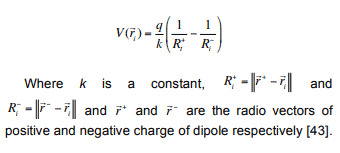Do we need to wake patients up during Cortical Surgery?
¿Se necesita mantener despiertos a los pacientes durante una cirugía cortical?
El Dr. Rafael García de Sola y su equipo analizan en este artículo como los potenciales evocados corticocorticales (CCEPs en inglés) han demostrado ser una técnica neurofisiológica fiable para mapear y monitorizar las regiones asociadas a la función del lenguaje en un pequeño grupo de pacientes anestesiados. En este artículo se analiza la posibilidad de no tener que tener al paciente despierto durante la cirugía de zonas que impliquen el lenguaje.
Rafael García de Sola
Director de la Cátedra UAM “Innovación en Neurocirugía”
Jefe del Servicio de Neurocirugía
Hospital Ntra. Sra. del Rosario
Madrid
Lorena Vega-Zelaya 1,2, Rafael G. Sola 3,4, Paloma Pulido 2,4,5 and Jesús Pastor 1,2,*
1 Clinical Neurophysiology, Hospital Universitario de la Princesa, Madrid, Spain
2 Instituto de Investigaciones Biomédicas del Hospital Universitario La Princesa, Madrid, Spain
3 Professor Emeritus of Neurosurgery, Universidad Autónoma de Madrid, Spain
4 Hospital Nuestra Señora del Rosario, Madrid, Spain
5 Neurosurgery, Hospital Universitario de la Princesa, Madrid, Spain
*Correspondence: jesus.pastor@salud.madrid.org (Jesús Pastor)
Published on 25-06-2018
Abstract
In recent years, a renewed fashion for awake surgery has appeared. In spite of its undoubted utility for scientific research, this technique has several limitations and flaws, usually not debated by parts of the scientific community.
We will discuss the aims and limitations of cortical surgery, especially the points relevant to protecting the patient. These objectives should define the guidelines that direct clinical practice. We will review the awake technique as well as various tools used in intraoperative neurophysiological monitoring (IONM) to explore and monitor several cortical functions during long surgeries. The main topics discussed include electrocorticography (ECoG) and cortically recorded evoked potentials (EP), including somatosensory, visual and auditory. Later, we will discuss methods to identify and survey motor functions as motor-evoked potentials, although they are elicited trans-cranially. Finally, we will briefly discuss a promising technique to monitor some language functions in anaesthetized patients, such as cortico-cortical evoked potentials (CCEP). We will address in depth some technical questions about electrical stimulation whose full relevance are not always considered.
Finally, we will discuss why, in the absence of empirical facts showing unequivocal superiority in post-surgical outcome, we have to awaken patients, especially when an alternate possibility exists without worst clinical results, as is the case for IONM.
Keywords
: Anaesthetized surgery, awake surgery, cortical mapping, cortico-cortical evoked potentials, intraoperative neurophysiological monitoring.
INTRODUCTION
In recent years, there has been a renewed interest in surgery in awake patients [1-3]. This procedure uses the asleep-awake-sleep anaesthetic technique, which consists of induction with propofol + sevoflurane and topical blocking with svedocain + lidocaine around the skin incision. During exploration, the patient must be awoken slowly by removing the sedation. Recently, a new anaesthetic, dexmedetomidine, has been introduced for this type of surgery and is considered the most effective option [4], not only for asleep-awake- asleep technique, but for the conscious sedation one [5].
The scientific interest and relevance of this technique are undisputed. However, its clinical necessity is yet to be demonstrated, the surgery under total anaesthesia with intraoperative neurophysiological monitoring (IONM) is obviously more comfortable for both patients and medical staff. The main question that remains to be answered, therein, is whether the safety of the two techniques is equivalent.
Until now, no systematic comparison has been performed between awake and sedate craniotomy for cortical or subcortical surgery, and a definite answer remains to be established. However, both positions, for and against awake craniotomy, can be argued in this interesting debate, which is the topic of this work.
In our institutions, we have systematically performed IONM on anaesthetized patients for cortical and subcortical surgery for more than fifteen years, and we are firmly convinced of the validity of this approach.
IONM is a set of neurophysiological techniques that cannot evaluate complex functions (i.e., language function, visual or cognitive performance) but can allow the identification primary/eloquent structures with great confidence. Recently, even some parts of the language have been ascertained by cortico-cortical potentials in anaesthetised patients.
Our aim in this review is to show the powerful set of IONM techniques for use during cortical and subcortical surgery and to discuss why we think that a rational use of them can avoid the stress induced by awakening the patient.
AIMS AND LIMITS OF CORTICAL SURGERY
From a surgical point of view, the cortex can be divided into two classes: i) the primary or eloquent cortex, which cannot be removed or injured because a permanent neurological deficit would appear, and ii) the non-eloquent cortex (secondary and association areas), regions whose function can be supplied by other areas or by means of plasticity.
Cortical surgery, especially neuro-oncology, is a great challenge for neurosurgeons from two perspectives: first, in some types of tumours, gross total resection (GTR) is the best predictor of outcome in terms of life expectancy, mostly for malignant gliomas [6,7]; second, a dominant goal of every surgery is to avoid new iatrogenic lesions. The relative weight of each of these principles can be changed based on individual considerations of the type of tumour, the structures affected, the life expectancy and even the social considerations of each patient. These features are particularly relevant to patients suffering from high- grade gliomas, for whom survival is directly related to the degree of tumour removal. Therefore, to maintain an adequate quality of life, the primary goal of surgery is to achieve GTR without compromising neurological function.
