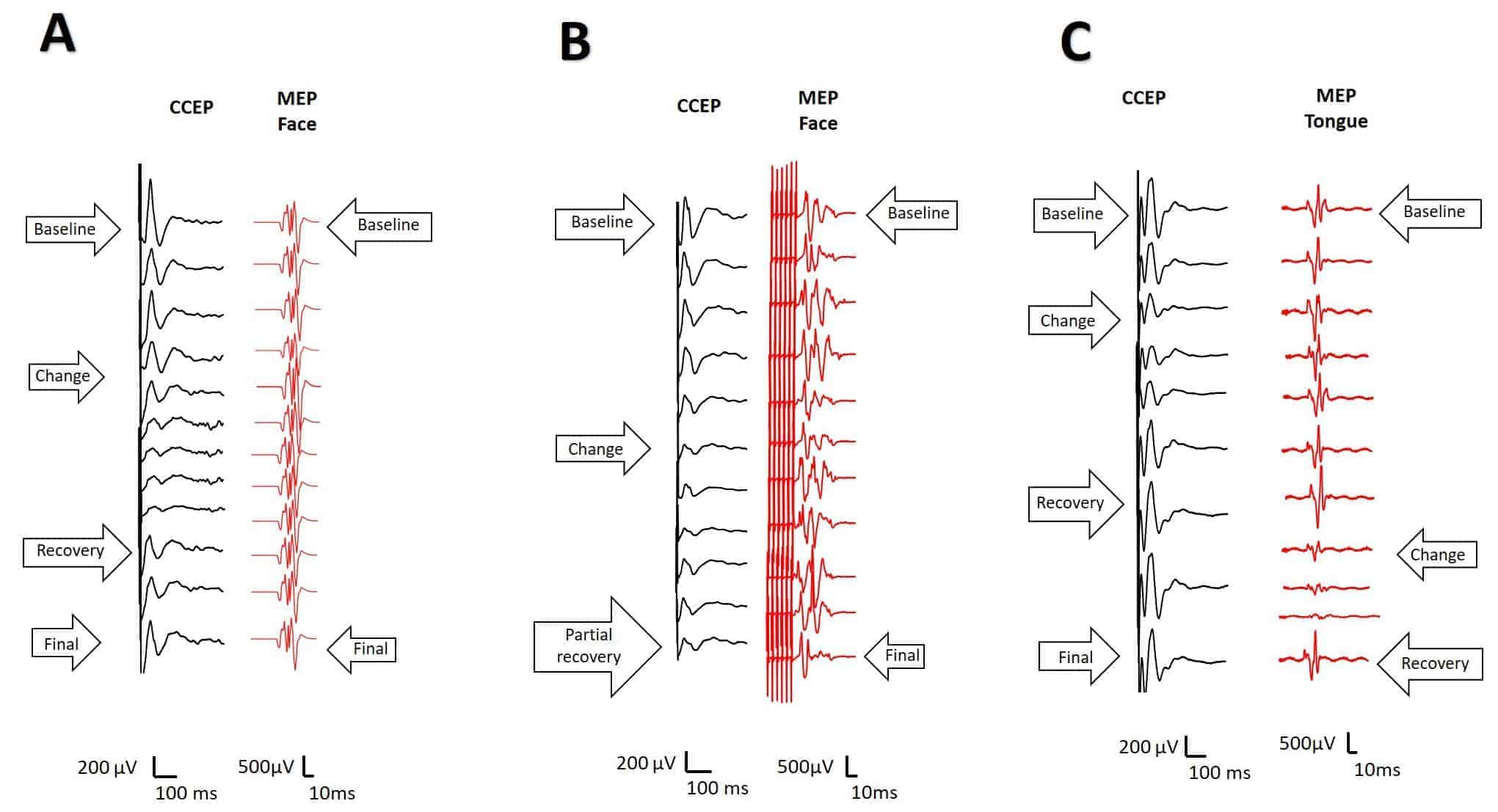
The remaining patients did not develop new neurological deficits in the postoperative period (Table 2). At the one year follow-up visit, five patients were asymptomatic, one of them still had mild dysarthria, and another still had ES. Symptomatology was similar for one, six, and twelve months after surgery. Although the number of patients in this study was quite limited, we have computed several variables regarding Se and Sp for the early and late post-surgery periods. These results are shown in Table 3.
