Right-sided vagus nerve stimulation therapy in adult patients after failed left-sided placement.
Authors
M. Navas García, E. Garcia Navarrete, J.M. Pascual, R. Carrasco, Shakur SF, R. Gil, R.García de Sola.
Neurosurgery Department, La Princesa University Hospital, Madrid, Spain.
Email: m_navas_garcia@hotmail.com
Abstract
Introduction and Objectives: Vagus Nerve Stimulation (VNS) is a non-pharmacological alternative therapy for decreasing seizure frequency in patients with refractory epilepsy, usually placed on the left vagus nerve. Nonetheless, left side VNS (L-VNS) can be hindered because of the presence of intraoperative complications, postoperative fibrosis, or even mechanical malfunction of the device, forcing L-VNS explant. Due to in animal models right side VNS (R-VNS) have shown to be as effective as left-sided stimulation in reducing the frequency of seizures, R-VNS could be considered in patients who may not tolerate L-VNS. The objective is to examine our experience of R-VNS placement in two patients with intractable epilepsy.
Methods: We performed a retrospective review of two adult patients, who underwent a R-VNS procedure due to complicated or failed previous L-VNS implantation.
Results: One of the patients was treated with L-VNS for 13 years but the device had to be removed because of mechanical malfunction. The second patient could not be implanted with L-VNS due to significant bleeding caused by the accidental tearing of an ectopic vein. Both patients were thought to be at high risk for left vagus nerve injury if L-VNS reimplantation was attempted, thus R-VNS was chosen. R-VNS therapy successfully reduced seizure activity without causing cardiac side effects.
Conclusions: We conclude that R-VNS placement is an alternative therapy in those patients who cannot undergo L-VNS implantation. Close follow-up and frequent ECG monitoring is recommended following each increase in stimulation intensity, in order to detect the presence of cardiac side effects.
Navas M, Navarrete EG, Pascual JM, Carrasco R, Nuñez JA, Shakur SF, Pastor J, Sola RG. Epilepsy Res. 2010 Jun; 90(1-2): 1-7
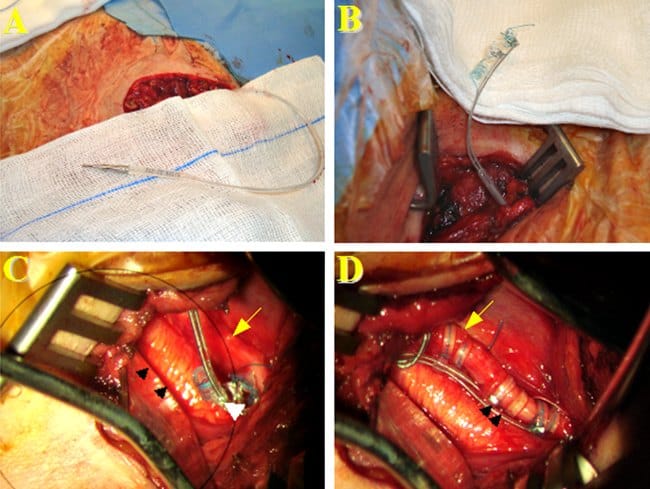
Patient 1
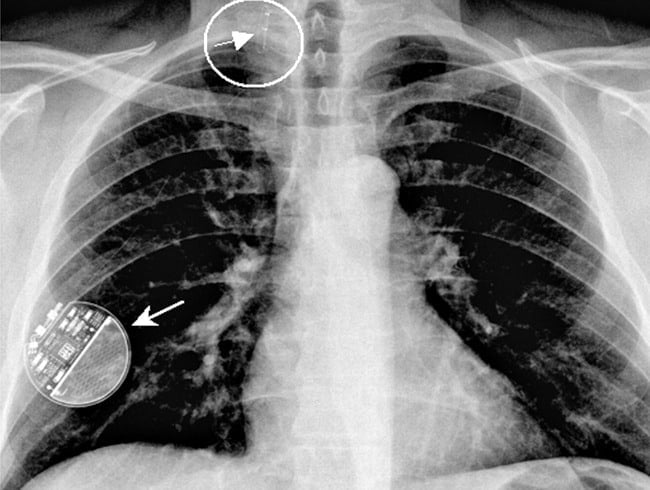
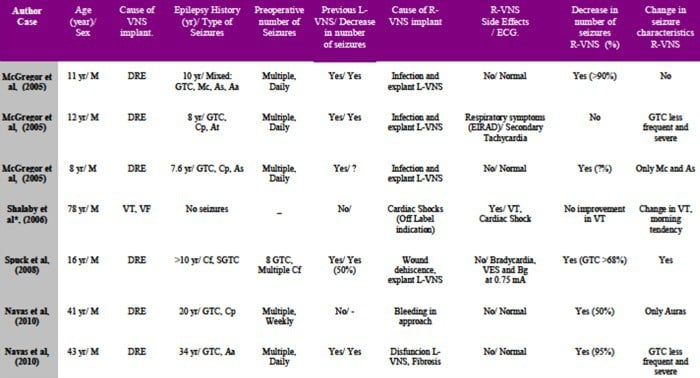
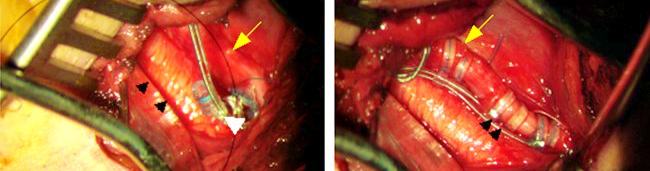
41-year-old patient suffering from refractary epilepsy since he was 20. Seizure frequency was approximately three times per week (complex focal and generalized tonic-clonic episodes). After a left temporal lobectomy and amygdalohippocampectomy, a 75% decrease in the total number of seizures was achieved (Engel III-A). However, two years after surgery the patient reported a seizure frequency similar to his preoperative seizure rate (Engel IV-B). A new video-EEG study showed undifferentiated bioelectrical basal activity, so the patient was considered for implantation of a VNS. The first attempt to implant a L-VNS via left anterolateral cervicotomy failed due to significant bleeding of thyroid veins and the presence of a thin vagus nerve that was tightly adhered to carotid sheath. Because the patient was at high risk of vascular injury