These considerations are extremely relevant at the time of making decisions during the surgery, as sometimes it is more important to remove more tumour, knowing that it is placed in a non-eloquent cortex, thus allowing longer survival without neurological deficits (although some transitory deficit can usually be observed) and preserving non-primary functions at the expense of leaving greater tumour volume, which will ultimately shorten the life expectancy.
With these options in mind, our goals during cortical/subcortical surgeries are two: first, to positively identify the eloquent regions and second, to preserve those structures (cortices and tracts coming from and going to) whose injury would induce permanent deficits. Our approach, therefore, is to maximize tumour resection until a primary/eloquent structure is reached.
CORTICAL AND SUBCORTICAL SURGERY IN AWAKE PATIENTS
After the craniotomy is completed and the anaesthesia is removed, the patient wakes up in the operation room. Usually, this process takes several minutes, and it is not unusual for the patient to become somewhat agitated. After a period of haemodynamical and emotional stabilization, functional mapping begins. Cortical stimulation, which is performed by the neurosurgeon, is usually achieved through a bipolar probe with ball-tips that are separated by 0.5 cm. It is common to use 60 Hz trains over 1-4 s. The pulse width is usually 1 ms, with a current intensity of 2.5-10 mA [8,9]. During stimulation, a set of neurological and neuropsychological tests that are validated by a neuropsychologist or the patient, less commonly, by the neurosurgeon or a neurologist.
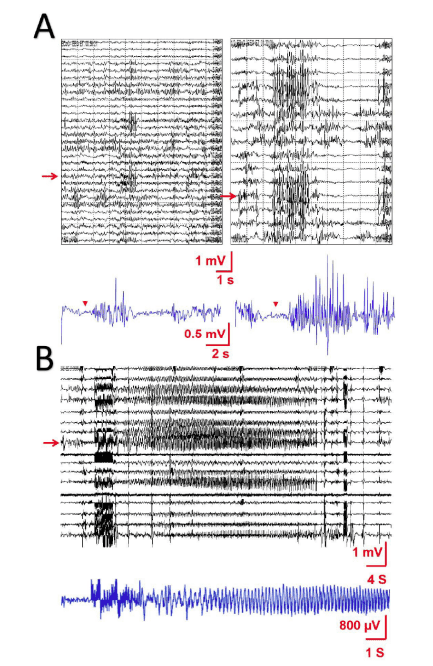
Positive responses (e.g., paraesthesia or muscular response) or alteration of complex functions (e.g., language arrest or other kinds of aphasia/paraphasia) are sought during mapping. Usually, no control of the cortex electrical activity by electrocorticography (ECoG) is performed. A low rate of intraoperative seizures has been reported (approximately 3-3.4%) [10], and some authors have concluded that control by ECoG is not mandatory [8]. However, this conclusion has been debated and remains to be validated [11]. Figure 1B shows how post-discharges appear after electrical stimulation and can be controlled by means of cold serum applied during the cortical surgery.
Subcortical pathways can be assessed by means of the same paradigm. This technique permits the identification of subcortical tracts other than IC or thalamo-cortical radiation [12].
TOOLKIT FOR IONM DURING CORTICAL SURGERY IN ANAESTHETIZED PATIENTS
We briefly expose and analyse the main techniques used by the neurophysiologist during IONM in cortical and subcortical surgery.
Electrocorticography
Several types of tumours that are located in the cortex can induce epilepsy or irritative activity, which are defined by the presence of a spike or sharp waves and combinations thereof. Hence, it is very relevant to assess the presence of these activities. ECoG can discriminate between different functional regions in the cortex, namely [13,14], i) the spiking area, where the irritative activity can be observed; ii) the lesional area, where abnormal slowing or loss of activity is observed; and iii) the non-pathological area, defined by the absence of the abovementioned activities. The identification of these regions is very important because it aids the surgeon in selecting the cortical region through which to approach the tumour, using the more injured region and avoiding the healthiest region [15,16].
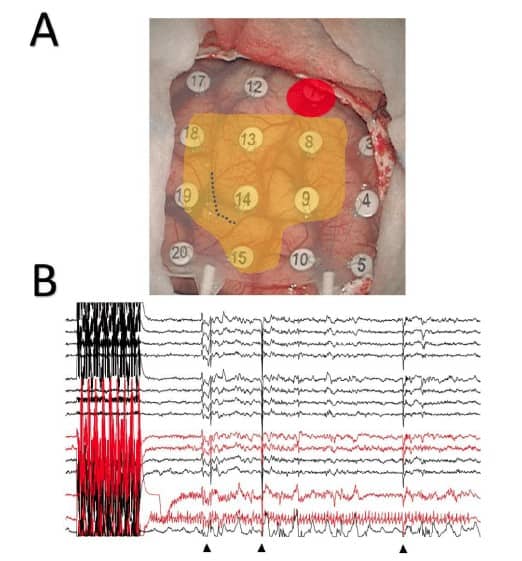
As we have stated previously, electrical stimulation can induce irritative activity or even clinical seizures (Figure 2A). Even electrocautery can inadvertently induce seizures (Figure 2B) that can then evolve. Thus, it is very important to monitor the presence of this activity during electrical stimulation of the cerebral cortex. Consequently, we propose the use of ECoG to assess the appearance of epileptiform activity not only during functional mapping but also throughout the electrical stimulation during tumour resection.
Some types of mathematical analysis can be helpful for the assessment of ECoG [16,17].
