MOYAMOYA DISEASE
Conferencia de la Doctora Marian García Pallero sobre la enfermedad de Moyamoya
Dra. Marian García Pallero (pulsar para ver más trabajos suyos)
Servicio de Neurocirugía, Hospital La Princesa. Madrid, España
INTRODUCTION
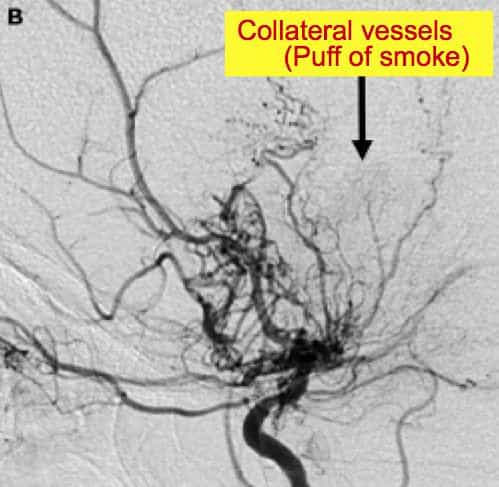
Moyamoya disease is a unique chronic progressive cerebrovascular disease characterized by bilateral stenosis or occlusion of the arteries around the circle of Willis with prominent arterial collateral circulation. «Moyamoya» is a Japanese word meaning puffy, obscure, or hazy like a puff of smoke in the air. Thus, the term was used to describe the smoky angiographic appearance of the vascular collateral network.
Moyamoya disease was first described in Japan in 1957. Many similar cases have subsequently been reported, mainly in Japan and other Asian countries. The disease is found less frequently in North America and Europe.
ETIOLOGY
The etiology of moyamoya disease is unknown. The high incidence among the Japanese and Asian population, together with a familial occurrence of approximately 10 to 15 percent of cases, strongly suggests a genetic etiology.
Accumulating evidence suggests that the RNF213 gene on chromosome 17q25.3 is an important susceptibility factor for MMD in East Asian populations. Although the mode of inheritance is not established, one study suggested that familial moyamoya is an autosomal dominant disease with incomplete penetrance.
Associated conditions
Classic angiographic findings of moyamoya vessels have been demonstrated in patients with other medical conditions. This has led some investigators to propose the term «moyamoya phenomenon» or «moyamoya syndrome» as an entity separate from true idiopathic moyamoya disease.
Patients with the angiographic appearance of moyamoya and with one of the well-recognized associated conditions (neurofibromatosis type 1, cranial irradiation, Down syndrome, and sickle cell disease) are classified as having moyamoya syndrome.
EPIDEMIOLOGY
In epidemiologic surveys conducted in Japan, the following observations have been made:
– The annual incidence of moyamoya is 0.35 to 0.94 per 100,000 population
– The prevalence of moyamoya is 3.2 to 10.5 per 100,000 population
– There is a female predominance, with a male-to-female ratio of 1:1.8 to 1:2.2
– A family history of moyamoya disease is present in 10.0 to 15.4 percent of patients
A study that analyzed hospital discharge data from Washington and California in the western United States found an estimated overall moyamoya incidence of 0.086 per 100,000. Among ethnic groups in California, the moyamoya incidence rate for Asians was 0.28 per 100,000. The incidence rates were lower for blacks, whites, and Hispanics (0.13, 0.06, and 0.03 per 100,000, respectively).
Age distribution
— Moyamoya disease and moyamoya syndrome occur in children and adults of all ages, although presentation in infancy is rare. A cohort study of 802 patients with moyamoya disease from China also demonstrated a bimodal age distribution, with a major peak in the 5 to 9 year old group and another peak in the 35 to 39 year old group.
CLINICAL FEATURES
The clinical manifestations of moyamoya are variable and the expression of disease may differ by age at the time of diagnosis.
– Ischemic cerebrovascular events, either TIA or infarction, are more prevalent than hemorrhagic events in children
– Hemorrhagic stroke (mainly intraparenchymal and intraventricular hemorrhage) is more common in adults
Among symptomatic patients with moyamoya disease, some have only one or a few events, while others have multiple recurrences.
In a study from Korea with 88 patients (36 of whom had revascularization surgery) from 1 to 75 years of age at presentation who were followed for 6 to 216 months, the following outcomes were reported:
– A single ischemic or hemorrhagic cerebrovascular episode was noted in 40 patients (45 percent).
– Recurrent ischemic and hemorrhagic cerebrovascular events occurred in 48 patients (55 percent).
