DBS-Estimulación cerebral profunda y agresión, editorial y respuesta
See the corresponding article, DOI: 10.3171/2013.4.JNS121639. ©AANS, 2013
Ver otros trabajos de la Dra. Cristina Torres
Editorial
Robert H. Howland, M.d.
Department of Psychiatry, University of Pittsburgh School of Medicine, Western Psychiatric Institute and Clinic, University of Pittsburgh Medical Center, Pittsburgh, Pennsylvania
Although deep brain stimulation (DBS) is an established neurosurgical treatment option for various movement disorders, the origins of DBS lie in the earlier use of electrical stimulation via implanted depth electrodes to study and treat emotional and behavioral disorders, which even included patients with aggressive behaviors.7 In their article, Torres and colleagues9 describe their experience using DBS targeting the posteromedial hypothalamus (PMH) in 4 male and 2 female patients who had severe chronic medication-resistant aggressiveness. The patients were 17–48 years old at the time of surgery. All of the patients had significant intellectual and neurobehavioral disabilities that necessitated institutionalized care, had had daily uncontrollable aggressiveness for at least 5 years, and had not responded to 5 or more medication trials. One patient had normal development until age 14, when his behavior problems commenced. A temporal lobe arteriovenous malformation and complex partial seizures were subsequently diagnosed. The other 5 patients had autism spectrum disorders and moderate-to-severe mental retardation, with developmental and behavioral problems that were apparent by age 2. Two of these 5 patients had had perinatal hypoxia, and 2 had seizure disorders. The final approval for DBS surgery was given after an extensive multidisciplinary evaluation, local ethics committee review, and informed consent from the patients’ parents or legal guardians.
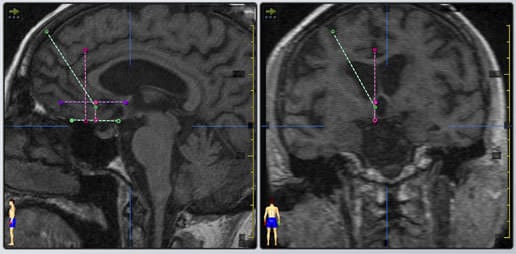
The PMH target was first localized using stereotactic procedures and then tested with microelectrode recordings and stimulation. Final electrode placement was established based on physiological responses (electrocardiographic, cervical muscle electromyographic, and electroencephalographic activity). Low-frequency lowvoltage stimulation was started within days of the surgery. Adverse effects with initial stimulation were assessed by monitoring the patients’ heart rate, blood pressure, and electrocardiographic and electroencephalographic activity. Follow-up visits were scheduled for 1 and 3 months after surgery and every 6 months thereafter. Self-aggression, outward aggression, general aggressiveness, and asocial behavior were rated using the Inventory for Client and Agency Planning (ICAP) before surgery and at subsequent visits. Laboratory studies (levels of lipids, proteins, and various hormones), body weight, and sleep patterns also were monitored. Regardless of therapeutic response, the stimulation frequency and voltage were gradually increased during the 2nd year of DBS (while other stimulation parameters were adjusted to avoid exceeding a predetermined maximum charge density).
Outcomes compared to baseline were reported only for the patients’ last follow-up visit (6–82 months after surgery). One patient died following a stroke 6 months after DBS surgery. Her family noted a progressive reduction in violent attacks during the period between surgery and the stroke. A second patient died nearly 3 years after DBS surgery but had no apparent benefit from DBS. Neither death was believed to be related to DBS. Of the remaining 4 patients, 3 showed significant improvements on ICAP measures of self-aggression, outward aggression, and general aggressiveness, with improvement noted from the 1st month of stimulation. One patient showed a significant improvement on self-aggression but not the other measures. There was no improvement for any patient on the ICAP measure of asocial behavior.
Subsequent increases in DBS stimulation frequency or voltage did not appear to result in increased therapeutic effects or adverse effects. Seizure frequency reduction was noted in one of the 3 patients with seizure disorders. Sleep patterns improved in 3 patients. Weight gain occurred in 3 patients and weight loss in 1 patient. There were no significant changes on laboratory measures.