Cortical Somatosensory Evoked Potentials
During surgery of the cortex, it is very common to use the phase reversal of SSEP to identify the transition between motor and somato-sensory areas, which usually occurs at the central sulcus (CS). This recording registers the cortical activity that is generated in the primary somatosensory cortex (Brodmann areas 3, 1 and 2) in response to a stimulus on a peripheral nerve [18], usually the median nerve of the upper limb (although it is not infrequent to stimulate the ulnaris) and the posterior tibialis of the lower limb.
Thalamo-cortical projections from the ventrocaudal nucleus synapse are present in layer IV of the primary somatosensory area. However, the rostral part of Brodmann area 3 is located in the anterior wall of the central sulcus, and thus, the current sources generated by these afferents can be modelled by a dipole oriented parieto-frontally rather than in a normal position relative to the surface. This dipole source results in a reversal of the polarity when registering the potential ahead of and behind the central sulcus. This phase reversal, therefore, determines the actual location of the transition between the motor area (normally anterior to the central sulcus) and the somatosensory regions (habitually posterior).
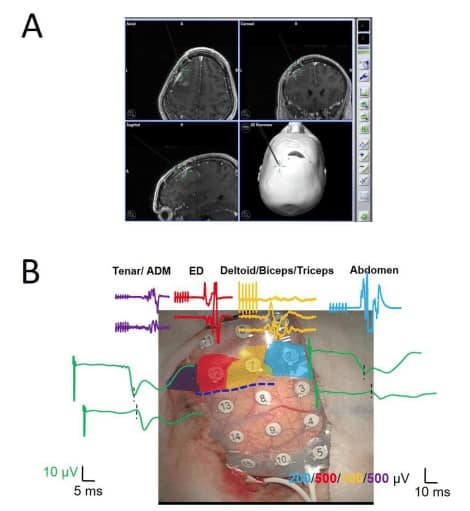
The somatosensory region corresponding to the forearm can be easily identified by the greater amplitude of the complex N1/P1/N2 waves [19]. A 20- electrode grid placed at the lateral region of the fronto- parietal transition can be very helpful. By contrast, for the lower-limb SSEP, a 4-8-electrode strip is placed at the medial region. The reference electrode should be placed as far away as possible, i.e., at the contralateral earlobe, whereas the ground electrode should be as close as possible, e.g., at the ipsilateral earlobe [15,17,18]. See Figure 3B for SSEP performed during a mapping.
It is important to perform a monopolar recording to identify the phase reversal because a differential montage can lead to very serious mistakes (see Appendix).
Cortical Auditory Evoked Potentials (cAEPs)
These potentials were initially thought to be generated in the primary auditory cortex (PAC), located deep in the white matter of the lateral fissure of the transverse gyrus of Heschl. However, other different areas, including the second auditory cortex (SAC) and the insula, are capable of eliciting cAEPs. There is considerable intersubject and interhemispheric variability [20], and the whole structure remains to be elucidated.
cAEPs are characterized by a series of waves, which can be systematized as follows:
- Short-latency waves: N13/P17/N30. These waves are typically recorded from the PAC. This complex is absent in the SAC.
- Intermediate-latency waves: peaks between 60 and 100 ms. These waves, which are always present in the SAC, can also be present in the PAC.
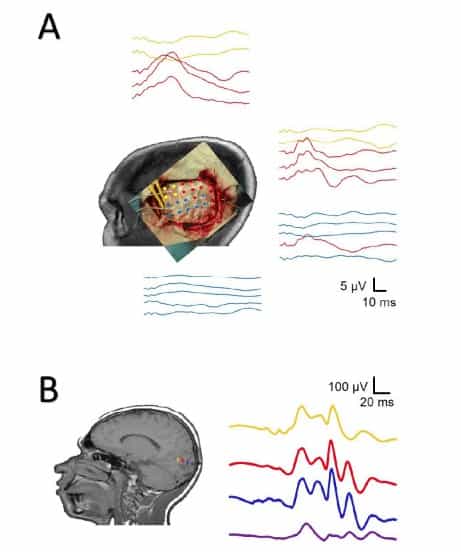
These potentials can be generated by auditory tones through earphones inserted in the external auditory channel. Considering the latencies of some waves, a minimum period of 250 ms should be used to average the signals. Therefore, a stimulation frequency of 2.18 would be adequate. A minimum intensity of 70 dB masking noise (sensation level) is applied to the contralateral ear (Figure 4A).
It is not uncommon to identify only the SAC, especially when cortical grids are used.
Visual Evoked Potentials (VEPs)
These waves exhibit the characteristics of near-field potentials generated from the primary visual cortex. In the surgery room, the only technique used for stimulation is the application of flashes of light. Although we think that VEP has undeniable utility, there have been some questions about its efficacy, mainly regarding the instability of the recording, the lack of correlation with the postoperative visual function and the high susceptibility to anaesthetic agents. However, more recent results have demonstrated stable recordings and a strong correlation with the postoperative visual function [21,22]. Therefore, intraoperative VEP monitoring will be mandatory for surgeries harbouring a risk of visual impairment [23].
Stimulation is performed by flashing light-emitting diodes stimulated at 2.18 Hz, with 10 µs pulses and a bandwidth of 10–1.000 Hz. We considered an increase in latency of 10% or a reduction in amplitude greater than 50% of the amplitude compared with the baseline as alarm criteria [16].
In some cases, VEP can be directly recorded from the cortical surface. In these cases, the potentials are much more stable, require fewer stimuli (in fact, a very small number of stimuli can induce the response) and are 2-3 orders of magnitude higher than the scalp recording [24] (Figure 4B).
