Usefulness of Reintervention in Recurrent Glioblastoma: An Indispensable Weapon for Increasing Survival
– BACKGROUND: Glioblastoma is the most frequent primary brain tumor and despite of complete treatment survival is still poor. The aim of this study is to define the utility of reoperation for improving survival in patients with recurrent glioblastoma, and determine other prognostic factors associated with longer survival.
– METHODS: We performed a retrospective analysis of those patients who underwent surgery and compared those who were operated two or more times and those who received surgery only once. We studied overall survival (OS), progression-free survival (PFS), and clinical variables that could be related with higher survival.
– RESULTS: A total of 121 patients were eligible for the study, of whom 31 (25%) underwent reoperation. The reoperation group had a mean and median increase survival of 10.5 and 16.4 months in OS and 3.5 and 2.7 months for PFS compared with the non-reoperation group (P <. 0.001 and 0.01, respectively). Although complications were higher in patients that underwent reintervention (19.3%) there was no statistical difference with complication rate in first surgery (12.4%, c2 [ 1.86; P [ 0.40). Cox multivariable analysis revealed that age (hazard ratio [HR] 1.03; 95% confidence interval [CI], 1.006e1.055; P [ 0.013), reoperation (HR, 0.48; 95% CI, 0.285e0.810; P [ 0.006), extent of resection 95% (HR, 0.547; 95% CI, 0.401e0.748; P <. 0.001), and complete adjuvant therapy (HR, 0.389; 95% CI, 0.208e0.726; P [ 0.003) were correlated with a higher OS.
– CONCLUSIONS: Reoperation and the extent of resection (EOR) are the only surgical variables that neurosurgeons can modify to improve survival in our patients. Higher EOR and reoperation rates in patients who can be candidates for second surgery, will increase OS and PFS.
![]()
INTRODUCTION
Glioblastoma is the most common primary brain tumor in adults, with a devastating prognosis.1,2 The incidence of glioblastoma is 5e10 cases per 100,000 persons per year, representing 65% of newly diagnosed gliomas.3 Despite the efforts to improve survival in affected patients, life expectancy is limited, ranging from 12 to 15 months, with <.10% of patients alive after 5 years.4 Typically, glioblastoma recurs after 1 year despite maximal treatment when surgery, chemotherapy, and radiotherapy are administered.4,5 To improve prognosis, new treatments and strategies have been developed for recurrence, based on the use of new angiogenesis inhibitors, targeted therapies, or gamma knife surgery.6,7 Surgical treatment has become more aggressive, and reoperation is a more frequently used approach for these patients. In 1968, Pool8 introduced reoperation for recurrent glioblastoma, and few studies have evaluated the impact of reoperation in glioblastoma recurrence, although it can be an important tool for increasing overall survival (OS) and progression-free survival (PFS) in selected patients. Some evidence has been published, mainly by Berger et al.6,9-11 and also by other authors, indicating similar results.12,13 However, there is a lack of information regarding the possible effectiveness of this strategy and possible prognostic factors. The aim of the present study was to elucidate the impact of reoperation for recurrent glioblastoma and to identify prognostic factors that can improve survival in these cases.
– Extent of resection
– Progression-free survival
– Recurrent glioblastoma
– Reoperation
– Survival
5-ALA: 5-aminolevulinic acid CI: Confidence interval
ECOG: Eastern Cooperative Oncology Group
EOR: Extent of resection GTR: Gross total resection HR: Hazard ratio
KPS: Karnofsky Performance Status
OS: Overall survival
PFS: Progression-free survival
Division of Neurosurgery, University Hospital La Princesa, Madrid, Spain
To whom correspondence should be addressed: Juan Delgado-Fernandez, M.D. [E-mail: juan.delgado.fdez@gmail.com]
Citation: World Neurosurg. (2017) 108:610-617. https://doi.org/10.1016/j.wneu.2017.09.062
Journal homepage: www.WORLDNEUROSURGERY.org
Available online: www.sciencedirect.com
1878-8750/$ -see front matter ª 2017 Elsevier Inc. All rights reserved.
METHODS
We retrospectively reviewed all patients diagnosed with and treated for de novo glioblastoma at our institution between January 2010 and December 2015, certified after histopathological examination. The hospital’s central database was reviewed for all patients who underwent surgery for intra-axial tumors. Those patients with complete medical records and confirmed glioblastoma, according to World Health Organization criteria, at the first surgery were eligible for the study. Patients who had incomplete records, were lost to follow-up, or underwent biopsy were excluded. The patients lost to follow-up were excluded as well. Patient survival was calculated between the time of surgery and last follow-up in those patients with complete follow-up.