– The most frequent neurologic deficit was hemiparesis in 64 percent of cases. Additional common sequelae were aphasia, seizure, altered mentation, and visual disturbance.
PATHOLOGY
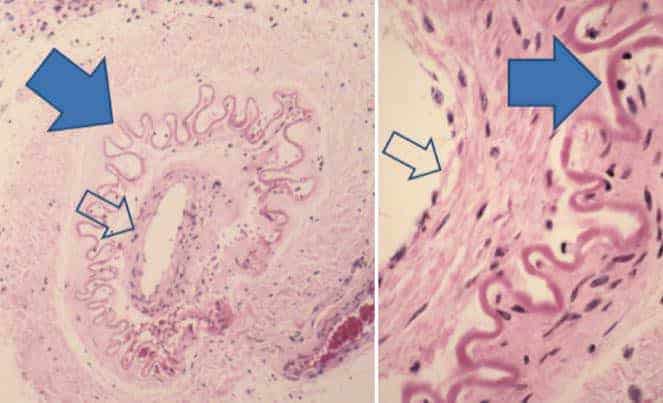
The pathology of the smaller perforating vessels in moyamoya is variable. Morphometric analysis suggests that some are dilated with relatively thin walls, while others are stenotic with thick walls.
In the picture, we can see the fibrosis and thickening of the intima and proliferation of smooth muscle cells (open arrows) and in-folding and chronic contraction of the internal elastic lamina (closed arrows), as well as notable absence of inflammatory or atherosclerotic changes.
Vascular stenosis
Pathologic vascular lesions appear in the large vessels of the circle of Willis and in the small collateral vessels. Bilateral concentric stenosis or occlusion is consistently found in the distal internal carotid arteries and the proximal anterior and middle cerebral arteries. Less frequently, the posterior circulation is affected, especially the posterior cerebral artery.
Collateral vessels
One of the hallmarks of moyamoya disease is the presence of a collateral meshwork of overgrown and dilated small arteries, the moyamoya vessels, that branch from the circle of Willis.
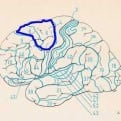
Leptomeningeal vessels are another source of collaterals in moyamoya (image 5). These collaterals result from dilatation of preexisting arteries and veins. In addition, transdural anastomoses, termed vault moyamoya, may develop from extracranial arteries such as the middle meningeal and superficial temporal arteries.
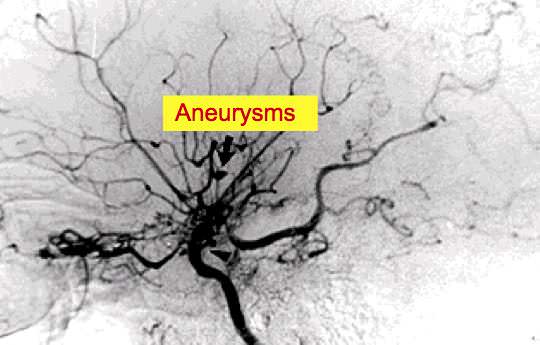
Aneurysms
– Large artery aneurysms can develop at vessel branching points in the circle of Willis and cause subarachnoid hemorrhage when they rupture.
– Aneurysms can also arise from the small collateral moyamoya vessels, choroidal arteries, or other peripheral collateral arteries. These small vessel aneurysms are the major cause of parenchymal (intracerebral) hemorrhage in moyamoya disease.
Extracranial involvement
In patients with moyamoya disease, stenosis due to fibrocellular intimal thickening may also affect the extracranial and systemic arteries, including the cervical carotid, renal, pulmonary, and coronary vessels. Involvement of the renal arteries has been most frequently reported.
NEUROIMAGING
Head CT
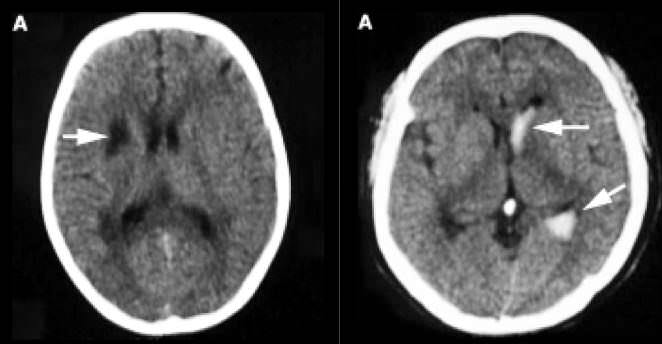
It´s an important study for the detection of brain infarction and hemorrhage.