Motor-Evoked Potentials (MEPs)
MEPs are the recordings that are obtained from muscles in response to stimulation of the motor system at different levels (cortex, inner capsule, cortico- spinal/cortico-bulbar tracts or spinal cord) [25]. Usually, transcranial electrical stimulation is not used, although in some patients, it is an extremely important technique [16,17,26], especially in subcortical surgery. Considering the amplitude of the response, these types of evoked potentials do not need to be averaged.
Transcranial Electrical Stimulation (TES) in Subcortical Surgery
This technique consists of the stimulation of the motor pathway by an electrical current delivered through electrodes placed outside the cranium, usually in the scalp [27].
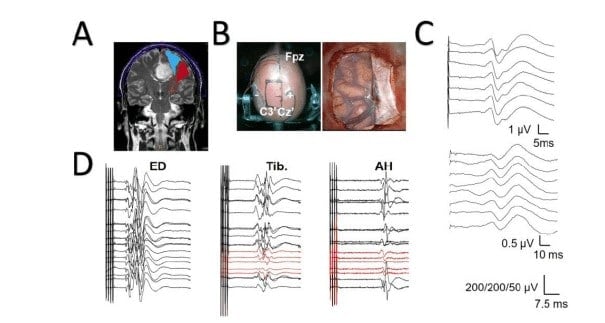
It is usually believed that TES excites the white matter of the inner capsule (IC) rather than cortical neurons. This distinction must be recognized and kept in mind by the neurophysiologist, especially in the case of surgery at the supratentorial level. However, for subcortical surgery, TES can be safely used under two conditions: i) high voltage (current) is not needed to elicit a response [26,28], and ii) a hemispheric stimulation is performed. With these preventions, TES elicits stable and specific responses that predict the postoperative outcome very well [29] (Figure 5).
Electrodes can be subdermal needles or cork- screws and are placed at different sites depending on the region to be stimulated.
The parameters used to elicit MEPs through TES are variable [30], but we use trains of 4-6 pulses with a 50-75 µs pulse width, an inter-stimulus interval (ISI) of 2 ms (i.e., 500 Hz) and voltage ranging from 120 to 450 V.
Direct Cortical Stimulation (DCS)
For this technique, electrodes are applied directly to the cortical/subcortical surface. Direct cortical stimulation (DCS) to identify the primary motor cortex (PMC) is accomplished using paired electrodes. Stimulation is performed using 4-6 pulse trains at 500 Hz (the reason we call this paradigm high frequency; this technique is also known as multipulse, which is misleading), with bi-phasic pulses of 150-200 µs in the duration/phase. Motor-evoked potentials are assessed using pairs of subdermal needles spaced approximately 2 cm apart that are inserted into the contralateral muscles, but surface electrodes attached to the skin can also be used. Depending on the location of the tumour, it is customary to use the following muscles: the orbicularis oculi, orbicularis oris, deltoid, brachial biceps, extensor digitorum carpal flexor, abductor pollicis brevis, abductor digiti minimi, quadriceps, tibialis anterior and abductor hallucis.
Stimulation is initiated at 4 mA and increased continuously in increments of 1-2 mA until a stable compound muscle action potential (CMAP) is recorded at a minimum amplitude of 30 µV or until an upper limit of 30 mA is achieved without eliciting a CMAP [16,17] (see Figure 2B).
An alternative strategy entails the use of Ojemann’s stimulation or low-frequency stimulation, which consists of a 50-60 Hz train that is 3-5 seconds in length and has a pulse width as high as 0.5 ms [3,31].
Although a systematic comparison between these two strategies remains to be performed, it is important to be aware that neither the electrical thresholds nor the muscle response or electrical safety are equivalent.
In this sense, it is important at this point to consider some electrophysiological variables concerning patient safety. See Appendix for details.
Although there are no well-defined limits for the abovementioned magnitudes, from the table, we can observe that Ojemann’s technique is the paradigm with the highest qtotal/train, and the stimulation for awake craniotomy has the highest ρmax.
Cortico-cortical Evoked Potentials (CCEPs)
As of several years ago, there is evidence that some auditory perceptive functions persist during anaesthesia [32], in addition to the presence of some auditory memory in the context of light general anaesthesia [33] or even the existence of brain responses during sleep [34]. These observations allow the supposition that complex networks associated with language are active during anaesthesia and, therefore, could be used for monitoring.
On the other hand, it is known from the end of the past century that electrical cortical stimulation can elicit responses even in the opposite hemisphere, by means of single-pulse electrical stimulation (SPES) [35]. The specificity for identification of the epileptogenic zone is still debated. However, what is beyond any doubt is that responses are not conducted through volume but driven through specific neural pathways. These cortico- cortical evoked potentials (CCEPs) have been used in IONM for language [36,37].
There are several pieces of evidence showing that complex language function can be monitored in anaesthetized patients.
In fact, during the last two years, we have used these CCEPs in IONM for the language function in patients who refused to be awake during surgery. Until now, our results have been robust but scarce, needing much more work before definite conclusions. However, we have now obtained an excellent agreement between intraoperative results and postoperative outcome.