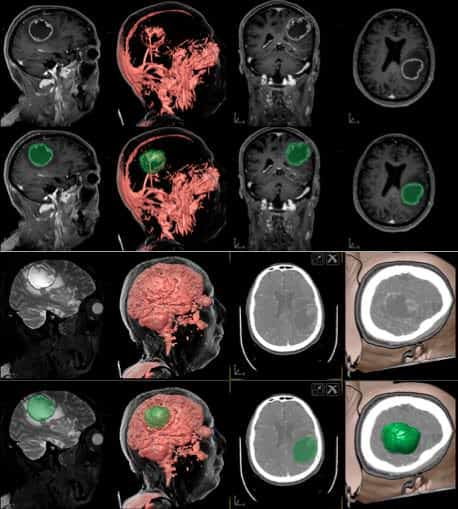
Clinical variables collected included date of birth; sex; age; date of diagnosis; initial symptoms; tumor location; location in an eloquent area; intraoperative use of 5-aminolevulinic acid (5-ALA), carmustine, or neuromonitoring; and volume at diagnosis calculated with the 3 largest diameters in axial and coronal views using the formula for an ellipsoid volume: Volume 4/3p (diameter 1 diameter 2 diameter 3), in cm.3 In our postoperative protocol, all patients underwent postoperative magnetic resonance imaging at 48e72 hours to assess surgical resection, and postoperative volume was calculated again. The percent removal was calculated as follows: (preoperative tumor volume postoperative tumor volume)/preoperative tumor volume. Functional status of the patient was calculated with preoperative and postoperative Karnofsky Performance Scale (KPS) and Eastern Cooperative Oncology Group (ECOG) performance status14 at diagnosis. Date of surgery, complications after surgery, inpatient stay, and in those patients who were reoperated, date of second surgery, postoperative KPS and ECOG scores and the presence of new complications were also recorded. Patients were classified into 3 groups according to the adjuvant therapy received: complete, comprising patients receiving complete radiotherapy and chemotherapy after surgery following the Stupp protocol; only radiotherapy, comprising patients who completed radiotherapy after surgery; and no therapy, comprising patients who did not receive adjuvant therapy or did not complete it. The patients also were classified into 3 groups according to the extent of resection (EOR) at initial surgery: gross total resection (GTR; >.95%), subtotal resection (50%e95%), and partial resection (<.50%). Secondary variables analyzed were KPS score 80 or <.80 and ECOG score <.2 or 2.
OS was defined as the period from the initial diagnosis of the
glioblastoma to the last follow-up or the date of death if registered, and PFS was defined as the period from the initial surgery to the date of recurrence.
Statistical analyses were performed with SPSS 21.0 (IBM, Armonk, New York, USA), with a P value 0.05 considered to indicate statistical significance. Student’s t test and ANOVA were used for quantitative variables, and the c2 test and Fisher’s exact test were used for qualitative variables. OS and PFS were compared using the Kaplan-Meier test, and multivariable analysis was performed using Cox proportional hazards regression with 95% confidence intervals. All variables associated with survival in univariable analysis (P <. 0.10) were included in the multivariable analysis.
![]()
RESULTS
Between January 2010 and December 2015, 157 patients were diagnosed with glioblastoma at our institution. Of these, 31 (20%) received only biopsy as treatment at our department because of deep or inaccessible location, and 5 were lost to follow-up. The remaining 121 patients who underwent initial surgery were divided into 2 groups: those who underwent only 1 surgery (n 90; 75%), and those who underwent 2 or more surgeries (n 31; 25%).
Our patients’ demographic and clinical data are summarized in Table 1. The mean patient age was 62.21 12.34 years, and 71 were male (58.7%). Preclinical status, as defined by ECOG score <.2, was seen in 76 patients (62.8%), and the median preoperative KPS score was 80 (range, 50e90). The most common location was the left temporal lobe (32 patients; 26.4%), followed by the right temporal lobe (27 patients; 22.3%) and the left frontal lobe (17 patients; 14%). Only 22 patients had a tumor located in an eloquent area (18.1%). The mean volume was 310 246 cm3, and the most frequent symptom was headache (33 patients; 27.3%), followed by hemiparesis (21 patients; 17.3%). In 38 patients (31.4%), 5-ALA was administered to guide the intervention, and intraoperative neuromonitoring was applied in 15 patients (12.4%). Gross total resection was achieved in 59 patients (48.8%), and EOR 50% was achieved in 104 patients (86%). Mean postoperative volume was 55.4 88.3 cm3, translating to a mean resection of 82.15% of the lesion. Splitting the cohort into 3 groups, the mean values were as follows: GTR, defined as a mean postoperative volume of 7.04 6.4 cm3 and 97.02 2.4% of mean resection; subtotal resection, defined as a mean postoperative volume of 56.02 47.4 cm3 and 79.95 14.4% of mean resection; and partial resection, defined as a mean postoperative volume of 221.46 118.7 cm3 and 36.36 19.6% of mean resection. Morbidity and mortality occurred in 15 patients who experienced complications after surgery (12.4%), with 3 patients (2.4%) who died due to complications and were excluded from the posterior analysis. The causes of death were acute subdural hematoma in 1 patient and pulmonary embolism in 2 patients. Other complications included hemiparesis in 4 patients (4.4%), acute subdural hematoma in 2 patients (2.2%), and 1 patient each with recurrent seizures, pulmonary thromboembolism, epidural hematoma, wound infection, pseudomeningocele, and refractory edema necessitating decompressive craniotomy. The mean inpatient length of stay was 18 9.6 days. At the end of this retrospective analysis, 111 patients had died (91.7%). Adjuvant therapy following the Stupp protocol, with complete temazolamide and radiotherapy treatment, was administered to 82 patients (67.8%).
Only 3 patients in the reoperation group underwent 3 surgeries
(9.6%). The most common location was the right temporal lobe (8 patients; 25.8%), followed by the left temporal lobe (7 patients; 22.6%). Intraoperative neuromonitoring was performed in 4 patients (13.3%) because of tumor location. Intraoperative agents included 5ALA in 17 patients (54.8%) and carmustine in 10 patients (32.2%). Postoperative complications after second surgery occurred in 19.3% of patients, including 3 patients with cerebrospinal fluid fistula and 3 patients with wound infection.