Infarction may involve cortical and subcortical regions (image 1). Dilatation of the sulci, accompanied by focal ventricular enlargement indicating volume loss, is usually found in the chronic phase of disease.
In patients with parenchymal hemorrhage, cranial CT usually shows a high density area indicating blood in the basal ganglia, thalamus, and/or ventricular system (image 2).
Brain MRI
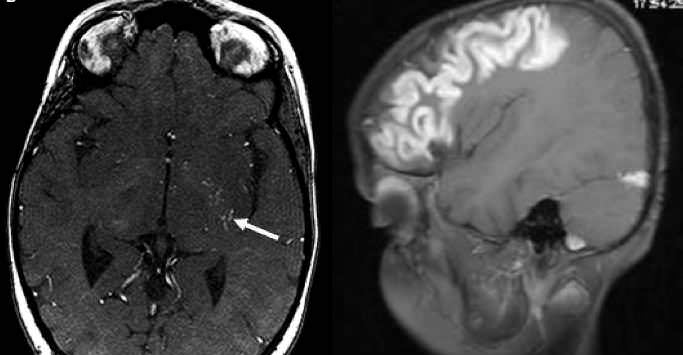
Brain MRI, particularly diffusion and perfusion MR techniques, is superior to CT scan for detection of small and/or acute ischemic brain lesions. In some cases, dilated collateral vessels in the basal ganglia and thalamus can be demonstrated as multiple punctate flow voids, a finding which is considered virtually diagnostic of moyamoya syndrome.
In both fluid-attenuated inversion recovery (FLAIR) images and post contrast T1 images may show a linear pattern of increased signal in the leptomeninges and perivascular spaces. This pattern has been termed the «ivy sign», since it resembles the appearance of ivy creeping on stones.The probable cause is slow retrograde collateral flow through engorged pial vessels via leptomeningeal anastomosis.
Angiography
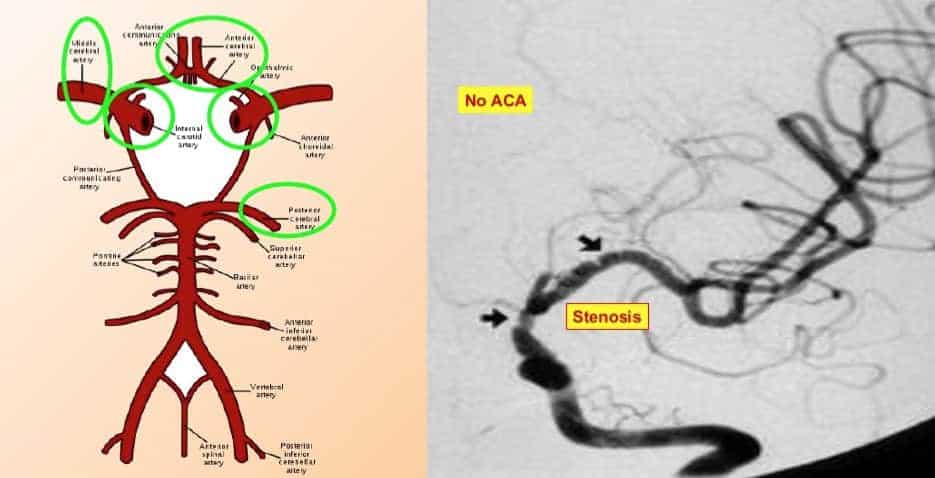
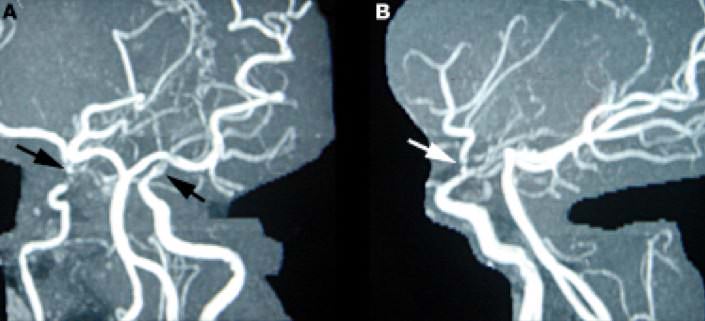
MR angiography (MRA) can demonstrate stenotic or occlusive lesions.
In the picture, we can see a severe narrowing of the distal part of the both carotid arteries (arrows). In addition, there is markedly reduced flow in the left middle cerebral artery and absence of both anterior cerebral arteries.