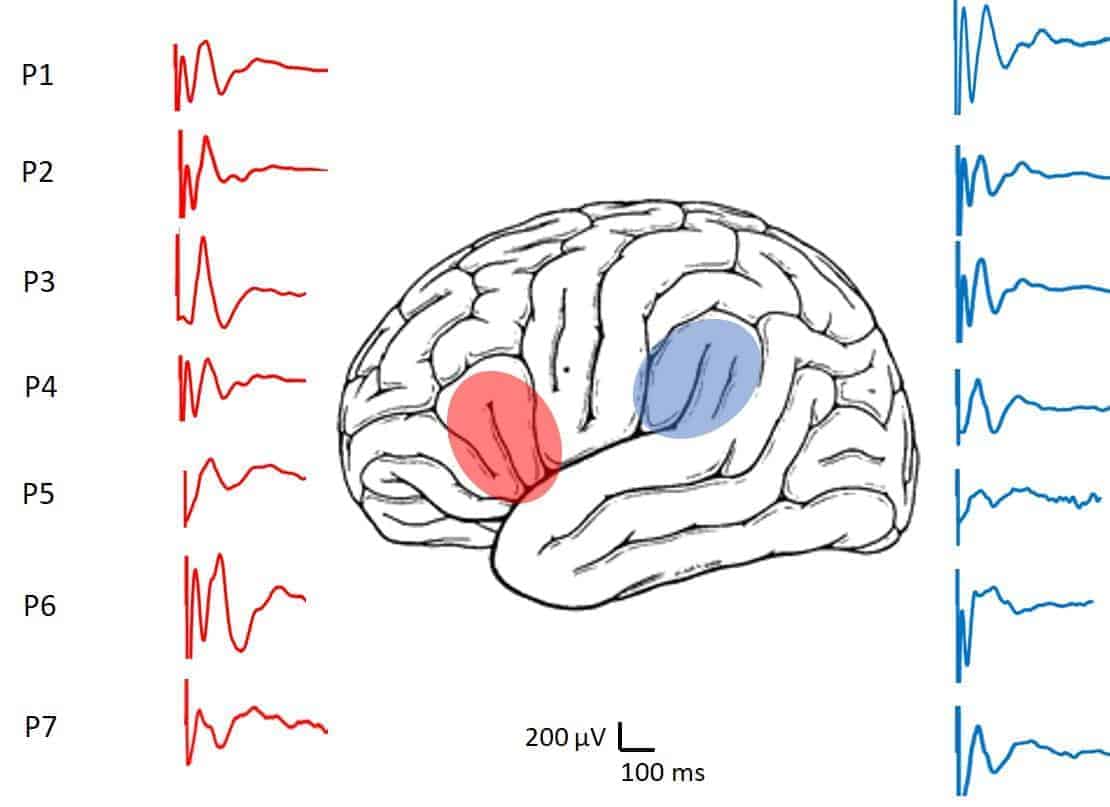
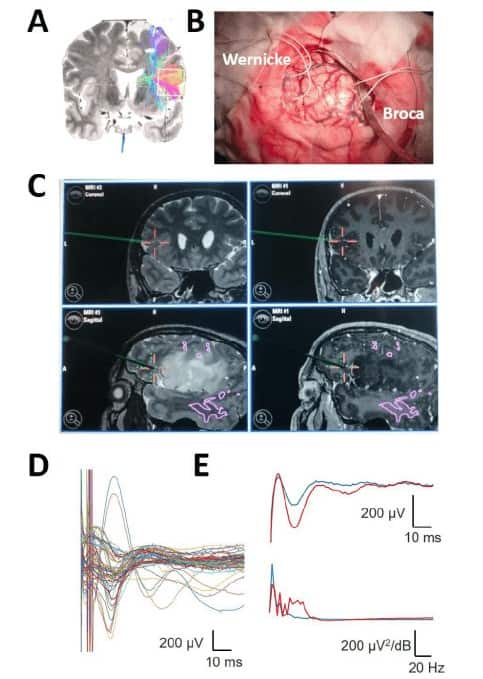
These preliminary results show the appearing of high-amplitude (> 600 µV) and stable signals during long periods of time, changing amplitude and/or latency in a reversible way during surgery (Figure 6).
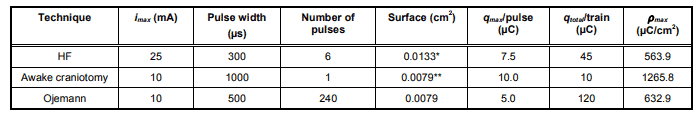
HF: high frequency. *The surface is calculated from a 1.3-mm diameter disc electrode. **The surface is calculated from a 1-mm diameter spherical electrode, assuming that only 1/4th of the surface is in contact with the cortex.
DISCUSSION
Surgery in awake patients is a fashion that has continuously grown since the last decade of the past century. It has been applied not only to cortical surgery but also to ponto-cerebellar angle surgery and trigeminal surgery. It is beyond the scope of this article to uncover the causes of this fact, which are probably multiple and complex.
Awake surgery is a powerful tool to study the human brain in a way not accessible to other techniques. Nevertheless, for clinical practice, we have other options that can offer sure results without awakening patients. In fact, awake surgery is not free of real and potential problems.
Electrical stimulation of the human cortex can induce seizures even in nonepileptic patients. Therefore, monitoring of the bioelectrical activity of the cortex should be recommended, even if the seizure rates is low, because we can reduce the probability of seizures by detecting post-discharges and impeding their evolution to seizures. In addition to this prevention, some authors have focused on possible secondary effects derived from awake surgery. A normal human response to such an exceptional situation as awake craniotomy can, for instance, result in the delayed appearance of unintentional distressing recollections of the event or some type of post- traumatic stress disorder (as yet undescribed), despite the satisfaction of the patient concerning the procedure [38].