and rebleeding, he was considered for placement of a R-VNS. The preoperative ECG and Holter monitoring did not show any abnormality. The patient subsequently underwent R-VNS (Cyberonics® 302 model) implantation, via right anterolateral cervicotomy and right para-axillary generator (Cyberonics® 102 model) placement. A standard lead test and device diagnostics were performed in the operating room without problems and no arrhythmias were observed. Holter monitoring performed over the next twenty-four hour was normal.The device was turned on four weeks after surgery; VNS parameters used were: on time 30 seconds, off time 5 min, frequency 30 Hz, 250 μs pulse width and 0.25 mA stimulation intensity with a monthly increase of 0.25 mA (2 mA at the last follow up). Following each increase in stimulation intensity, we performed a continuous 24-hour electrocardiogram in order to rule out the presence of arrthythmias or impairments in hearth rate; all of the Holter studies were normal. One year after right VNS implantation, the patient reported improved quality of life (Karnofsky 70), as well as a decrease in number (50%) and intensity of seizures.
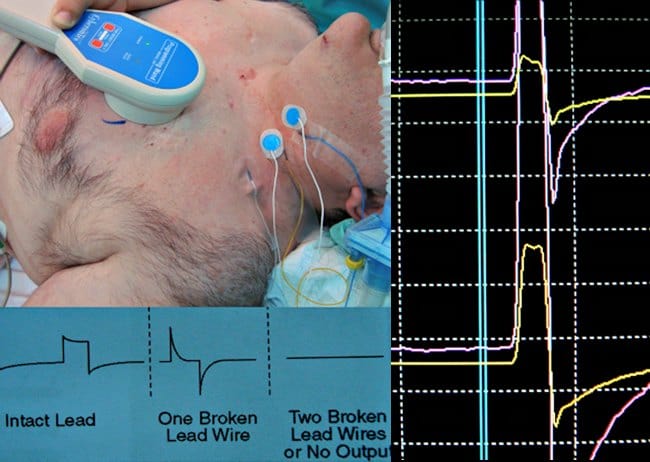
Patient 2
43-year-old man suffering from refractory epilepsy since he was 9 years-old. He reported daily atypical absence seizures with automatisms and occasional generalized tonic-clonic episodes. The patient underwent L-VNS placement at another institution when he was 27 years-old, and complete control of seizure activity was achieved for 13 years. At the time of subsequent battery replacement, the device became non-functional and the patient experienced a significant increase in seizure frequency. The patient was transferred to our neurosurgical department for further evaluation. L-VNS telemetry revealed a device dysfunction, high impedance values, and full battery. Before L-VNS explant, intraoperative Evoked Potential Monitoring (EPM) was used to analyze the stimulus waveform from the neck, for verification of an electrical discontinuity. The L-VNS generator and distal lead were removed, but the proximal lead was hardly attached at left vagus nerve due to dense scarring; local scarred tissue and the short distance of available vagus nerve did not allow placement of a new L-VNS. Therefore, a R-VNS (Cyberonics® 302 model) and a right para-axillary generator (Cyberonics® 102 model) were implanted. Intraoperative test stimulation ( 1 mA, 5 s) did not show any cardiac side effects. R-VNS was turned on two days after surgery. VNS parameters used were: on time 30 seconds, off time 5 min, frequency 30 Hz, 250 μs pulse width and 0.25 mA stimulation intensity with a monthly increase of 0.25 mA. The stimulation parameters were increased progressively to 1 mA over the next six next months; each increase in stimulation parameters was followed by Holter monitoring; side effects, particularly cardiac symptoms, were not detected. At last follow-up, 7 months after surgery, a decrease in 95% of the total number of seizures was noted. The patient’s antiepileptic drug regimen remained unchanged for 7 months after surgery.