In addition, MRA can visualize the collateral «moyamoya vessels» in the basal region, although it is less sensitive to smaller vessel occlusion than conventional cerebral angiography.
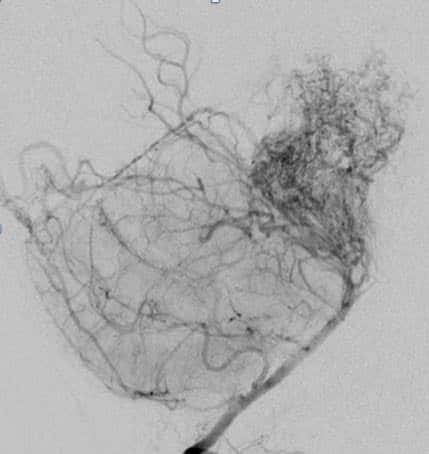
Conventional cerebral angiography is the gold standard for the diagnosis of moyamoya disease.
The picture shows a cerebral angiogram (lateral projection) demonstrates total occlusion of the left internal carotid artery and collateral flow from the external carotid system.
The Suzuki angiographic stages of moyamoya disease:
Suzuki and colleagues followed patients with moyamoya disease and classified the angiographic progression into six stages:
– Stage 1 – Narrowing of carotid fork only
– Stage 2 – Initiation of basal moyamoya with dilatation of all main cerebral arteries
– Stage 3 – Intensification of moyamoya together with reduction of flow in the middle and anterior cerebral arteries
– Stage 4 – Minimization of moyamoya vessels; the proximal portions of the posterior cerebral arteries become involved
– Stage 5 – Reduction of moyamoya and absence of all main cerebral arteries
– Stage 6 – Disappearance of moyamoya vessels; the cerebral circulation is supplied only by the external carotid system
Moyamoya disease clinical cases
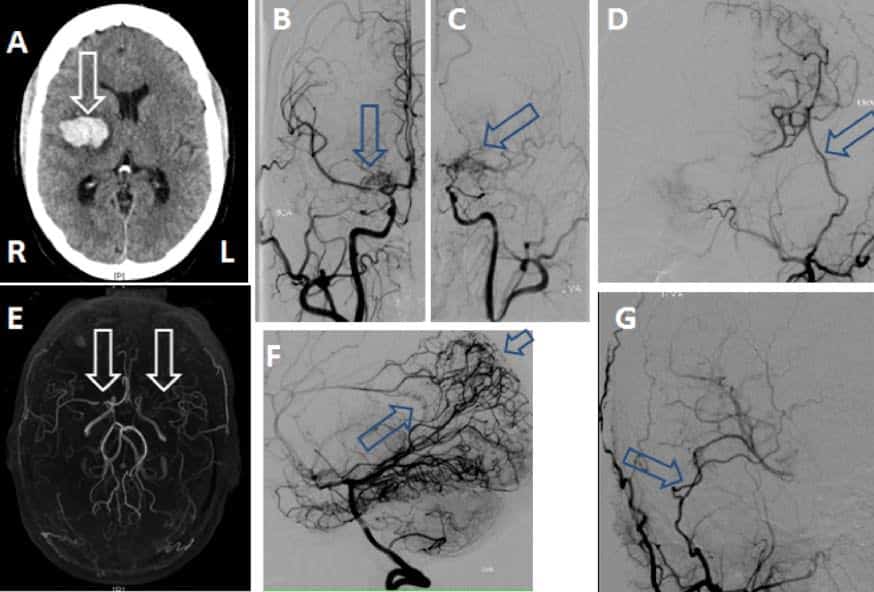
This figure illustrates one of the typical presentations and diagnostic findings in an adult patient with hemorrhagic moyamoya disease. The patient was found to have an intraparenchmal hemorrhage(arrow) on plain CT (A). MRA (E) showed severe stenosis of the supraclinoid carotid and middle cerebral artery on the right (arrow) and occlusion of the middle cerebral artery on the left (arrow). Cerebral angiography (B right, C left) revealed classic findings consistent with moyamoya with basal vessel narrowing and collateral vessel changes including bilateral hypertrophy of the fine perforating arteries as well as collateral development from the posterior circulation (F) with extensive leptomeningeal collateral vessels (small arrow) and hypertrophied splenial vessels (large arrow) supplying the anterior cerebral arteries.