The Se and Sp were high enough to assure confidence, as indicated J. More relevant, the PPV and NPV were also high, which means that the presence of CCEP alterations during IONM predicts the existence of postsurgical language deficits with high probability. On the contrary, the absence of warning events during IONM are associated with a high probability of a language function that is similar to function previous to surgery.

Discussion
In this study, we showed that CCEPs can reliably identify cortical regions related to language function. CCEPs can also be used to monitor patients under general anesthesia because they are easy to obtain and reliable for predicting postsurgical outcomes of language function.
Intraoperative CCEP monitoring for language function is usually performed with the patient awake as a complementary technique. Therefore, this is one of the first works addressing its usefulness in an group patients under general anesthesia with most of them not awake. Giampiccolo et al. [20], recently described the usefulness of CCEP monitoring in patients under general anesthesia with results very similar to those obtained in this study.
Obviously, an unconsciouss patient has a great risk to suffer iatrogenic injuries, especially these patients in which the surgery is performed in extremely complex regions near the FLA, PLA, PMC, PSSC and subcortical structures such as the inner capsulae, thalamo-cortical radiations, or arquate fascicle. Further, most of these patients (all of them in our study) suffered from ES. Therefore, it is of great importance to monitor all of these structures and functions with a comprehensive neurophysiological approach. Consequently, MEP, SSEP, and maybe others, as cortical evoked potentials are mandatory. By the same reasoning, ECoG can precociously prevent or identify the presence of ES [21].
Understanding the neural connectivity between cortical eloquent areas and white matter pathways (e.g., cortical and association fibers related to language or motor functions) is extremely important to preserve brain function when surgically treating brain lesions. Some of them have been extensively identified in conscious patients [22]. Further, in the last two decades, it has been suggested that CCEP connectivity can be recorded between the FLA and PLA, such as in awake anesthetized patients [16, 18].
The reliability of this technique for identifying language areas had been previously described [11, 16, 18, 23, 24]. In our study, we performed mapping and monitoring of the language areas in anesthetized patients with excellent clinical results. The PPV and NPV were high enough to confirm the intense correlation between the presence of CCEP alteration and the appearance of new language deficits. On the contrary, the absence of CCEP warning was highly correlated with the absence of new language deficits. Nevertheless, as we recognized above, the number of patients was small and we must be cautious in the acceptance of these values. This high correlation between changes in CCEPs and post-op functions was demonstrated in the patient operated on with the sleep-awake technique [11] in which transitory changes in CCEPs were strictly associated with transitory language deficits.
Although the mechanism underlying CCEPs remains unclear, an accepted theory is that one of the transmission mechanisms could be cortico-cortical propagation directly conveyed through white matter fibers [25]. We have observed some anisotropy in the responses of FLA and PLA areas. Although no differences in latencies have been observed, it was suggested that long neural connections share some symmetry from the FLA to PLA, and in the reversal direction, amplitudes were higher for CCEPs obtained from the PLA compared to the FLA. Potentials are the result of extracellular currents from the underlying cortex. Therefore, the cortical structure, including the number and types of neurons and synapses, as well as its synchronization would be responsible for the amplitude and dynamics of the potentials. Therefore, it is conceivable that cortical regions with different cyto-architecture (Brodmann’s area 40 for Broca’s area and for Wernicke’s area) give rise to different potentials. Nevertheless, much work is needed to understand the sources of these potentials.
CCEPs have proven to be an effective technique for the identification and monitoring of language areas, so their use in anesthetized patients should not be limited to those who cannot tolerate awake surgery. An important matter to note is that, in awake surgery, 4–23% of postoperative deterioration of speech function has been described, despite negative intraoperative cortical mapping [26, 27]. Besides the presence of language deficits, the limitations of awake surgery must be considered seriously [21]. It is a stressful situation for the patient. Hence, patients must have both adequate cognitive function and emotional maturity. In fact, the Japan Society for Awake Surgery Guidelines limits the target patient population to those ranging from 15–65 years of age, although with some limitations, awake craniotomy can be used in the pediatric population [28]. Nevertheless, use in mentally handicapped patients remains problematic or impossible. In addition, although it is currently accepted that the intracranial pain-sensitive structures are limited to the dura mater and its feeding areas, and pain can be adequately controlled by topical anesthesia of the skin, bone, and dura, it has been observed recently that the pia and small cerebral vessels are also pain sensitive, inducing sharp, intense, and briefly painful events [29].