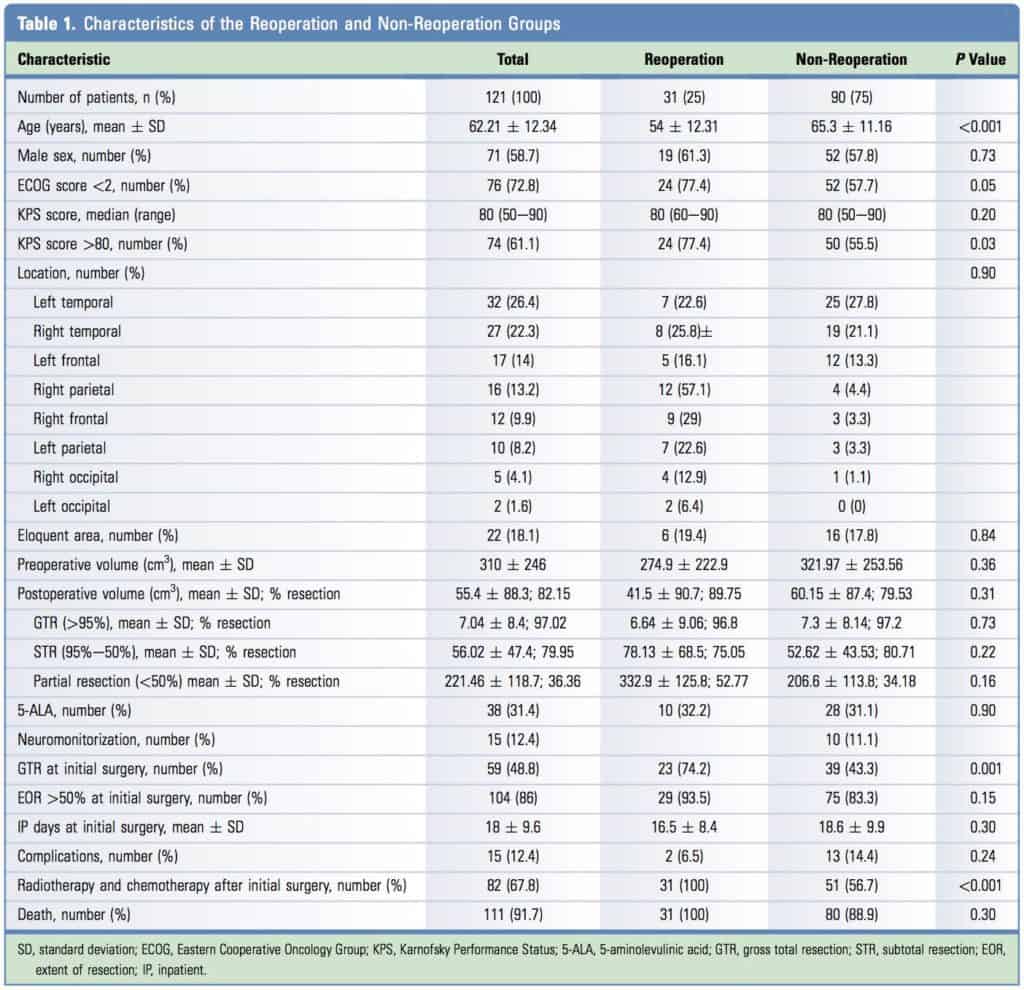
The reoperation group was significantly younger than the non reoperation group (mean age, 54 12.31 years vs. 65.3 11.16 years; P <. 0.001). There was no difference in preoperative status between the 2 groups as measured by the KPS score (P >. 0.05), but better ECOG status was associated with a trend toward better status in the reoperation group (P 0.05). Moreover, the patients with a preoperative KPS score 80 had a greater probability of undergoing reoperation (P <. 0.05). The rate of GTR was significantly higher in the reoperation group (74.2% vs. 43.3%; P 0.001), and complete adjuvant therapy was administered more frequently in the reoperation group (100% vs. 56.7%; P <. 0.001). There were no significant differences in preoperative and
postoperative volumes between the 2 groups. There was a significant trend toward undergoing reoperation for those patients with a postoperative KPS score >80 (P <. 0.01). There was no statistical difference between the patients who experienced complications with the first surgery and those who experienced complications after reintervention (12.4% vs. 19.3%; c2 1.86; P 0.40). There also were no differences in recurrence location in the total cohort or in the reoperated and non-reoperated groups. All the other clinical and demographic data did not show differences between groups.