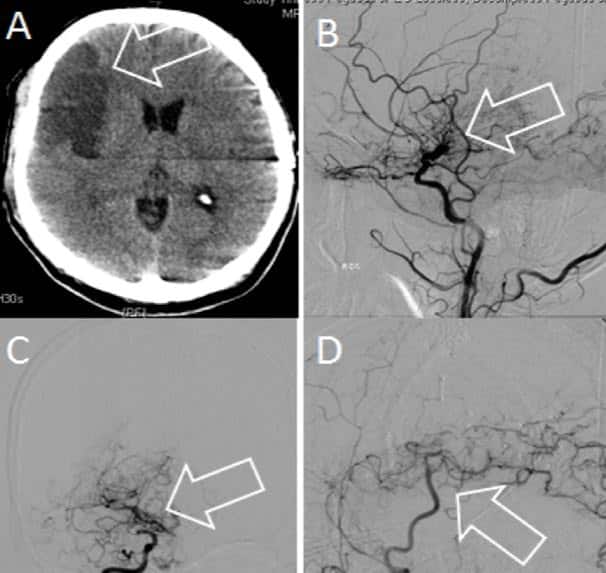
This figure illustrates an acute infarction (arrow) on CT (A) caused by ischemic moyamoya in an adult. Cerebral angiography showed bilateral internal carotid occlusions and extensive fine basal collateral (moyamoya) vessels (B and C). Direct superficial temporal to middle cerebral artery bypass (arrow) with filling of the temporal cortical vessels (D)
DIAGNOSIS
Moyamoya disease is one of the differential diagnoses of stroke in children or young adults.
Diagnostic criteria — Diagnostic criteria for idiopathic moyamoya disease proposed by a Japanese research committee include the following major requirements:
– Stenosis or occlusion at the terminal portion of the internal carotid artery and at the proximal portion of the anterior and middle cerebral arteries on magnetic resonance angiography (MRA)
– Abnormal vascular networks in the basal ganglia on MRA; these networks can also be diagnosed by the presence of multiple flow voids on brain MRI
– Angiographic findings are present bilaterally; cases with unilateral angiographic findings are considered probable
– The following conditions should be excluded: Arteriosclerosis, autoimmune disease, brain neoplasm, a history of cranial irradiation,down syndrome, head trauma, neurofibromatosis, meningitis
ACUTE MANAGEMENT
For children and adults with moyamoya and acute stroke, acute treatment is mainly symptomatic and directed towards reducing elevated intracranial pressure, improving cerebral blood flow, and controlling seizures.
During hospitalization for acute stroke or surgery, the following management issues deserve special attention, especially in children with moyamoya:
– Precautions to minimize crying and hyperventilation.
– Pain control measures.
– Avoidance of hypotension, hypovolemia, hyperthermia, and hypocarbia
– Administration of supplemental oxygen.
SECONDARY PREVENTION
There is no curative treatment for moyamoya disease.
Secondary prevention for patients with symptomatic moyamoya is largely centered on surgical revascularization techniques: The goal of surgical treatment for moyamoya disease is to reduce the risk of ischemic stroke by improving the cerebral circulation. Thus, surgical procedures are used most often for patients with ischemic-type moyamoya who have cognitive decline or progressive symptoms.
Antiplatelet agents, have been use to treat some patients with moyamoya disease or moyamoya syndrome, particularly those who are asymptomatic or have mildly symptomatic ischemic disease, or those considered to have a high risk for poor surgical outcome.
Oral anticoagulants are seldom used in children with ischemic moyamoya disease because of the risk of hemorrhage after incidental trauma and because of the difficulty maintaining therapeutic levels. In adults, hemorrhage is the predominant manifestation of moyamoya, and anticoagulation is generally not indicated.
Revascularization surgery:
Surgical techniques can be divided into direct and indirect revascularization procedures and their combinations:
– Direct revascularization: Superficial temporal artery to middle cerebral artery (MCA) bypass or middle meningeal artery to MCA bypass are the most common direct techniques. Direct methods are technically difficult to perform in children because of the small size of donor and/or recipient vessels.