The limitations of awake surgery must be considered seriously. During such surgeries, the patient is awake with the head fixed and covered with cloth and may be kept awake for up to 2 hours. Hence, patients must have both adequate cognitive function and the emotional maturity necessary to withstand such an environment. In fact, the Japan Society for Awake Surgery Guidelines limits the target patient population to those ranging from 15-65 years of age, although with some limitations, awake craniotomy can be used in the paediatric population [39]. Nevertheless, use in mentally handicapped patients remains problematic or impossible. In addition, although it is currently accepted that the intracranial pain-sensitive structures are limited to the dura mater and its feeding, and pain can be adequately controlled by topical anaesthesia of the skin, bone and dura, it has been observed recently that the pia and small cerebral vessels are also pain sensitive, inducing sharp, intense and brief painful events [40].
Probably the most shocking fact about the justification of awake surgery is that no differences in the immediate postoperative motor status, extent of resection have been found between IONM in anaesthetized patients and stimulation during awake craniotomy [41], although no detailed evaluation has been performed for the different techniques or surgeries. If there is not a clear difference in the post- surgical outcome, it is difficult to understand why we need to stress the patient and the medical team when we can instead perform a systematic, calm and reliable mapping and monitoring of most of the brain functions. Obviously, there are some functions, such as cognitive and language functions, that cannot be effectively assessed in anaesthetized patients. In our opinion, these kinds of patients should be the only candidates for awake surgery. However, even this statement must be nuanced. There are some promising data suggesting that some functions related to language can be mapped and monitored in anaesthetized patients. However, this option needs much more work before it can be demonstrated.
Therefore, for selected patients, an awake craniotomy is currently an option to reduce the risk of surgery-related neurological deficits, especially for language mapping. However, the benefits and risks of this type of procedure should be carefully considered, and the decision should serve the interests of the patient.
We have entitled this work with a question. Although we are conscious that a lot of work must be done before to have an unequivocal answer, our feeling and clinical compromise is to answer no.
APPENDIX
The effect of any type of electrical stimulation on the neural tissue is mediated by the total amount of charge applied to the system and the duration of application [42]. The electric current (i, in mA) is defined as follows:
Another relevant value concerning safety is the maximum charge density ( qtotal ), which is defined as qmax/A (µC/cm2/phase), where A is the area (usually in cm2). This parameter directly depends on the size and shape of the stimulation electrode.
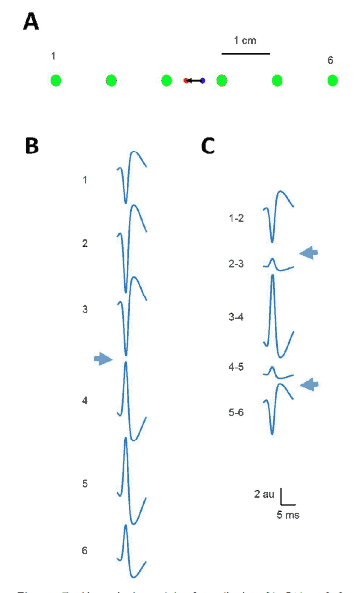
The configuration recording for phase-reversal identification during IONM is quite relevant, although no sufficient attention has been payed. The most used model for SSEP is the dipolar one [19]. Therefore, when an array of electrodes is placed in a parallel way to dipole, a phase reversal must appear in monopolar montage because some electrodes are closer to the to the opposite polarity. However, when we use a differential montage, algebraic combination of monopolar potentials gives rise to the appearing of two phase-reversals. This fact can induce serious mistakes about the identification of central sulcus.
We have addressed this fact (Figure 7) by means of a simple numerical model performed in Matlab® R2016 (Mathworks, Natick, USA). Dipolar potential (V) to different points ri (namely, electrodes of a strip) can be computed according to this expression:
Therefore, differential recordings must be strictly avoided during identification of the central sulcus.
Acknowledgment
This work was financed by a grant from the Ministerio de Sanidad FIS PI17/02193 and was partially supported by FEDER (Fonds Europeen de Developpement Economique et Regional).
References
[1] Duffau H, Capelle L, Sichez J, Faillot T, Abdennour L, Law Koune JD, et al. Intra-operative direct electrical stimulations of the central nervous system: the Salpetrier experience with 60 patients. Acta Neurochir (Wien) 1999; 141: 1157-1167. https://doi.org/10.1007/s007010050413
[2] Peruzzi P, Puente E, Bergese S, Chiocca EA. Intraoperative MRI (ioMRI) in the setting of awake craniotomies for supratentorial glioma resection. Acta Neurochir 2011; Suppl 109: 43-48.
https://doi.org/10.1007/978-3-211-99651-5_7
[3] De Witt Hamer PC, Robles SG, Zwinderman AH, Duffau H, Berger MS. Impact of intraoperative stimulation brain mapping on glioma surgery outcome: a meta-analysis. J Clin Oncol 2012; 30: 2559-2565. https://doi.org/10.1200/JCO.2011.38.4818
[4] Smith DJ, Howie MB. General Anesthesia: Intravenous and Inhalational Agents. Modern Pharmacology with clinical applications, 6th ed. Eds: Ch. R. Craig, R.E. Stitzel; Lippincott, Williams, Wilkins; Philadelphia 2003; pp. 291-309.