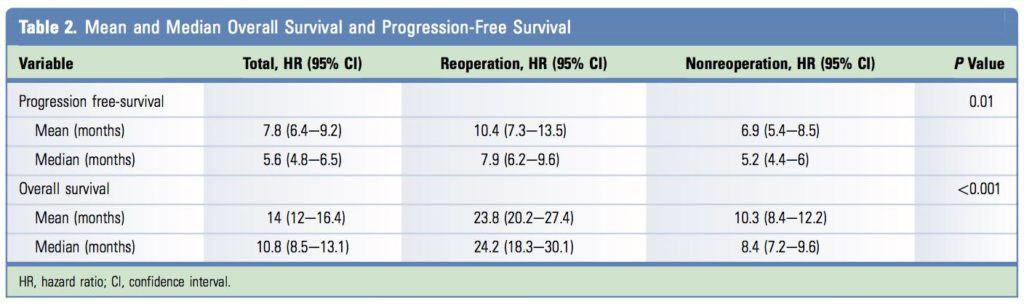
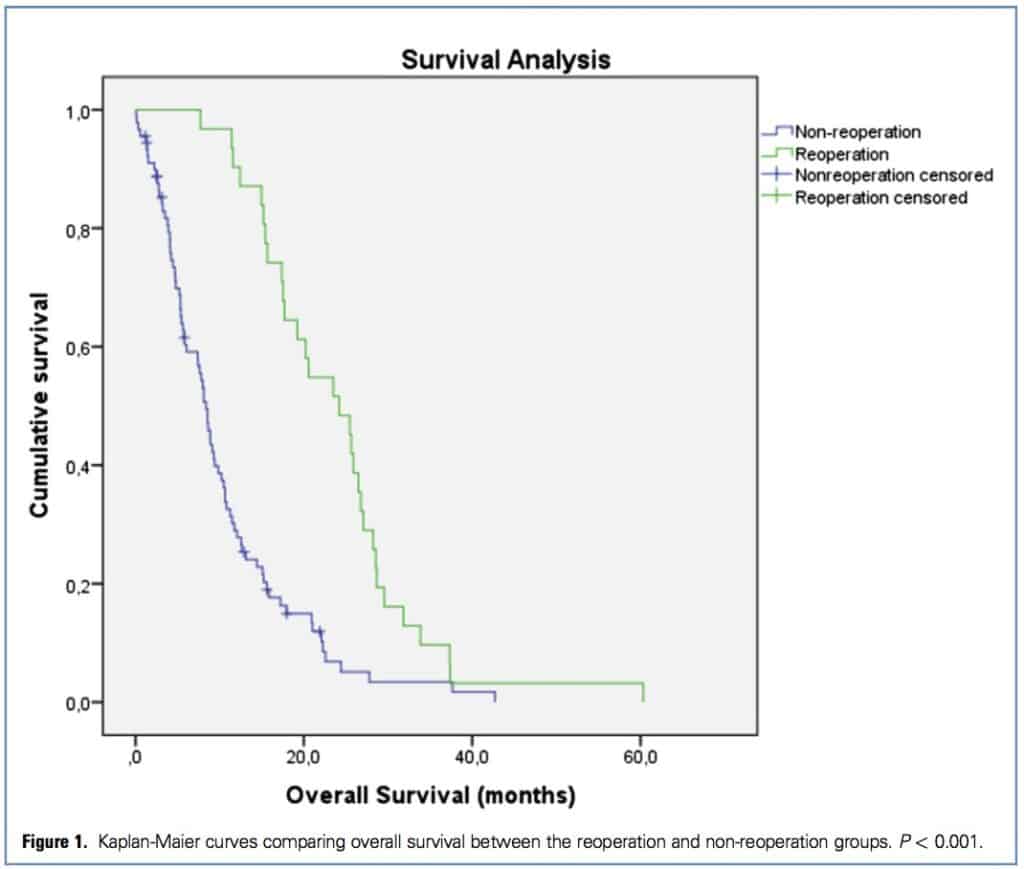
For the entire cohort, the mean and median OS were 14.0 months (95% CI, 12e16.4 months) and 10.8 months (95% CI, 8.5e13.1 months), respectively. In the non-reoperation group, mean and median OS were 10.3 months (95% CI, 8.4e12.2 months) and 8.4 months (95% CI, 7.2e9.6 months), respectively, and in the reoperation group, mean and median OS were 23.8 months (95% CI, 20.2e27.4 months) and 24.2 months (95% CI, 18.3e30.1 months), respectively (P <. 0.001) (Figure 1).
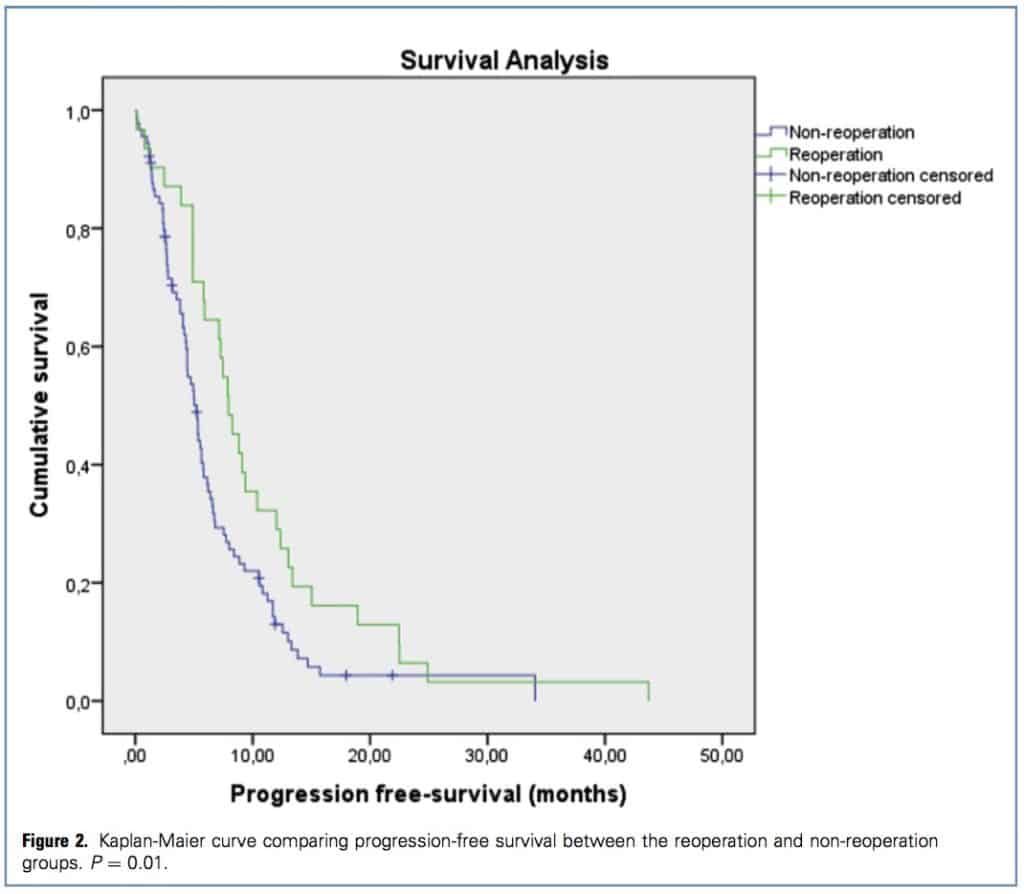
Mean and median PFS were 7.8 months (95% CI, 6.4e9.2 months) and 5.6 months (95% CI, 4.8e6.5 months), respectively, for the entire cohort. There were significant differences in PFS between the non-reoperation and reoperation groups (mean, 6.9 months [95% CI, 5.4e8.5 months] vs. 5.2 months [95% CI, 4.4e6 months]; median, 10.4 months [95% CI, 7.3e13.5 months] vs. 7.9 months [95% CI, 6.2e9.6 months]; P 0.01) (Figure 2). Thus, the difference between groups was 10.5 months and 16.4 months for mean and median OS, respectively, and 3.5 months and 2.7 months for mean and median PFS, respectively (Table 2).
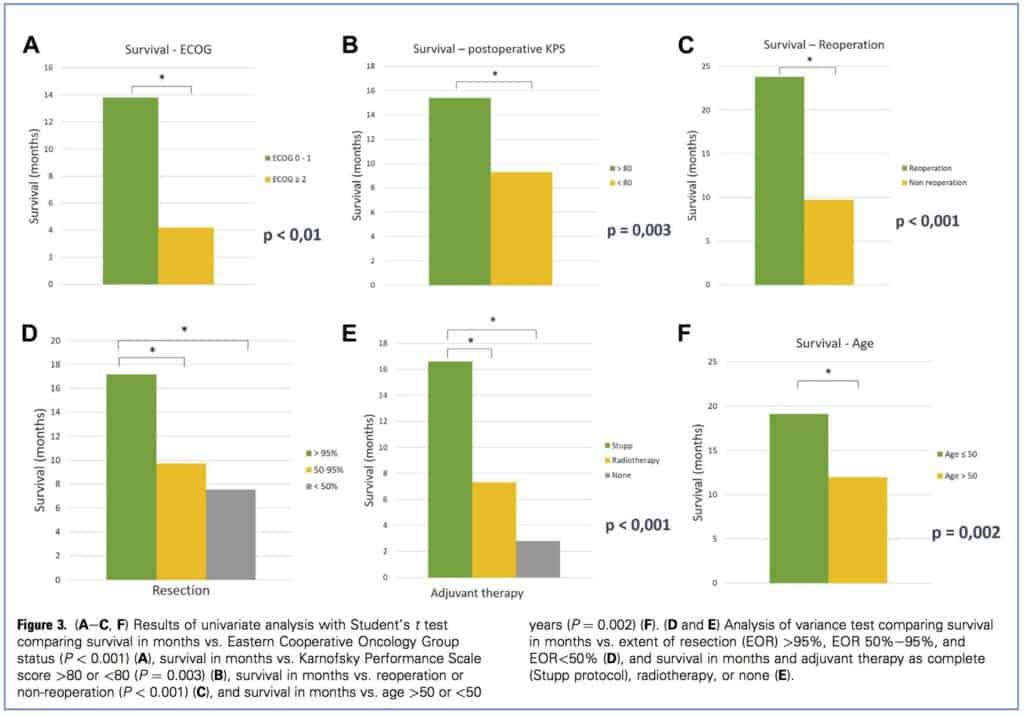