The apparent positive benefits, tolerability, and safety of PMH DBS demonstrated in this study should be considered in the context of several important methodological limitations: small sample size, limited set of clinical assessment measures, allowance of postsurgical medication changes, lack of sham control, and use of unblinded assessments.
A small sample size makes it impossible to identify and confirm any patient characteristics that might be associated with favorable or unfavorable outcomes. For example, it is notable that the oldest patient (who was 48 years old and did not develop significant behavior problems until age 14) had no apparent benefit from DBS. Moreover, the second-oldest patient (a 37-year-old woman) showed improvement in self-aggression but not in other aggressive behaviors. The remaining 4 patients, who had better overall responses to DBS, were much younger (17–23 years old). Hence, future studies should enroll patients with a broad age range to determine whether age of onset of aggression, duration of aggression, or age at implantation are significantly associated with outcome. Also, there might be important neurobiological sex differences that are relevant to pathological forms of aggression.1 Larger studies enrolling sufficient numbers of male and female patients to be able to assess the influence of sex on outcome with PMH DBS are necessary. Finally, a larger number of patients receiving PMH DBS obviously will permit a more thorough evaluation of the safety and tolerability of this treatment.
The ICAP, a standardized rating scale for evaluating the service needs of individuals with intellectual disabilities, comprehensively includes adaptive behavior subscales, maladaptive behavior indices, and maladaptive behavior subscales. For the present study, however, the authors reported on only a small number of selected maladaptive behavior measures from the ICAP, which rated certain behaviors as “aggressive” (that is, harmful to self or others, destructive to property, or disruptive) or “asocial” (that is, socially offensive or uncooperative). In addition to self-injury and aggression, individuals with severe autism spectrum disorders and/or intellectual disabilities have a wide variety of aberrant behaviors, psychopathology, and social/functional impairment.3 The study reported no effect of DBS on “asocial” behaviors, and the authors indicated that they did not expect to modify these behaviors with stimulation. I would not necessarily agree with this expectation, however, especially based on such a small number of patients. Long-standing (at least 5 years in these patients) severely disruptive behaviors, manifested by selfinjury or aggression, are likely to affect other aspects of patients’ behaviors and functioning and to influence how others rate such behaviors and functioning. It would be useful to know whether a reduction in aggressiveness with PMH DBS results in any changes on other ICAP measures of adaptive and maladaptive behaviors, especially as they relate to the need for ongoing care and supervision, social and communication skills, personal and community living skills, and individual general functioning. It would also be important to determine whether these patients became more amenable to behavioral interventions as a result of DBS. In addition to the ICAP, future studies might include other measures that have been developed specifically for assessing self-injury, aggression, and violence.
The changes in sleep and weight seen in this study deserve further study using standardized measures of sleep, caloric intake, and activity. For example, wrist actigraphy could be used for monitoring sleep and activity levels in these patients.2 Sleep electroencephalographic
studies would be technically difficult to perform in this patient population, but such studies would provide important information about sleep physiology with PMH DBS. Measuring energy expenditure directly in patients receiving PMH DBS also would be of interest, but this type of measurement is difficult in humans.8
The authors state that “it seems unlikely that medication changes would account” for the observed therapeutic effects, but one wonders why prescribers made subsequent medication changes when patients’ aggressive behaviors began to improve early in the course of DBS. A more careful characterization of patients’ medication history, their medication regimen at the time of surgery, and the reasons for subsequent medication changes would have been helpful. Also, the authors only provide ICAP data at 2 time points—baseline and last follow-up visit. The authors state that “after the first months of DBS, the patients’ condition remained stable, with discrete fluctuations that were mostly related to stressful events.” How bad were these discrete fluctuations, and did they lead to changes in medication or other treatment? Reporting ICAP data at each follow-up visit, describing the nature of intervening “stressful events,” and quantifying medication changes would have been useful. As the authors note, a study design in which medications are kept constant would address this potential confounding factor. It would also be important to determine whether treatment with DBS is sufficiently effective to allow for safe reduction or even discontinuation of any medications.