CCEP monitoring in sleeping patients could be an adequate alternative, since it would also have four added advantages over awake surgery, (i) it is better tolerated, (ii) it is not subject to subjectivity, such as the intraoperative evaluation of verbal responses, (iii) it can be performed in patients with preoperative language deficits, in whom an intraoperative neurological evaluation cannot be carried out, and (iv) can be monitored constantly, not only at some time points before or between different stages of tumor removal.
The disadvantages of this technique are the necessity of an extensive craniotomy to allow adequate placement of the grid and also the increase in surgical times. It could be argued as well that the charge density during SPES to obtain CCEPs is high and could be harmful because it is several times higher than 30 μC/cm2. However, this limit is currently debated because it was defined for chronic stimulation. On the other hand, it is well recognized that macroscopic electrodes at the cortical surface behave differently than microscopic intracerebral electrodes and therefore the theoretical limit needs to be better defined [30]. On the other hand, it has been estimated that a charge density of the same magnitude (1269 μC/cm2) for the paradigm used during awake craniotomy [20] is not safer than in anesthetized patients. Nevertheless, the possibility of cortical electrical injury must be considered and, consequently, the patients should be followed after surgery (e.g., by means of periodic EEG examination).
Finally, we would like to pose a technical question not previously mentioned in the literature. The use of a notch filter gives rise to spurious waveforms quite similar to the true CCEPs. This can be misleading to the neurophysiologist, considering the presence of a positive response when in fact there is not. This bias can have dramatic results if there is erroneous identification of the functional areas. Therefore, it is extremely important to be conscious of this phenomenon.
Perhaps the most surprising fact used to justify awake surgery is that no differences were observed in the immediate postoperative motor status extent of resection between IONM in anesthetized patients and stimulation during awake craniotomy [25, 26, 31], although no detailed evaluation has been performed for the different techniques or surgeries. If there is no clear difference in the postsurgical outcomes, it is difficult to understand why it is necessary to stress the patient and the medical team when, instead, a systematic, calm and reliable mapping and monitoring of most brain functions, including language, can be done.
We hope that in the near future the CCEP technique will be widespread and equally accepted as motor or somatosensory evoked potentials for the assessment of motor and somatosensory pathways.
Conclusions
CCEPs have proven to be a reliable neurophysiological technique for mapping and monitoring the regions associated with language function in a small group of anesthetized patients. The high correlation between warning events and postsurgical outcomes suggests high sensitivity and specificity. Nevertheless, the small number of patients studied to date suggests these results should be considered cautiously.
Abbreviations
CCEP, cortico-cortical evoked potentials; DCS, direct cortical stimulation; DTI, diffusion tensor imaging; ECoG, electrocorticography; ES, epileptic seizure; FLA, frontal language area; FN, false negative; FP, false positive; GTR, gross total resection; fMRI, functional magnetic resonance imaging; IONM, intraoperative neurophysiological monitoring; MRI, magnetic resonance imaging; PLA, parietal language area; PMC, primary motor cortex; PNV, predictive negative value; PPV, predictive positive value; Se, sensibility; Sp, specificity; SPES, single pulse electrical stimulation; STR, subtotal resection; TN, true negative; TP, true positive.
Author Contributions
These should be presented as follows: JP and RGS designed the research study. JP and LVZ performed the research. PP and RGS provided help and advice on surgery and data recording. JP and LVZ analyzed the data. Initials JP and LVZ wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Ethics Approval and Consent to Participate
The study observed the principles of the Helsinki’s Declaration and was approved by the Ethics Committee of the Hospital Universitario de la Princesa (Ref. 3109), and each patient provided wrote informed consent before surgery.
Acknowledgment
Not applicable.
Funding
This work was financed by a grant from the Ministerio de Sanidad FIS PI17/02193 and was partially supported by FEDER (Fonds Europeen de Developpement Economique et Regional).
Conflict of Interest
The authors declare no conflict of interest. JP is serving as one of the editorial board members of this journal and the guest editor of the special issue on “Brain Stimulation and Neuroimaging”. We declare that JP had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to GR.