Initially, we performed univariate analysis to identify prognostic factors of higher OS. Postoperative ECOG score <.2 (13.8 months vs.4.2 months; P <. 0.01), postoperative KPS score 80 (15.4 months vs. 9.3 months; P 0.003) and reoperation (23.8 months vs. 9.7 months; P <. 0.001) were associated with better OS. EOR values were divided in 3 groups: >.95%, 50%e95%, and <.50%. Comparing these groups, the EOR >95% group had better OS compared with the 50%e95% group (mean difference, 6.9 T 2 months; P ¼ 0.002) and the <.50% group (mean difference, 9.1 T 2.8 months; P 0.004); there was no difference between the 50%e 95% EOR group and the <.50% EOR group. Adjuvant therapy was analyzed comparing patients who received chemotherapy and radiotherapy administered following the Stupp protocol and those who only received radiotherapy, and the group that did not receive adjuvant therapy. In this case, patients who followed the Stupp protocol had a better OS (9.3 2.2 months over patients who received only radiotherapy and 13.2 2.9 months over patients who did not receive adjuvant therapy; P <. 0.001). Age and postoperative volume showed a negative correlation with OS (Pearson’s r for age 0.429; P <. 0.001). Patients age >.50 years had worse OS than younger patients (12 months vs. 19.1 months; P 0.002) (Figure 3AeE). Postoperative volume also demonstrated a negative correlation with OS (Pearson’s r for postoperative volume 0.214; P 0.02). Finally, we observed a trend toward better survival results in those patients with preoperative KPS score 80 (13.4 months vs. 10.3 months; P 0.07).