The authors note that the PMH has been identified as a particular region of interest for the treatment of pathological aggression (using ablative procedures or DBS),6 but neural circuits involving connections between the mediobasal hypothalamus, amygdala, frontal cortex, and other regions are relevant.4 One limitation of this report is a lack of pretreatment and posttreatment neurobiological measures that could be used to characterize neural circuits of interest and to identify predictors of outcome. For example, functional MRI, PET, SPECT, magnetoencephalography, and diffusion tensor imaging would be potentially useful tools in future studies of PMH DBS for pathological aggression. The one patient who most clearly did not respond to DBS in this report had a temporal lobe arteriovenous malformation.
The design of future studies to confirm the effectiveness of PMH DBS for pathological aggression deserves careful consideration. A prospective randomized trial comparing active and sham DBS for a period of time, using independent blinded raters, followed by open-label treatment and extended follow-up is one approach. However, given the likely heterogeneity of the intended patient population, it is not clear to me that the randomized treatment groups would be sufficiently similar with respect to clinical and demographic characteristics to be able to fairly assess the effectiveness of DBS or predictors of outcome. Also, enrolling enough patients to achieve a sample size with sufficient power to evaluate outcomes of interest is potentially problematic and might require multicenter collaboration. As an alternative, a sham-controlled within-subject crossover design, using independent blinded raters, may be more feasible and clinically informative. Each patient could be randomized to receive an initial phase of double-blind sham or active treatment, then a second phase of double-blind crossover treatment, and finally extended open-label treatment. In their study, Torres and colleagues planned to titrate stimulation “dosing” upward during the course of follow-up, regardless of clinical benefit. Because they noted an improvement in behavior from the 1st month of stimulation and no apparent benefit with higher stimulation “doses,” it would be important to determine whether tolerance develops to the initial benefits of “low-dose” stimulation and whether subsequent behavioral flare-ups might then improve with stimulation adjustments.
To date, only a small number of cases in which PMH DBS was used for pathological aggression have been described in the literature, and a number of questions should be addressed. Should this procedure be offered only in the context of participation in a clinical trial or clinical registry? How do we decide whether a patient with pathological aggression is an appropriate candidate for treatment with PMH DBS? Self-injury, aggression, and violence can be significant problems for patient populations other than individuals with profound intellectual disabilities or autism spectrum disorders. Should all patients with pathological aggression be considered appropriate candidates? If not, then why not? What drug treatment and behavioral treatment algorithms should be used before declaring that a patient has uncontrollable aggression? What threshold of severity, frequency, and/or chronicity of aggressiveness should be used to determine eligibility? Torres and colleagues are to be commended for their multidisciplinary and ethical approach to the use of PMH DBS in these 6 patients, but proceeding with caution is still advised.5 (http://thejns.org/doi/abs/10.3171/2013.1.JNS122308).
Disclosure
The author reports receiving research grant support from Medtronic.
References
1. Anderson DJ: Optogenetics, sex, and violence in the brain: implications for psychiatry. Biol Psychiatry 71:1081–1089, 2012
2. Calogiuri G, Weydahl A, Carandente F: Methodological issues for studying the rest-activity cycle and sleep disturbances: a chronobiological approach using actigraphy data. Biol Res Nurs 15:5–12, 2013
3. Didden R, Sturmey P, Sigafoos J, Lang R, O’Reilly MF, Lancioni GE: Nature, prevalence, and characteristics of challenging behavior, in Matson JL (ed): Functional Assessment for Challenging Behaviors. New York: Springer, 2012, pp 25–44
4. Fumagalli M, Priori A: Functional and clinical neuroanatomy of morality. Brain 135:2006–2021, 2012
5. Gilbert F, Vranic A, Hurst S: Involuntary and voluntary invasive brain surgery: ethical issues related to acquired aggressiveness. Neuroethics 1:115–128, 2013
6. Haller J: The neurobiology of abnormal manifestations of aggression—a review of hypothalamic mechanisms in cats, rodents, and humans. Brain Res Bull 93:97–109, 2013
7. Hariz MI, Blomstedt P, Zrinzo L: Deep brain stimulation between 1947 and 1987: the untold story. Neurosurg Focus
29(2):E1, 2010
8. Melanson EL, Dykstra JC, Szuminsky N: A novel approach for measuring energy expenditure in free-living humans. Conf Proc IEEE Eng Med Biol Soc 2009:6873–6877, 2009
9. Torres CV, Sola RG, Pastor J, Pedrosa M, Navas M, García-Navarrete E, et al: Long-term results of posteromedial hypothalamic deep brain stimulation for patients with resistant aggressiveness. Clinical article. J Neurosurg [epub ahead of print June 7, 2013. DOI: 10.3171/2013.4.JNS121639]
Response
Cristina V. Torres, M.d., PH.d.,1
Rafael García de Sola, M.d., PH.d.,1 Jesús Pastor, M.d., PH.d.,2 Manuel Pedrosa, M.d., PH.d.,1 Marta Navas, M.d.,1 Eduardo García-Navarrete, M.d., PH.d.,1, Elena Ezquiaga, M.d., PH.d.,3 and Eduardo García-Camba, M.d., PH.d.3
Departments of 1 Neurosurgery, 2 Clinical Neurophysiology, and 3 Psychiatry, University Hospital La Princesa, UAM, Madrid, Spain
We would like to thank Dr. Howland for his thoughtful editorial comments on our study. We wish to add some additional data and discussion.