[5] Suero Molina E, Schipmann S, Mueller I, Wölfer J, Ewelt C, Maas M, Brokinkel B, Stummer W. Conscious sedation with dexmedetomidine compared with asleep-awake-asleep craniotomies in glioma surgery: an analysis of 180 patients. J Neurosurg 2018 Jan 12: 1-8. https://doi.org/10.3171/2017.7.JNS171312
[6] Stummer W, Reulen HJ, Novotny A, Stepp H, Tonn JC. Fluorescence-guided resections of malignant gliomas-an overview. Acta Neurochir Suppl 2003; 88: 9-12. https://doi.org/10.1007/978-3-7091-6090-9_3
[7] Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ. Fluorescence-guided surgery with 5- aminolevulinic acid for resection of malignant glioma: a randomized controlled multicenter phase III trial. Lancet Oncol 2006; 7: 392-401.
https://doi.org/10.1016/S1470-2045(06)70665-9
[8] Boetto J, Bertram L, Moulinié G, Herbet G, Moritz-Gasser S, Duffau H. Low Rate of Intraoperative Seizures During Awake Craniotomy in a Prospective Cohort with 374 Supratentorial Brain Lesions: Electrocorticography Is Not Mandatory. World Neurosurg 2015; 84(6): 1838-44. https://doi.org/10.1016/j.wneu.2015.07.075
[9] Li T, Bai H, Wang G, Wang W, Lin J, Gao H, et al. Glioma localization and excision using direct electrical stimulation for language mapping during awake surgery. Exp Ther Med 2015; 9(5): 1962-1966. https://doi.org/10.3892/etm.2015.2359
[10] Hervey-Jumper SL, Li J, Lau D, Molinaro AM, Perry DW, Meng L, et al. Awake craniotomy to maximize glioma resection: methods and technical nuances over a 27-year period. J Neurosurg 2015; 123(2): 325-39. https://doi.org/10.3171/2014.10.JNS141520
[11] Vega-Zelaya L, Sola RG, Pastor J. Intraoperative Neurophysiological Monitoring in Neurooncology. En: Neurooncology. Edited by: A. Agarwal. InTech 2016; pp: 207-249. ISBN 978-953-51-2425-2.
[12] Duffau H, Capelle L, Sichez N, Denvil D, Lopes M, Sichez JP, et al. Intraoperative mapping of the subcortical language pathways using direct stimulations. An anatomo-functional study. Brain 2002;125(Pt 1): 199-214. https://doi.org/10.1093/brain/awf016
[13] Pastor J. Conceptos de Neurofisiología en Neurocirugía Funcional, in Navarrete EG y Sola RG (ed): Neurocirugía Funcional y Estereotáxica. Barcelona: Viguera Editores, 2011, pp 17-27.
[14] Vega-Zelaya L, Torres CV, Garnes-Camarena O, Ortega GJ, García-Navarrete E, Navas M, et al. Electrocorticographic evidence and surgical implications of different physiopathologic subtypes of temporal epilepsy. Clin Neurophysiol 2014; 125: 2349-2357. https://doi.org/10.1016/j.clinph.2014.03.027
[15] Pastor J, Pulido P, Sola RG. Neurophysiological assisted transsulcal approach to a high-grade glioma without affect neither motor nor somatosensory function. Rev Neurol 2013; 56 (7): 370-374.
https://doi.org/10.1007/s00701-013-1864-0
[16] Vega-Zelaya L, Pastor J. Intraoperative neurophysiological monitoring techniques for the resection of malignant brain tumors located in eloquent cortical areas. Austin J Neurosurg 2015; 2(4): 1038.
[17] Pastor J, Vega-Zelaya L, Pulido P, Garnés-Camarena O, Abreu A, Sola RG. Role of intraoperative neurophysiological monitoring during fluorescence-guided resection surgery. Acta Neurochir (Wien) 2013; 155(12).
[18] Pastor J. Neurofisiología Intraoperatoria, in Navarrete EG y Sola RG (ed): Neurocirugía Funcional y Estereotáxica. Barcelona: Viguera Editores 2011; pp. 589-604.
[19] Mauguière F. Somatosensory evoked potentials: normal responses, abnormal waveforms and clinical applications in neurological diseases. In Niedermeyer E, Lopes da Silva A, eds. EEG, basic principles, clinical applications and related fields. Baltimore: Lippincott, Williams & Wilkins 1993; pp. 1014-58.
[20] Liégeois-Chauvel C, Trécuchon-Dafonseca A, Régis J, Marquis P, Chauvel P. Auditory evoked potentials in the definition of eloquent cortical areas. In: Presurgical assessment of the epilepsies with clinical neurophysiology and functional imaging. Eds: F. Rosenow, H. O Lüders. Elsevier, Amsterdam 2004; 305-316.
[21] Wiedemayer H, Fauser B, Armbruster W, Gasser T, Stolke D. Visual evoked potentials for intraoperative neurophysiologic monitoring using total intravenous anesthesia. J Neurosurg Anesthesiol 2003; 15: 19-24. https://doi.org/10.1097/00008506-200301000-00004
[22] Ota T, Kawai K, Kamada K, Kin T, Saito N. Intraoperative monitoring of cortically recorded visual response for posterior visual pathway. J Neurosurg 2010; 112: 285-294. https://doi.org/10.3171/2009.6.JNS081272
[23] Kodama K, Goto T, Sato A, Sakai K, Tanaka Y, Hongo K. Standard and limitation of intraoperative monitoring of the visual evoked potential. Acta Neurochirurgica 2010; 152: 643-648.doi: 10.1007/s00701010-0600-2.
[24] Torres Díaz C, Pastor J, Rocio E, Sola RG. Continuous monitoring of cortical visual evoked potentials by means of subdural electrodes in surgery on the posterior optic pathway. A case report and review of the literature. Rev Neurol 2012; 55(6): 343-348.