The model obtained with Cox multivariable analysis showed a significance level of P <. 0.001, and revealed that age (hazard ratio [HR], 1.03; 95% CI, 1.006e1.055; P 0.013), reoperation (HR, 0.48; 95% CI, 0.285e0.810; P 0.006), EOR >95% (HR,0.547; 95% CI, 0.401e0.748; P <. 0.001) and adjuvant therapy following the Stupp protocol (HR, 0.389; 95% CI, 0.208e0.726; P 0.003) were the variables most closely correlated with a better OS (Table 3).
![]()
DISCUSSION
In recent years, there has been an increased interest in the role of surgery in the treatment of both low and high-grade gliomas. This is due to a lack of consensus on the treatment of recurrence and progression of this lesions.5,15 Our data show that 25% of patients undergo reoperation, which correlates with other previously published clinical series with reintervention rates ranging from 10% to 30%, meaning that roughly 1 of 4 patients are considered for surgery.5,16,17 Prognosis of patients with glioblastoma is still very poor, and median survival is approximately 15 months,6,9,13 which agrees with our present data. However, in recent years, some authors have reported longer survival periods in relation to re-resection and chemoradiotherapy as standardized treatment.12,15,18 On the other hand, it should be noted that in our study the overall complication rate was 12.4%, and the rate was higher in the non-reoperation group compared with the reoperation group after the first surgery (14.4% vs. 6.5%), with no significant difference between the 2 groups (Table 1). This is comparable to data reported in the literature.6,19 Complication rates were not statistically significantly higher in the reoperation group (12.4% vs. 19.3%; c2 1.86; P 0.40), either in the literature or in our data, even in those series with successive reoperations.6,12,15 These findings demonstrate the value of surgery in improving survival with low morbidity.
We observed some differences between our reoperation and non-reoperation groups. In our reoperation group, the patients were younger and in better functional condition according to KPS and ECOG scores, as has also been shown in previously published studies.12,13,18 These between-group differences have led to the hypothesis that the better characteristics of patients in the reoperation group may be more important than the reoperation itself.18,20 However, Tully et al.13 demonstrated in a multivariate analysis that reoperation improves survival independent of other prognostic factors, as we also found in the present series (Table 3). As for prognostic factors that increase patient survival, multiple studies have confirmed, as we found here, that age, functional status as measured by KPS or ECOG score, and the degree of resection are prognostic factors.6,9,13,18,19,21-24 Most published reports agree that preoperative KPS score is a prognostic factor for longer survival6,19,22,23; however, in our series, preoperative KPS score alone was not, but a better general postoperative status as measured by ECOG or KPS score, was correlated with better OS. This has also been reported by Bloch et al.,9 and it is readily apparent how a better postoperative status before recurrence would benefit OS and PFS. Although these characteristics appear to be the most frequently cited in the literature, others that were not correlated with prognosis in our study include volume,21-23 location in a noneloquent area,22 presence of a lesion with cystic component,21 and successive reoperations.12 Our multivariable analysis did not show a correlation for volumen, because our method for volume analysis was not as accurate as used in other studies.25 However, although volume has been reported as a risk factor for shorter survival,22,23 published data do not agree on the implications of residual volume.13,18 For example, Bloch et al.9 reported no difference between total and subtotal resection as the first procedure, obtaining the best prognosis when complete resection was performed during the reintervention. Nevertheless, they recommended obtaining a complete resection initially because of the longer survival in the patients who underwent complete resection in both procedures. In addition, we found no correlation between recurrence and location; however, this factor has not been frequently related with survival,22 and a recent study of 1229 lowand high-grade gliomas showed no correlation between eloquent or non-eloquent areas and survival,21 as reported previously.9,19 Finally, some recent articles have reported improved OS with multiple resections. Chaichana et al.12 retrospectively studied 578 patients from Johns Hopkins Hospital and found improvements in OS of 6.8, 15.5, 22.4, and 26.6 months in patients who underwent 1, 2, 3, and 4 surgeries, respectively. This represented a relative risk (RR) of shortened survival of 3.4, compared with patients who underwent 2 surgeries (RR 0.688), 3 surgeries (RR 0. 614), or 4 surgeries (RR 0.600). Similar results have been published by Ringel et al.,15 showing the best survival in patients with additional re-resections.