Appendix
Computation of charge density (ρ)
References
[1] Kunieda T, Yamao Y, Kikuchi T, Matsumoto R. New Approach for Exploring Cerebral Functional Connectivity: Review of Cortico-cortical Evoked Potential. Neurologia Medico-Chirurgica (Tokyo). 2015; 55: 374–382.
[2] Yamao Y, Suzuki K, Kunieda T, Matsumoto R, Arakawa Y, Nakae T, et al. Clinical impact of intraoperative CCEP monitoring in evaluating the dorsal language white matter pathway. Human Brain Mapping. 2017; 38: 1977–1991.
[3] Matsumoto R, Nair DR, LaPresto E, Najm I, Bingaman W, Shibasaki H, et al. Functional connectivity in the human language system: a cortico-cortical evoked potential study. Brain. 2004; 127: 2316–2330.
[4] Matsumoto R, Nair DR, Ikeda A, Fumuro T, LaPresto E, Mikuni N, et al. Parieto-frontal network in humans studied by cortico-cortical evoked potential. Human Brain Mapping. 2012; 33: 2856–2872.
[5] Swann NC, Cai W, Conner CR, Pieters TA, Claffey MP, George JS, et al. Roles for the pre-supplementary motor area and the right inferior frontal gyrus in stopping action: Electrophysiological responses and functional and structural connectivity. NeuroImage. 2012; 59: 2860–2870.
[6] Enatsu R, Gonzalez-Martinez J, Bulacio J, Kubota Y, Mosher J, Burgess RC, et al. Connections of the limbic network: a corticocortical evoked potentials study. Cortex. 2015; 62: 20–33.
[7] Kubota Y, Enatsu R, Gonzalez-Martinez J, Bulacio J, Mosher J, Burgess RC, et al. In vivo human hippocampal cingulate connectivity: a corticocortical evoked potentials (CCEPs) study. Clinical Neurophysiology. 2013; 124: 1547–1556.
[8] Matsuzaki N, Juhász C, Asano E. Cortico-cortical evoked potentials and stimulation-elicited gamma activity preferentially propagate from lower- to higher-order visual areas. Clinical Neurophysiology. 2013; 124: 1290–1296.
[9] Valentin A. Responses to single pulse electrical stimulation identify epileptogenesis in the human brain in vivo. Brain. 2002; 125: 1709–1718.
[10] Lacruz ME, García Seoane JJ, Valentin A, Selway R, Alarcón G. Frontal and temporal functional connections of the living human brain. European Journal of Neuroscience. 2007; 26: 1357–1370.
[11] Vega-Zelaya L, Navas M, Martín P, Pastor J. Utility of Intraoperative Cortico-Cortical Evoked Potentials for the Evaluation of Language Function during Brain Tumor Resection. Surgical Case Reports. 2020; 3: 1–4.
[12] Matsumoto R, Imamura H, Inouchi M, Nakagawa T, Yokoyama Y,Matsuhashi M, et al. Left anterior temporal cortex actively engages in speech perception: A direct cortical stimulation study. Neuropsychologia. 2011; 49: 1350–1354.
[13] Bizzi A, Nava S, Ferrè F, Castelli G, Aquino D, Ciaraffa F, et al. Aphasia induced by gliomas growing in the ventrolateral frontal region: Assessment with diffusion MR tractography, functional MR imaging and neuropsychology. Cortex. 2012; 48: 255–272.
[14] De Witt Hamer PC, Robles SG, Zwinderman AH, Duffau H, Berger MS. Impact of Intraoperative Stimulation Brain Mapping on Glioma Surgery Outcome: a Meta-Analysis. Journal of Clinical Oncology. 2012; 30: 2559–2565.
[15] Vega-Zelaya L, Pastor J. Intraoperative Neurophysiological Monitoring Techniques for the Resection of Malignant Brain Tumors Located in Eloquent Cortical Areas. Austin Journal of Neurosurgery. 2015; 2: 1038.
[16] Saito T, Tamura M, Muragaki Y, Maruyama T, Kubota Y, Fukuchi S, et al. Intraoperative cortico-cortical evoked potentials for the evaluation of language function during brain tumor resection: initial experience with 13 cases. Journal of Neurosurgery. 2014; 121: 827–838.