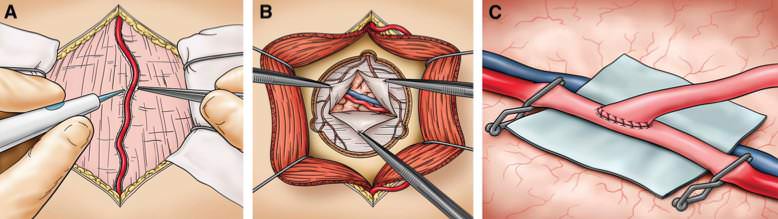
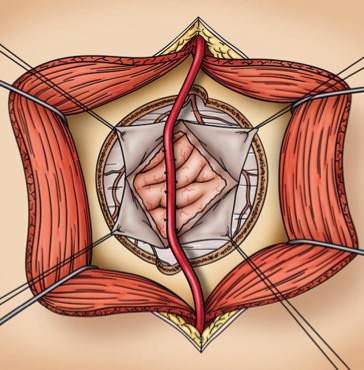
The picture shows a superficial temporal artery (STA)–to–middle cerebral artery (MCA) bypass. A, after opening of the skin, the STA is dissected. B, a craniotomy is performed with multiple burr holes to preserve the middle meningeal artery, and the dura is opened to find a recipient branch of the MCA. C, the anastomosis is performed.
– Indirect revascularization is preferred at other centers, particularly in cases where the cortical recipient artery is not available for anastomosis. The technique aims to promote the development of a new vascular network over time.
Indirect techniques include the following:
-
– Encephaloduroarteriosynangiosis and a modification called pial synangiosis (Encephaloduroarteriosynangiosis. This procedure involves suturing a branch of the superficial temporal artery with its associated cuff of galea connective tissue to the pial surface of the cerebral cortex)
– Encephalomyosynangiosis, Encephaloarteriosynangiosis, Encephalodurogaleosynangiosis, Omentum transplantation, Craniotomy with inversion of the dura, Multiple burr holes without vessel synangiosis, Cervical sympathectomy.
Complications of surgery
Complications of surgery are perioperative stroke, cerebral hyperperfusion syndrome and subdural hematoma.
The reported incidence of clinically symptomatic cerebral hyperperfusion syndrome following bypass surgery for moyamoya disease ranges from 15 to 47 percent.
SURGICAL ADVANCES:
Several advances pertaining to the surgical procedure itself have occurred in recent years. One is the introduction of indocyanine green videoangiography into the armamentarium for evaluating graft patency and flow at the time of anastomosis.
Another advance is the application of an intraoperative perivascular flow probe that uses ultrasonic transit-time principles to directly measure blood flow (rather than velocity) to assess the patency and flow of a direct bypass.
Finally, there have been efforts to reduce or eliminate the ischemia time during construction of a surgical bypass with the idea that this might reduce perioperative ischemic complications. One such technique is the excimer laser–associated nonocclusive anastomosis. This involves sewing a platinum ring into a donor vessel, which is then attached to the recipient vessel. A laser catheter is then inserted through a slit in the donor vessel and used to make the arteriotomy. The advantage of this technique is that there is no need for temporary occlusion of the recipient vessel.
Another approach to reduce temporary artery occlusion time during bypass is the use of an automated anastomosis system that can complete a vascular anastomosis within seconds. One such system (the C-Port Flex-A Anastomosis System, Cardica, Inc) has been successfully used in humans for high-flow bypasses. However, modifications to the device will be required to permit its use for STA-to-MCA bypass procedures.
Efficacy of surgery
There have been no randomized, controlled studies to determine the effectiveness of surgical treatment for moyamoya. The available evidence consists of retrospective case series and case reports.
– A systematic review published in 2005 identified 55 studies with data for 1156 children (mainly from Japan) who had surgical revascularization for moyamoya disease or moyamoya syndrome. Most patients were treated with indirect surgical techniques or a combination of direct and indirect methods (73 and 23 percent, respectively). Over a mean postoperative follow-up of 58 months, the following observations were reported:
- •Symptomatic benefit, defined as complete disappearance or reduction in symptomatic cerebral ischemia, was reported in 1003 children (87 percent).
•The perioperative stroke rate was 4 percent
•There was no statistically significant difference in outcome between the indirect and direct plus combined treatment groups
There are no controlled studies directly comparing medical and surgical therapy for moyamoya, and indirect evidence is equivocal. In a large survey study from Japan, there was no statistically significant difference in outcome between patients with moyamoya disease treated with revascularization surgery or medical therapy. However, in another large survey study from Japan, children with moyamoya who were initially managed nonoperatively developed progressive symptoms that prompted surgery in 38 percent, a rate much higher than the rate of reoperation in children initially managed with surgery.
Conclusions
Advances in neuroimaging have led to an increased recognition of the disease worldwide.
Surgical revascularization using either the indirect or direct method is widely used for the treatment of moyamoya.
The use of revascularization for the treatment of pediatric and adult patients presenting with ischemic symptoms is supported by multiple cohort studies, but its efficacy has not been established by randomized controlled trials.