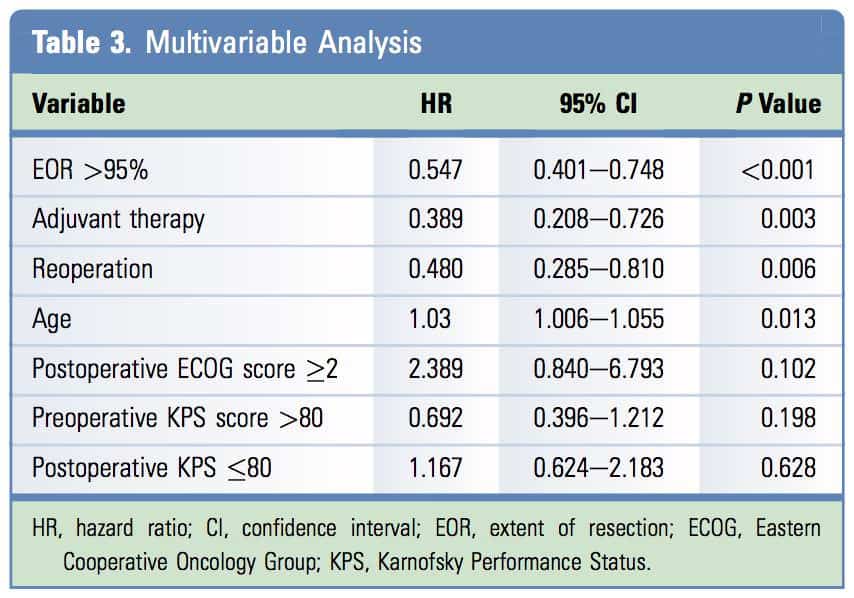
Several recent reports have established that the extent of resection is one of the most valuable prognostic factors for glioblastoma. Lacroix et al.24 were the first to show that both resection and its extension influenced survival. Analyzing the results from 416 patients treated for glioblastomas, the authors showed an increase in survival with resections >98%. Subsequently, Sanai et al.,23 in a series of 500 patients, concluded that resection of at least 78% of the tumor is necessary to significantly increase survival. That same group compared patients who underwent reoperation by checking for differences in survival between patients who had undergone initial complete resection followed by a second surgery with another complete resection or subtotal resection and subsequently another incomplete or a complete resection had been achieved. The authors concluded that although survival was best in the first case, there were no differences when a complete resection was obtained in the reintervention.9 Faced with this, our data do show that complete and >.95% resections are associated with increased survival, whereas 50%e95% resections do not improve survival. However, we could not provide a more accurate determination of the minimum volume required to increase survival. This contrasts with the data recently published by Tully et al.13 reporting a benefit of >50% resection, compared with data previously published by Sanai et al.19,23 Current surgical treatments for glioblastomas are becoming more aggressive and the goal should be to do an initial complete surgical resection as this will result in increased survival. In relation to this hypothesis, the resection of proximal areas to the tumor where it could spread are identified as hyperintensity regions on magnetic resonance imaging fluid-attenuated inversion recovery (FLAIR) sequences, as has been recently reported by Li et al.21 in low-grade gliomas. Those authors showed increased survival in patients treated by resection of >.53.2% of the volume as shown in FLAIR sequences in the first surgery with respect to those whose resection, was less that this percentage without presenting a higher incidence of complications, which has been verified previously by other groups.19,21 Finally, in addition to the degree of resection, which in our study demonstrated a protective effect in our multivariate model (HR, 0547; 95% CI, 0.401e0.748; P <. 0.001), other significant factors were age (HR, 1.03; 95% CI, 1.006e1.055; P 0.013), treatment with the Stupp protocol (HR, 0.389; 95% CI, 0.208e0.726; P 0.003), and reoperation (HR, 0.48; 95% CI, 0.285e0.810; ;P 0.006). Similar results have been shown in previously published series6,9,13,18,19,21-24; however, we did not observe correlations with preoperative functional status (KPS or ECOG score) or preoperative recurrence,6,9,19,21-24 volume,22,23 sex, location,22 presence of necrosis,21,24 or presence of cysts,21 as have been reported previously.
Our study has 2 main limitations. First, its retrospective design makes it more difficult to obtain stronger conclusions and to obtain a homogeneous group for analysis, making standardization of postoperative treatment a challenge. In our series, it is remarkable that only 68% of the patients received the Stupp protocol, which has already shown high efficacy. Second is the absence of analysis of O6-methylguanine-methyltransferase and isocitrate dehydrogenase 1 status in our patients. Recent literature, mainly the new World Health Organization classification of gliomas,26 has identified these as 2 main prognostic factors that may alter our approach to choosing treatment for our patients. These new molecular findings are meant to change the prognosis and management of high-grade gliomas, and new studies taking these factors into account are needed to expand our knowledge of how reintervention can improve survival in these patients. However, for our retrospective analysis, all of this information was not available in our database, mainly for the early cases.
Finally, radiosurgery also has been proposed for unresected residual or progressive high-grade gliomas,7 and has been also compared with patients who underwent reoperation,27 concluding that it is too soon to make a general recommendation, and more studies are needed. However, gamma knife surgery and radiosurgery are options for recurrent small glioblastomas, with a lower complication rate, possibly providing a new approach for some patients.
![]()
CONCLUSION
In conclusion, patients with recurrent glioblastoma will benefit from reoperation, increasing their OS and PFS. Extent of resection remains one of the main prognostic factors for improved survival in high-grade gliomas, and new studies are proving that the resection of peritumoral regions where the tumor is already spreading will improve survival without increasing morbidity. Other prognostic factors that will ameliorate survival in our patients are young age and maximal adjuvant therapy from initial diagnosis. However, not all patients will be eligible for reoperation, and new therapies, such as gamma knife surgery, can provide a second resection in those cases.
REFERENCES
1. Dolecek TA, Propp JM, Stroup NE, Kruchko C. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2005-2009. Neuro Oncol. 2012; 14(Suppl 5):v1-v49.
2. Preusser M, de Ribaupierre S, Wöhrer A, Erridge SC, Hegi M, Weller M, et al. Current concepts and management of glioblastoma. Ann Neurol. 2011;70:9-21.