[17] Goodglass H, Kaplan E, Barresi B. The assessment of aphasia and related disorders. Lippincott Williams & Wilkins: Philadelphia, USA. 2001
[18] Yamao Y, Matsumoto R, Kunieda T, Arakawa Y, Kobayashi K, Usami K, et al. Intraoperative dorsal language network mapping by using single-pulse electrical stimulation. Human Brain Mapping. 2014; 35: 4345–4361.
[19] Spiegel MR, Schiller J, Srinivasan RA. Probabilidad y Estadística. McGraw-Hill: Bogotá. 2003.
[20] Giampiccolo D, Parmigiani S, Basaldella F, Russo S, Pigorini A, Rosanova M, et al. Recording cortico-cortical evoked potentials of the human arcuate fasciculus under general anaesthesia. Clinical Neurophysiology. 2021; 132: 1966–1973.
[21] Vega-Zelaya L, Sola RG, Pulido P, Pastor J. Do we need to awake patients up during cortical surgery? Journal of Cancer Research Updates. 2018, 7: 84–96.
[22] Duffau H, Gatignol P, Mandonnet E, Peruzzi P, Tzourio-Mazoyer N, Capelle L. New insights into the anatomo-functional connectivity of the semantic system: a study using cortico-subcortical electrostimulations. Brain. 2005; 128: 797–810.
[23] Yoshimoto T, Maruichi K, Itoh Y, Takamiya S, Kaneko T. Monitoring Corticocortical Evoked Potentials during Intracranial Vascular Surgery. World Neurosurgery. 2019; 122: e947–e954.
[24] Nakae T, Matsumoto R, Kunieda T, Arakawa Y, Kobayashi K, Shimotake A, et al. Connectivity Gradient in the Human Left Inferior Frontal Gyrus: Intraoperative Cortico-Cortical Evoked Potential Study. Cerebral Cortex. 2020; 30: 4633–4650.
[25] Yamao Y, Matsumoto R, Kikuchi T, Yoshida K, Kunieda T, Miyamoto S. Intraoperative Brain Mapping by Cortico-Cortical Evoked Potential. Frontiers in Human Neuroscience. 2021; 15: 635453.
[26] Kim SS, McCutcheon IE, Suki D, Weinberg JS, Sawaya R, Lang FF, et al. AWAKE CRANIOTOMY for BRAIN TUMORS near ELOQUENT CORTEX. Neurosurgery. 2009; 64: 836–846.
[27] Sanai N, Mirzadeh Z, Berger MS. Functional Outcome after Language Mapping for Glioma Resection. New England Journal of Medicine. 2008; 358: 18–27.
[28] Delion M, Terminassian A, Lehousse T, Aubin G, Malka J, N’Guyen S, et al. Specificities of Awake Craniotomy and Brain Mapping in Children for Resection of Supratentorial Tumors in the Language Area. World Neurosurgery. 2015; 84: 1645–1652.
[29] Fontaine D, Almairac F, Santucci S, Fernandez C, Dallel R, Pallud J, et al. Dural and pial pain-sensitive structures in humans: new inputs from awake craniotomies. Brain. 2018; 141: 1040–1048.
[30] Cogan SF, Ludwig KA, Welle CG, Takmakov P. Tissue damage thresholds during therapeutic electrical stimulation. Journal of Neural Engineering. 2016; 13: 021001.
[31] Nguyen HS, Sundaram SV, Mosier KM, Cohen-Gadol AA. A method to map the visual cortex during an awake craniotomy. Journal of Neurosurgery. 2011; 114: 922–926.
Related
Cirugía de la epilepsia temporal: Contribución de los estudios preoperatorios
Jesús Pastor, Guillermo J. Ortega, Iván Herrera-Peco, Marta Navas-García, Eduardo G. Navarrete, Concepción Alonso-Cerezo, Paloma Pulido-Rivas, García de Sola
Tema 10: Neurocirugía Funcional. Parte IV – Cirugía de la Epilepsia
Dr. García de Sola, Dra. Paloma Pulido-Rivas
Neural activation patterns and connectivity in visual attention during number and non-number processing: An ERP study using Ishihara pseudoisochromatic plates
Faraj Al-Marri et al., Journal of Integrative Neuroscience, 2018