Deep brain stimulation, a widespread treatment for movement disorders, is nowadays being experimentally used to treat severe medication-resistant psychiatric disorders. Studies assessing the safety and effectiveness of DBS for psychiatric diseases involve specific challenges, as they raise serious ethical concerns. Severe aggressiveness in the context of mental impairment is a devastating problem that tremendously compromises the patients’ and caregivers’ quality of life. However, a surgical approach for these patients is especially problematic as the patients are unable to provide informed consent. In any experimental procedure involving psychiatric patients, doctors need to ensure that there is a favorable risk-benefit ratio for the individual patient and that the patients’ rights and interests are preserved.8 Our purpose was to substitute DBS, which is considered a reversible modality, for the permanent hypothalamic lesion proposed by Sano and Mayanagi.7
The small size of our series makes it impossible to define selection criteria for candidates; we agree with Dr. Howland that studies enrolling larger numbers of patients are necessary to identify clinical variables associated with positive responses to stimulation. We also believe that further research on the underlying causes of pathological aggressiveness would help better define different pathophysiological types of pathological aggressiveness and determine which of them are suitable for treatment with PMH DBS. Specific patterns of brain damage, such as temporal and frontal brain injury, hypothalamic and temporal lobe tumors, temporal lobe epilepsy, and Alzheimer disease, have been linked to pathological aggressiveness. Attention deficit/hyperactivity disorder and schizophrenia are also associated with abnormal violence, although in these cases structural brain damage cannot be detected.1,6
Aggressive behaviors associated with different diseases appear to differ in their clinical manifestations and responsiveness to medication; also, self-aggression and outward aggression might correspond to different pathophysiological substrates, as previously suggested.2,4–6,9,10 Imbalance in testosterone/serotonin and testosterone/cortisol ratios, as well as alterations in levels of other neurotransmitter, such as vasopressin, serotonin, and catecholamines, have been involved in the pathophysiology of abnormal aggression.9,10. As mentioned by Dr. Howland, we believe that identifying the biological measures that distinguish the different pathophysiological subtypes of aggressiveness is fundamental to determine which variables might predict positive treatment responses and which variables might normalize with treatment. Positron emission tomography studies, which have shown interesting results in psychiatric patients with a history of violence, might be used as a biological measure of treatment outcome. Investigations focusing on metabolism abnormalities in aggressive patients have suggested that decreased metabolism in the temporal and frontal cortices and hyperactivity of the limbic system, including structures such as the amygdala and hypothalamus, might be involved in the etiology of pathological aggression.9 In our study, only 3 patients underwent PET and their results were heterogeneous. A larger group of patients is necessary to allow for any conclusions regarding the possible utility of PET in the presurgical selection of candidates and monitoring of the treatment response.