3. Ohgaki H, Kleihues P. Epidemiology and etiology of gliomas. Acta Neuropathol. 2005;109:93-108.
4. Stupp R, Mason W, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352:987-996.
5. Weller M, Cloughesy T, Perry JR, Wick W. Standards of care for treatment of recurrent glioblastoma: are we there yet? Neuro Oncol. 2013;15:4-27.
6. Hervey-Jumper SL, Berger MS. Reoperation for recurrent high-grade glioma: a current perspective of the literature. Neurosurgery. 2014;75:491-499 [discussion: 498-499].
7. Niranjan A, Kano H, Iyer A, Kondziolka D, Flickinger JC, Lunsford LD. Role of adjuvant or salvage radiosurgery in the management of unresected residual or progressive glioblastoma multiforme in the pre-bevacizumab era. J Neurosurg. 2015;122:757-765.
8. Pool JL. The management of recurrent gliomas.
Clin Neurosurg. 1968;15:265-287.
9. Bloch O, Han SJ, Cha S, Sun MZ, Aghi MK, McDermott MW, et al. Impact of extent of resection for recurrent glioblastoma on overall survival: clinical article. J Neurosurg. 2012;117: 1032-1038.
10. Clark AJ, Butowski NA, Chang SM, Prados MD, Clarke J, Polley MY, et al. Impact of bevacizumab chemotherapy on craniotomy wound healing. J Neurosurg. 2011;114:1609-1616.
11. Clark AJ, Lamborn KR, Butowski NA, Chang SM, Prados MD, Clarke JL, et al. Neurosurgical management and prognosis of patients with glioblastoma that progresses during bevacizumab treatment. Neurosurgery. 2012;70:361-370.
12. Chaichana KL, Zadnik P, Weingart JD, Olivi A, Gallia GL, Blakeley J, et al. Multiple resections for patients with glioblastoma: prolonging survival. J Neurosurg. 2013;118:812-820.
13. Tully PA, Gogos AJ, Love C, Liew D, Drummond KJ, Morokoff AP. Reoperation for recurrent glioblastoma and its association with survival benefit. Neurosurgery. 2016;79:678-689.
14. Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649-655.
15. Ringel F, Pape H, Sabel M, Krex D, Bock HC, Misch M, et al. Clinical benefit from resection of recurrent glioblastomas: results of a multicenter study including 503 patients with recurrent glioblastomas undergoing surgical resection. Neuro Oncol. 2016;18:96-104.
16. Barbagallo GM, Jenkinson MD, Brodbelt AR. “Recurrent” glioblastoma multiforme, when should we reoperate? Br J Neurosurg. 2008;22: 452-455.
17. Gorlia T, Stupp R, Brandes AA, Rampling RR, Fumoleau P, Dittrich C, et al. New prognostic factors and calculators for outcome prediction in patients with recurrent glioblastoma: a pooled analysis of EORTC Brain Tumour Group phase I and II clinical trials. Eur J Cancer. 2012;48: 1176-1184.
18. Ortega A, Sarmiento JM, Ly D, Nuño M, Mukherjee D, Black KL, et al. Multiple resections and survival of recurrent glioblastoma patients in the temozolomide era. J Clin Neurosci. 2016;24: 105-111.
19. Oppenlander ME, Wolf AB, Snyder LA, Bina R, Wilson JR, Coons SW, et al. An extent of resection threshold for recurrent glioblastoma and its risk for neurological morbidity. J Neurosurg. 2014;120: 846-853.
20. Franceschi E, Bartolotti M, Tosoni A, Bartolini S, Sturiale C, Fioravanti A, et al. The effect of reoperation on survival in patients with recurrent glioblastoma. Anticancer Res. 2015;35:1743-1748.
21.
Li YM, Suki D, Hess K, Sawaya R. The influence of maximum safe resection of glioblastoma on sur vival in 1229 patients: can we do better than grosstotal resection? J Neurosurg. 2016;124:977-988.
22. Yong RL, Wu T, Mihatov N, Shen MJ, Brown MA, Zaghloul KA, et al. Residual tumor volume and patient survival following reoperation for recur rent glioblastoma. J Neurosurg. 2014;121:802-809.
23. Sanai N, Polley MY, McDermott MW, Parsa AT, Berger MS. An extent of resection threshold for newly diagnosed glioblastomas. J Neurosurg. 2011; 115:3-8.
24. Lacroix M, Abi-Said D, Fourney DR, Gokaslan ZL, Shi W, DeMonte F, et al. A multivariate analysis of
416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg. 2001;95:190-198.
25. Meier R, Porz N, Knecht U, Loosli T, Schucht P, Beck J, et al. Automatic estimation of extent of resection and residual tumor volume of patients with glioblastoma. J Neurosurg. 2017;127:798-806.
26. Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, et al. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol. 2016;131: 803-820.
27. Skeie BS, Enger PØ, Brøgger J, Ganz JC, Thorsen F, Heggdal JI, et al. Gamma knife surgery versus reoperation for recurrent glioblastoma multiforme. World Neurosurg. 2012;78:658-669.
Conflict of interest statement: The authors declare that the article content was composed in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Received 25 July 2017; accepted 9 September 2017
Citation: World Neurosurg. (2017) 108:610-617. https://doi.org/10.1016/j.wneu.2017.09.062
Journal homepage: www.WORLDNEUROSURGERY.org
Available online: www.sciencedirect.com
1878-8750/$ see front matter ª 2017 Elsevier Inc. All rights reserved.