[25] Pastor J. Diagnóstico funcional: Neurofisiología. En: Cirugía raquimedular. Eds. C Botella, RG Sola, A Isla. Viguera Editores, Barcelona; pp: 99-114 ISBN 978-84-92931-26-2, 2014.
[26] Vega-Zelaya L, Sola RG, Pastor J. Intraoperative Neurophysiological Monitoring in Neuro-oncology, Neurooncology Amit Agrawal, IntechOpen 2016. DOI: 10.5772/63241. Available from: https://www.intechopen.com/ books/neurooncology-newer-developments/intraoperative- neurophysiological-monitoring-in-neuro-oncology.
[27] Deletis, V. Intraoperative Neurophysiology and Methodologies Used to Monitor the Functional Integrity of the Motor System. Neurophysiology in Neurosurgery. Eds: V. Deletis, J.L. Shils. Academic Press, London 2002; pp. 25-54.
[28] Vega-Zelaya L, Pastor J. Differential excitation of the inner capsule by hemispheric and trans-hemispheric electrical stimulation. Clinical and mathematical evidence. Internal Medicine Review 2018 In press.
[29] Pastor J, Perla-Perla P, Pulido-Rivas P, Sola RG. Hemispheric transcranial electrical stimulation: clinical results. Rev Neurol 2010; 51: 65-71.
[30] Szelényi A, Kothbauer KF, Deletis V. Transcranial electric stimulation for intraoperative motor evoked potential monitoring: stimulation parameters and electrode montages Clin Neurophysiol 2007; 118: 1586-95. https://doi.org/10.1016/j.clinph.2007.04.008
[31] Chang EF, Clark A, Smith JS, Polley MY, Chang SM, Barbaro NM, et al. Functional mapping-guided resection of low-grade gliomas in eloquent areas of the brain: improvement of long-term survival. Clinical article. J Neurosurg 2011; 114(3): 566-73. https://doi.org/10.3171/2010.6.JNS091246
[32] Lubke GH, Kerssens C, Phaf H, Sebel PS. Dependence of explicit and implicit memory on hypnotic state in trauma patients. Anesthesiology 1999; 90(3): 670-80. https://doi.org/10.1097/00000542-199903000-00007
[33] Iselin-Chaves IA, Willems SJ, Jermann FC, Forster A, Adam SR, Van der Linden M. Investigation of implicit memory during isoflurane anesthesia for elective surgery using the process dissociation procedure. Anesthesiology 2005; 103(5): 925-33.
https://doi.org/10.1097/00000542-200511000-00005
[34] Kouider S, Andrillon T, Barbosa LS, Goupil L, Bekinschtein TA. Inducing task-relevant responses to speech in the sleeping brain. Curr Biol 2014; 24(18): 2208-2214. https://doi.org/10.1016/j.cub.2014.08.016
[35] Alarcón G, Jiménez-Jiménez D, Valentín A, Martín-López D. Characterizing EEG Cortical Dynamics and Connectivity with Responses to Single Pulse Electrical Stimulation (SPES). Int J Neural Syst 2017 Nov 23: 1750057. https://doi.org/10.1142/S0129065717500575
[36] Yamao Y, Matsumoto R, Kunieda T, Arakawa Y, Kobayashi K, Usami K, et al. Intraoperative dorsal language network mapping by using single-pulse electrical stimulation. Hum Brain Mapp 2014; 35(9): 4345-61. https://doi.org/10.1002/hbm.22479
[37] Tamura Y, Ogawa H, Kapeller C, Prueckl R, Takeuchi F, Anei R, et al. Passive language mapping combining real-time oscillation analysis with cortico-cortical evoked potentials for awake craniotomy. J Neurosurg 2016; 125(6): 1580-1588. https://doi.org/10.3171/2015.4.JNS15193
[38] Milian Ml, Tatagiba M, Feigl GC. Patient response to awake craniotomy – a summary overview. Acta Neurochir (Wien) 2014; 156(6): 1063-70.
https://doi.org/10.1007/s00701-014-2038-4
[39] Delion M, Terminassian A, Lehousse T, Aubin G, Malka J, N’Guyen S, et al. Specificities of Awake Craniotomy and Brain Mapping in Children for Resection of Supratentorial Tumors in the Language Area. World Neurosurg 2015; 84(6): 1645-52.
https://doi.org/10.1016/j.wneu.2015.06.073
[40] Fontaine D, Almairac F, Santucci S, Fernandez C, Dallel R, Pallud J, et al. Dural and pial pain-sensitive structures in humans: new inputs from awake craniotomies. Brain 2018; 141(4): 1040-1048.
https://doi.org/10.1093/brain/awy005
[41] Nguyen HS, Sundaram SV, Mosier KM, Cohen-Gadol AA. A method to map the visual cortex during an awake craniotomy. J Neurosurg 2011; 114(4): 922-926. https://doi.org/10.3171/2010.11.JNS101293
[42] Merrill DR, Bikson M, Jefferys JGR. Electrical stimulation of excitable tissue: design of efficacious and safe protocols. Journal of Neuroscience Methods 2005; 141: 171-198. https://doi.org/10.1016/j.jneumeth.2004.10.020
[43] Plonsey R, Barr RC. Bioelectricity. A quantitative approach. Third Edition. Spinger, New York 2007; pp. 30-32.
Received on 15-05-2018 Accepted on 18-05-2018 Published on 25-06-2018