In regard to the clinical assessment of these patients, we agree with Dr. Howland that optimizing the outcome measures improves our ability to monitor patients’ changes with treatment. In fact, we have recently operated on a seventh patient, not included in this study because of his short duration of follow-up, and we are carefully recording the number of hours that he has been restrained per day preand postoperatively. Other measures and specific scales would complete and improve the evaluation of patients with abnormal aggressiveness undergoing treatment, although we considered that the ICAP subscales utilized in our study reflected the clinical changes experienced by our patients. We would like to mention that patients’ medications were mainly reduced for those patients whose condition improved with DBS. In some cases, patients were transferred to different institutions at some point in their follow-up or spent longer periods of time with their families, so it was difficult to maintain the medication regimens unchanged.
Finally, we share Dr. Howland’s concern about the future research on PMH DBS for pathological aggression and urge extreme caution; we would also like to highlight the importance of the questions he brought up in his editorial. Given the large number of psychiatric diseases associated with abnormal aggressiveness, it is especially challenging to define which patients could be potential candidates for surgical treatment.
The prevalence of severe pathological aggressiveness and self-injurious behavior in patients with mental impairment has been reported to be approximately 10% and 5%, respectively. Studies suggest that disruptive behavior, especially serious physical attacks and self-injury, are not only prevalent but also persistent over time in individuals with mental impairment.2,4,5,10 Although in most cases, early behavioral interventions and medication can control its manifestations, there is still a subgroup of patients who manifest severe symptoms and whose quality of life is significantly reduced. Another concern with respect to these patients is the frequent prescription of multiple psychotropic medications in high doses, due to the potential adverse effects. Moreover, the continued use of physical restraint has given rise to ethical concerns as well, as it involves rare but potentially fatal complications, including sudden death, risk of heart disease, overarousal, and obesity.3
The advent of DBS made sham-controlled studies possible and also allowed within-subject designs to be implemented.8 However, some other problems interfere with the assessment of results in these types of studies, such as the existence of an insertion effect in some cases and the unknown duration of the stimulation effect once stimulation has been discontinued. A better understanding of the mechanisms of the therapeutic action of DBS might help to clarify these issues and to improve the design of these studies.
Given the extreme disability and vulnerability of these patients, as mentioned in our paper, it is necessary to continue in the search of effective treatments for them, while proceeding with the utmost caution, always guaranteeing that the highest ethical requirements are in place.
References
1. Casat CD, Pearson DA, Van Davelaar MJ, Cherek DR: Methylphenidate effects on a laboratory aggression measure in children with ADHD. Psychopharmacol Bull 31:353–356, 1995
2. Cooper SA, Smiley E, Jackson A, Finlayson J, Allan L, Mantry D, et al: Adults with intellectual disabilities: prevalence, incidence and remission of aggressive behaviour and related factors. J Intellect Disabil Res 53:217–232, 2009
3. Davison SE: The management of violence in general psychiatry. Adv Psychiatr Treat 11:362–370, 2005
4. Emerson E, Kiernan C, Alborz A, Reeves D, Mason H, Swarbrick R, et al: Predicting the persistence of severe selfinjurious behavior. Res Dev Disabil 22:67–75, 2001
5. Green VA, O’Reilly M, Itchon J, Sigafoos J: Persistence of early emerging aberrant behavior in children with developmental disabilities. Res Dev Disabil 26:47–55, 2005
6. Haller J, Mikics E, Halász J, Tóth M: Mechanisms differentiating normal from abnormal aggression: glucocorticoids and serotonin. Eur J Pharmacol 526:89–100, 2005
7. Sano K, Mayanagi Y: Posteromedial hypothalamotomy in the treatment of violent, aggressive behaviour. Acta Neurochir Suppl (Wien) 44:145–151, 1988
8. Schermer M: Ethical issues in deep brain stimulation. Front Integr Neurosci 5:17, 2011
9. Siever LJ: Neurobiology of aggression and violence. Am J Psychiatry 165:429–442, 2008
10. Totsika V, Toogood S, Hastings RP, Lewis S: Persistence of challenging behaviours in adults with intellectual disability over a period of 11 years. J Intellect Disabil Res 52:446–457, 2008
Please include this information when citing this paper: published online June 7, 2013; DOI: 10.3171/2013.1.JNS122308.